Abstract
Free full text

Cloning, partial sequence, expression, and antigenic analysis of the filamentous hemagglutinin gene of Bordetella pertussis.
Abstract
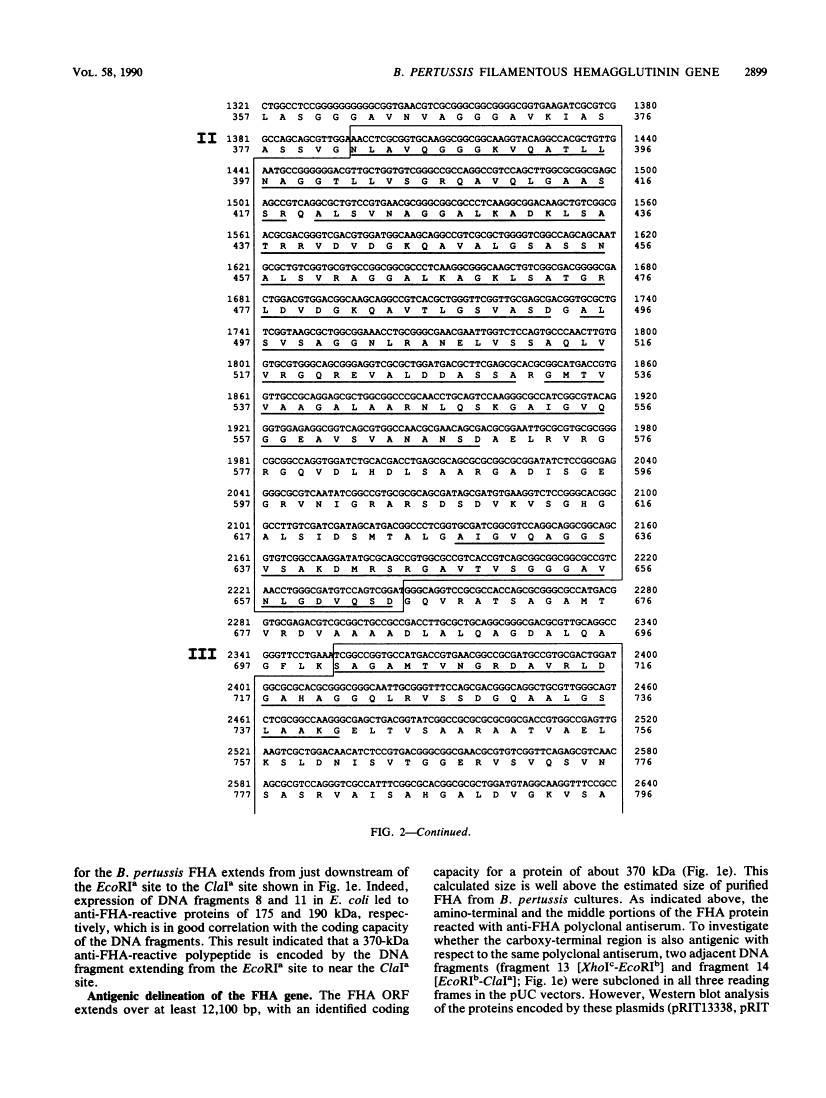
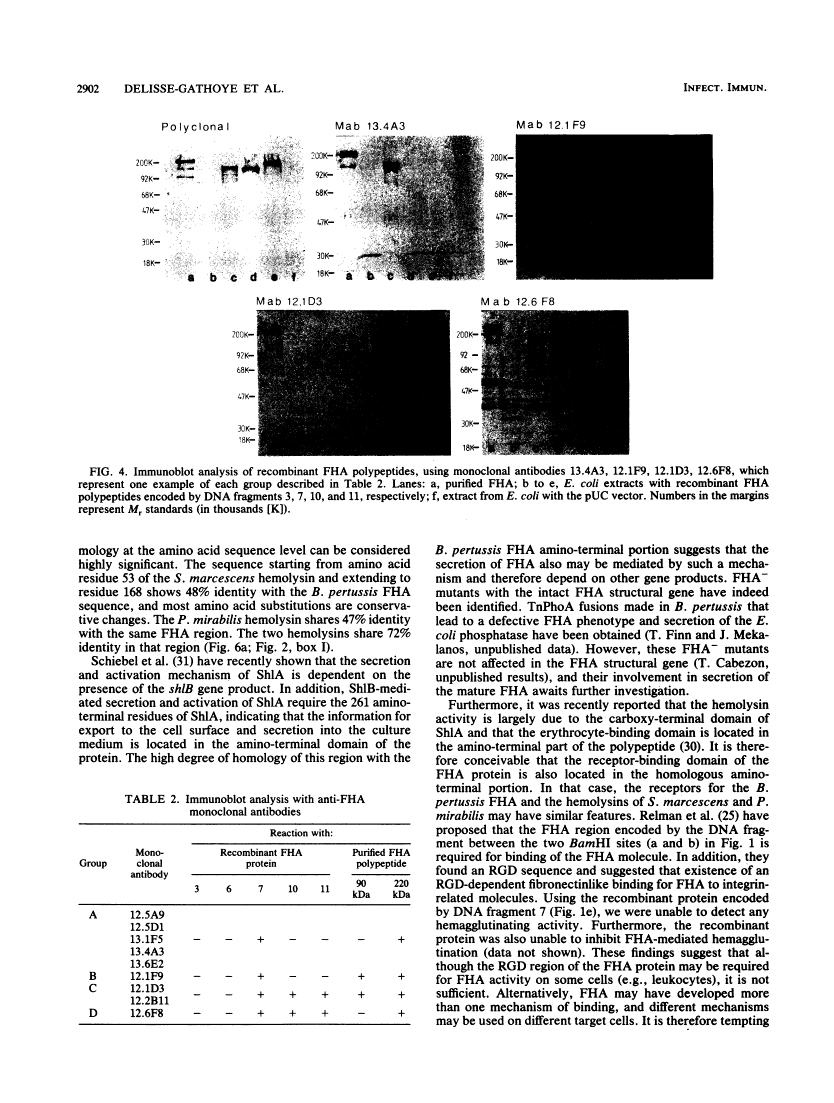
The gene coding for the filamentous hemagglutinin (FHA), one of the main factors involved in mediating adherence of Bordetella pertussis to ciliated host cells, was cloned in Escherichia coli, and the 3,500-base-pair nucleotide sequence encoding the amino-terminal region was determined. Molecular cloning, together with the characterization of recombinant FHA-related proteins produced in E. coli, revealed that the primary translation product is a protein of about 370 kilodaltons (kDa). The mature 220-kDa FHA polypeptide secreted by B. pertussis is most probably generated by proteolytic processing that eliminates a carboxy-terminal portion of about 150 kDa. The 1,087 amino-terminal residues of the predicted FHA sequence showed a number of remarkable features. Extensive homology to the Serratia marcescens and Proteus mirabilis hemolysin proteins was found between amino acids 91 and 205 of the FHA sequence, suggesting involvement of this FHA domain in host cell binding or secretion of FHA from B. pertussis. In addition, two regions containing repetitive amino acid sequences were identified. One region, extending from residues 382 to 664, was formed by six repeats, and a second, extending from residues 701 to 912, contained three repeats. The reactivities of several recombinant FHA-derived proteins with a panel of monoclonal antibodies identified at least four epitopes composing an immunoreactive domain present in the carboxy-terminal moiety of the mature FHA.
Full text
Full text is available as a scanned copy of the original print version. Get a printable copy (PDF file) of the complete article (2.2M), or click on a page image below to browse page by page. Links to PubMed are also available for Selected References.
Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Barbieri JT, Rappuoli R, Collier RJ. Expression of the S-1 catalytic subunit of pertussis toxin in Escherichia coli. Infect Immun. 1987 May;55(5):1321–1323. [Europe PMC free article] [Abstract] [Google Scholar]
- Brown DR, Parker CD. Cloning of the filamentous hemagglutinin of Bordetella pertussis and its expression in Escherichia coli. Infect Immun. 1987 Jan;55(1):154–161. [Europe PMC free article] [Abstract] [Google Scholar]
- Chen EY, Seeburg PH. Supercoil sequencing: a fast and simple method for sequencing plasmid DNA. DNA. 1985 Apr;4(2):165–170. [Abstract] [Google Scholar]
