Abstract
Free full text

Expression of striatal D1 dopamine receptors coupled to inositol phosphate production and Ca2+ mobilization in Xenopus oocytes.
Abstract
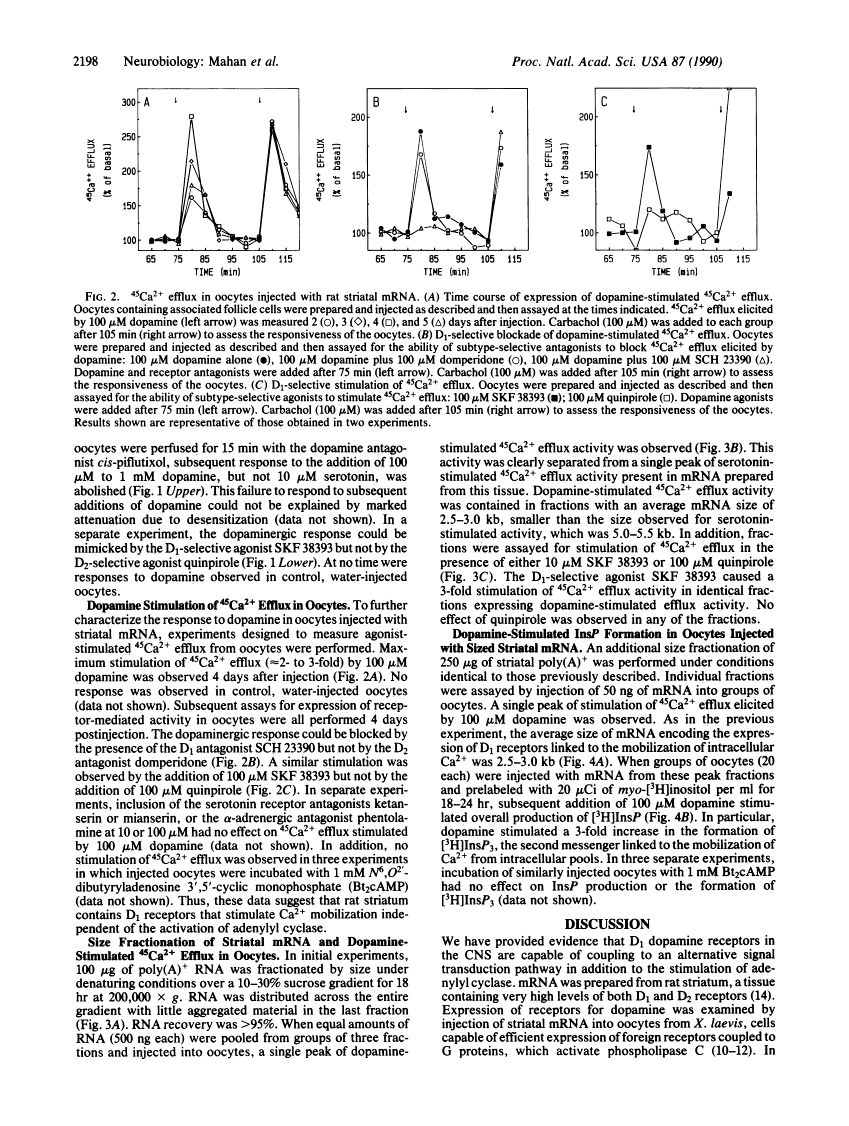
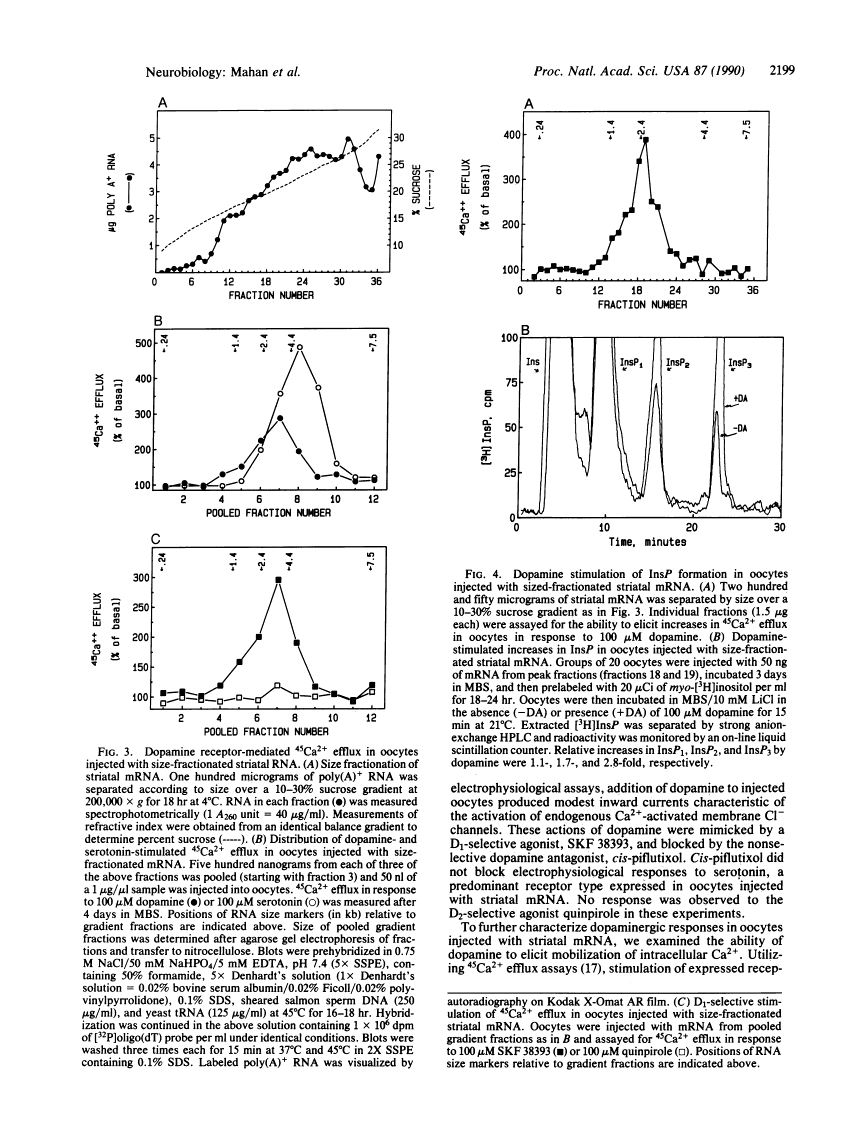
Expression of central nervous system receptors for dopamine was examined by injection of poly(A)+ RNA (mRNA) from rat striatum into oocytes from Xenopus laevis. Electrophysiological measurements in mRNA-injected oocytes indicated that addition of 100 microM dopamine induced an inward current (40-100 nA) that was consistent with the activation of endogenous Ca2(+)-dependent Cl- channels. This current was also elicited by addition of the selective D1 agonist SKF 38393 but not by the selective D2 agonist quinpirole. Prior addition of the dopaminergic antagonist cis-piflutixol completely abolished dopamine-induced currents but had no effect on currents produced by serotonin. Using 45Ca2+ efflux assays, addition of 100 microM dopamine to injected oocytes stimulated efflux 2- to 3-fold. This increase was mimicked by SKF 38393 and was blocked by the D1-selective antagonist (+)SCH 23390 but not by the D2-selective antagonist domperidone. No increase in 45Ca2+ efflux was seen with 100 microM quinpirole. Size fractionation of striatal mRNA yielded a single peak (2.5-3.0 kilobases) of D1 receptor-mediated 45Ca2+ efflux activity in injected oocytes. In addition, dopamine stimulation of oocytes injected with peak fractions and prelabeled with myo-[3H]inositol caused a 3-fold increase in [3H]inositol 1,4,5-triphosphate [( 3H]InsP3) formation. No effect on [3H]InsP3 production or 45Ca2+ efflux was observed, however, in injected oocytes incubated with 1 mM N6,O2'-dibutyryladenosine 3',5'-cyclic monophosphate. Thus, in addition to D1 receptors that stimulate adenylyl cyclase, rat striatum contains D1 receptors that can couple to InsP3 formation and mobilization of intracellular Ca2+.
Full text
Full text is available as a scanned copy of the original print version. Get a printable copy (PDF file) of the complete article (1.1M), or click on a page image below to browse page by page. Links to PubMed are also available for Selected References.
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Freissmuth M, Casey PJ, Gilman AG. G proteins control diverse pathways of transmembrane signaling. FASEB J. 1989 Aug;3(10):2125–2131. [Abstract] [Google Scholar]
- Creese I, Sibley DR, Hamblin MW, Leff SE. The classification of dopamine receptors: relationship to radioligand binding. Annu Rev Neurosci. 1983;6:43–71. [Abstract] [Google Scholar]
- Freedman JE, Weight FF. Single K+ channels activated by D2 dopamine receptors in acutely dissociated neurons from rat corpus striatum. Proc Natl Acad Sci U S A. 1988 May;85(10):3618–3622. [Europe PMC free article] [Abstract] [Google Scholar]
- Vallar L, Meldolesi J. Mechanisms of signal transduction at the dopamine D2 receptor. Trends Pharmacol Sci. 1989 Feb;10(2):74–77. [Abstract] [Google Scholar]
- Bigornia L, Suozzo M, Ryan KA, Napp D, Schneider AS. Dopamine receptors on adrenal chromaffin cells modulate calcium uptake and catecholamine release. J Neurochem. 1988 Oct;51(4):999–1006. [Abstract] [Google Scholar]
- Felder CC, Jose PA, Axelrod J. The dopamine-1 agonist, SKF 82526, stimulates phospholipase-C activity independent of adenylate cyclase. J Pharmacol Exp Ther. 1989 Jan;248(1):171–175. [Abstract] [Google Scholar]
- Felder CC, Blecher M, Jose PA. Dopamine-1-mediated stimulation of phospholipase C activity in rat renal cortical membranes. J Biol Chem. 1989 May 25;264(15):8739–8745. [Abstract] [Google Scholar]
- Williams JA, McChesney DJ, Calayag MC, Lingappa VR, Logsdon CD. Expression of receptors for cholecystokinin and other Ca2+-mobilizing hormones in Xenopus oocytes. Proc Natl Acad Sci U S A. 1988 Jul;85(13):4939–4943. [Europe PMC free article] [Abstract] [Google Scholar]
- McIntosh RP, Catt KJ. Coupling of inositol phospholipid hydrolysis to peptide hormone receptors expressed from adrenal and pituitary mRNA in Xenopus laevis oocytes. Proc Natl Acad Sci U S A. 1987 Dec;84(24):9045–9048. [Europe PMC free article] [Abstract] [Google Scholar]
- Snutch TP. The use of Xenopus oocytes to probe synaptic communication. Trends Neurosci. 1988 Jun;11(6):250–256. [Abstract] [Google Scholar]
- Kaneko S, Kato K, Yamagishi S, Sugiyama H, Nomura Y. GTP-binding proteins Gi and Go transplanted onto Xenopus oocyte by rat brain messenger RNA. Brain Res. 1987 Dec;427(1):11–19. [Abstract] [Google Scholar]
- Okayama H, Kawaichi M, Brownstein M, Lee F, Yokota T, Arai K. High-efficiency cloning of full-length cDNA; construction and screening of cDNA expression libraries for mammalian cells. Methods Enzymol. 1987;154:3–28. [Abstract] [Google Scholar]
- Aviv H, Leder P. Purification of biologically active globin messenger RNA by chromatography on oligothymidylic acid-cellulose. Proc Natl Acad Sci U S A. 1972 Jun;69(6):1408–1412. [Europe PMC free article] [Abstract] [Google Scholar]