- Devereux J, Haeberli P, Smithies O. A comprehensive set of sequence analysis programs for the VAX. Nucleic Acids Res. 1984 Jan 11;12(1 Pt 1):387–395. [Europe PMC free article] [Abstract] [Google Scholar]
- Duffaud G, Inouye M. Signal peptidases recognize a structural feature at the cleavage site of secretory proteins. J Biol Chem. 1988 Jul 25;263(21):10224–10228. [Abstract] [Google Scholar]
- Fickett JW. Recognition of protein coding regions in DNA sequences. Nucleic Acids Res. 1982 Sep 11;10(17):5303–5318. [Europe PMC free article] [Abstract] [Google Scholar]
- Glaser P, Ladant D, Sezer O, Pichot F, Ullmann A, Danchin A. The calmodulin-sensitive adenylate cyclase of Bordetella pertussis: cloning and expression in Escherichia coli. Mol Microbiol. 1988 Jan;2(1):19–30. [Abstract] [Google Scholar]
- Hanna Z, Fregeau C, Préfontaine G, Brousseau R. Construction of a family of universal expression plasmid vectors. Gene. 1984 Oct;30(1-3):247–250. [Abstract] [Google Scholar]
- Irons LI, Ashworth LA, Wilton-Smith P. Heterogeneity of the filamentous haemagglutinin of Bordetella pertussis studied with monoclonal antibodies. J Gen Microbiol. 1983 Sep;129(9):2769–2778. [Abstract] [Google Scholar]
- Knapp S, Mekalanos JJ. Two trans-acting regulatory genes (vir and mod) control antigenic modulation in Bordetella pertussis. J Bacteriol. 1988 Nov;170(11):5059–5066. [Europe PMC free article] [Abstract] [Google Scholar]
- Laemmli UK. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. [Abstract] [Google Scholar]
- Locht C, Barstad PA, Coligan JE, Mayer L, Munoz JJ, Smith SG, Keith JM. Molecular cloning of pertussis toxin genes. Nucleic Acids Res. 1986 Apr 25;14(8):3251–3261. [Europe PMC free article] [Abstract] [Google Scholar]
- Locht C, Cieplak W, Marchitto KS, Sato H, Keith JM. Activities of complete and truncated forms of pertussis toxin subunits S1 and S2 synthesized by Escherichia coli. Infect Immun. 1987 Nov;55(11):2546–2553. [Europe PMC free article] [Abstract] [Google Scholar]
- Locht C, Keith JM. Pertussis toxin gene: nucleotide sequence and genetic organization. Science. 1986 Jun 6;232(4755):1258–1264. [Abstract] [Google Scholar]
- Nicosia A, Bartoloni A, Perugini M, Rappuoli R. Expression and immunological properties of the five subunits of pertussis toxin. Infect Immun. 1987 Apr;55(4):963–967. [Europe PMC free article] [Abstract] [Google Scholar]
- Nicosia A, Perugini M, Franzini C, Casagli MC, Borri MG, Antoni G, Almoni M, Neri P, Ratti G, Rappuoli R. Cloning and sequencing of the pertussis toxin genes: operon structure and gene duplication. Proc Natl Acad Sci U S A. 1986 Jul;83(13):4631–4635. [Europe PMC free article] [Abstract] [Google Scholar]