- Sumikawa K, Houghton M, Smith JC, Bell L, Richards BM, Barnard EA. The molecular cloning and characterisation of cDNA coding for the alpha subunit of the acetylcholine receptor. Nucleic Acids Res. 1982 Oct 11;10(19):5809–5822. [Europe PMC free article] [Abstract] [Google Scholar]
- Tu CP, Cohen SN. 3'-end labeling of DNA with [alpha-32P]cordycepin-5'-triphosphate. Gene. 1980 Jul;10(2):177–183. [Abstract] [Google Scholar]
- Christensen AV, Arnt J, Hyttel J, Larsen JJ, Svendsen O. Pharmacological effects of a specific dopamine D-1 antagonist SCH 23390 in comparison with neuroleptics. Life Sci. 1984 Apr 16;34(16):1529–1540. [Abstract] [Google Scholar]
- Hoyer D. Molecular pharmacology and biology of 5-HT1C receptors. Trends Pharmacol Sci. 1988 Mar;9(3):89–94. [Abstract] [Google Scholar]
- Jope RS, Li X. Inhibition of inositol phospholipid synthesis and norepinephrine-stimulated hydrolysis in rat brain slices by excitatory amino acids. Biochem Pharmacol. 1989 Feb 15;38(4):589–596. [Abstract] [Google Scholar]
- Bunzow JR, Van Tol HH, Grandy DK, Albert P, Salon J, Christie M, Machida CA, Neve KA, Civelli O. Cloning and expression of a rat D2 dopamine receptor cDNA. Nature. 1988 Dec 22;336(6201):783–787. [Abstract] [Google Scholar]
- Monsma FJ, Jr, McVittie LD, Gerfen CR, Mahan LC, Sibley DR. Multiple D2 dopamine receptors produced by alternative RNA splicing. Nature. 1989 Dec 21;342(6252):926–929. [Abstract] [Google Scholar]
- Boyson SJ, McGonigle P, Molinoff PB. Quantitative autoradiographic localization of the D1 and D2 subtypes of dopamine receptors in rat brain. J Neurosci. 1986 Nov;6(11):3177–3188. [Abstract] [Google Scholar]
- Battaglia G, Norman AB, Hess EJ, Creese I. D2 dopamine receptor-mediated inhibition of forskolin-stimulated adenylate cyclase activity in rat striatum. Neurosci Lett. 1985 Aug 30;59(2):177–182. [Abstract] [Google Scholar]
- Battaglia G, Norman AB, Hess EJ, Creese I. Forskolin potentiates the stimulation of rat striatal adenylate cyclase mediated by D-1 dopamine receptors, guanine nucleotides, and sodium fluoride. J Neurochem. 1986 Apr;46(4):1180–1185. [Abstract] [Google Scholar]
- Mailman RB, Schulz DW, Kilts CD, Lewis MH, Rollema H, Wyrick S. The multiplicity of the D1 dopamine receptor. Adv Exp Med Biol. 1986;204:53–72. [Abstract] [Google Scholar]
- De Keyser J, Dierckx R, Vanderheyden P, Ebinger G, Vauquelin G. D1 dopamine receptors in human putamen, frontal cortex and calf retina: differences in guanine nucleotide regulation of agonist binding and adenylate cyclase stimulation. Brain Res. 1988 Mar 8;443(1-2):77–84. [Abstract] [Google Scholar]
- Watling KJ, Dowling JE. Dopaminergic mechanisms in the teleost retina. I. Dopamine-sensitive adenylate cyclase in homogenates of carp retina; effects of agonists, antagonists, and ergots. J Neurochem. 1981 Feb;36(2):559–568. [Abstract] [Google Scholar]
- Bonner TI. The molecular basis of muscarinic receptor diversity. Trends Neurosci. 1989 Apr;12(4):148–151. [Abstract] [Google Scholar]
- Hartig PR. Molecular biology of 5-HT receptors. Trends Pharmacol Sci. 1989 Feb;10(2):64–69. [Abstract] [Google Scholar]
- Kobilka BK, Matsui H, Kobilka TS, Yang-Feng TL, Francke U, Caron MG, Lefkowitz RJ, Regan JW. Cloning, sequencing, and expression of the gene coding for the human platelet alpha 2-adrenergic receptor. Science. 1987 Oct 30;238(4827):650–656. [Abstract] [Google Scholar]
- Regan JW, Kobilka TS, Yang-Feng TL, Caron MG, Lefkowitz RJ, Kobilka BK. Cloning and expression of a human kidney cDNA for an alpha 2-adrenergic receptor subtype. Proc Natl Acad Sci U S A. 1988 Sep;85(17):6301–6305. [Europe PMC free article] [Abstract] [Google Scholar]
- Cotecchia S, Schwinn DA, Randall RR, Lefkowitz RJ, Caron MG, Kobilka BK. Molecular cloning and expression of the cDNA for the hamster alpha 1-adrenergic receptor. Proc Natl Acad Sci U S A. 1988 Oct;85(19):7159–7163. [Europe PMC free article] [Abstract] [Google Scholar]
Associated Data
Articles from Proceedings of the National Academy of Sciences of the United States of America are provided here courtesy of National Academy of Sciences
Full text links
Read article at publisher's site: https://doi.org/10.1073/pnas.87.6.2196
Read article for free, from open access legal sources, via Unpaywall:
https://europepmc.org/articles/pmc53653?pdf=render
Citations & impact
Impact metrics
Citations of article over time
Alternative metrics
Article citations
Dopamine internalization via Uptake<sub>2</sub> and stimulation of intracellular D<sub>5</sub>-receptor-dependent calcium mobilization and CDP-diacylglycerol signaling.
Front Pharmacol, 15:1422998, 25 Oct 2024
Cited by: 0 articles | PMID: 39525629 | PMCID: PMC11543475
Effects of DPTQ, a novel positive allosteric modulator of the dopamine D1 receptor, on spontaneous eye blink rate and spatial working memory in the nonhuman primate.
Psychopharmacology (Berl), 240(5):1033-1048, 24 Mar 2023
Cited by: 0 articles | PMID: 36961560 | PMCID: PMC10102062
Neurotransmitters, neuropeptides and calcium in oocyte maturation and early development.
Front Cell Dev Biol, 10:980219, 23 Sep 2022
Cited by: 5 articles | PMID: 36211465 | PMCID: PMC9537470
Review Free full text in Europe PMC
Dopamine D1/D5 Receptor Signaling Is Involved in Arrhythmogenesis in the Setting of Takotsubo Cardiomyopathy.
Front Cardiovasc Med, 8:777463, 04 Feb 2022
Cited by: 6 articles | PMID: 35187102 | PMCID: PMC8855058
Functional Selectivity of Dopamine D1 Receptor Signaling: Retrospect and Prospect.
Int J Mol Sci, 22(21):11914, 03 Nov 2021
Cited by: 8 articles | PMID: 34769344 | PMCID: PMC8584964
Review Free full text in Europe PMC
Go to all (105) article citations
Data
Similar Articles
To arrive at the top five similar articles we use a word-weighted algorithm to compare words from the Title and Abstract of each citation.
Dopamine receptors expressed in the Xenopus oocytes injected with bovine striatal mRNA.
Brain Res Mol Brain Res, 7(2):183-187, 01 Feb 1990
Cited by: 6 articles | PMID: 2160046
Stimulation of a dopamine D1 receptor enhances inositol phosphates formation in rat brain.
J Pharmacol Exp Ther, 253(3):987-992, 01 Jun 1990
Cited by: 127 articles | PMID: 1972756
Dopamine D2 receptor stimulation inhibits inositol phosphate generating system in rat striatal slices.
Brain Res, 456(2):235-240, 01 Jul 1988
Cited by: 38 articles | PMID: 2974746
[Studies on the transmembrane signalling mechanism of the brain using Xenopus oocytes].
Yakugaku Zasshi, 110(5):304-314, 01 May 1990
Cited by: 0 articles | PMID: 2165528
Review