- Oda M, Cowell JL, Burstyn DG, Manclark CR. Protective activities of the filamentous hemagglutinin and the lymphocytosis-promoting factor of Bordetella pertussis in mice. J Infect Dis. 1984 Dec;150(6):823–833. [Abstract] [Google Scholar]
- Poole K, Schiebel E, Braun V. Molecular characterization of the hemolysin determinant of Serratia marcescens. J Bacteriol. 1988 Jul;170(7):3177–3188. [Europe PMC free article] [Abstract] [Google Scholar]
- Relman DA, Domenighini M, Tuomanen E, Rappuoli R, Falkow S. Filamentous hemagglutinin of Bordetella pertussis: nucleotide sequence and crucial role in adherence. Proc Natl Acad Sci U S A. 1989 Apr;86(8):2637–2641. [Europe PMC free article] [Abstract] [Google Scholar]
- Runeberg-Nyman K, Engström O, Löfdahl S, Ylöstalo S, Sarvas M. Expression and secretion of pertussis toxin subunit S1 in Bacillus subtilis. Microb Pathog. 1987 Dec;3(6):461–468. [Abstract] [Google Scholar]
- Sato H, Sato Y. Bordetella pertussis infection in mice: correlation of specific antibodies against two antigens, pertussis toxin, and filamentous hemagglutinin with mouse protectivity in an intracerebral or aerosol challenge system. Infect Immun. 1984 Nov;46(2):415–421. [Europe PMC free article] [Abstract] [Google Scholar]
- Sato Y, Kimura M, Fukumi H. Development of a pertussis component vaccine in Japan. Lancet. 1984 Jan 21;1(8369):122–126. [Abstract] [Google Scholar]
- Schiebel E, Braun V. Integration of the Serratia marcescens haemolysin into human erythrocyte membranes. Mol Microbiol. 1989 Mar;3(3):445–453. [Abstract] [Google Scholar]
- Schiebel E, Schwarz H, Braun V. Subcellular location and unique secretion of the hemolysin of Serratia marcescens. J Biol Chem. 1989 Sep 25;264(27):16311–16320. [Abstract] [Google Scholar]
- Tuomanen E, Weiss A. Characterization of two adhesins of Bordetella pertussis for human ciliated respiratory-epithelial cells. J Infect Dis. 1985 Jul;152(1):118–125. [Abstract] [Google Scholar]
- Uphoff TS, Welch RA. Nucleotide sequencing of the Proteus mirabilis calcium-independent hemolysin genes (hpmA and hpmB) reveals sequence similarity with the Serratia marcescens hemolysin genes (shlA and shlB). J Bacteriol. 1990 Mar;172(3):1206–1216. [Europe PMC free article] [Abstract] [Google Scholar]
- Vieira J, Messing J. The pUC plasmids, an M13mp7-derived system for insertion mutagenesis and sequencing with synthetic universal primers. Gene. 1982 Oct;19(3):259–268. [Abstract] [Google Scholar]
- von Heijne G. A new method for predicting signal sequence cleavage sites. Nucleic Acids Res. 1986 Jun 11;14(11):4683–4690. [Europe PMC free article] [Abstract] [Google Scholar]
- Weiss AA, Falkow S. Genetic analysis of phase change in Bordetella pertussis. Infect Immun. 1984 Jan;43(1):263–269. [Europe PMC free article] [Abstract] [Google Scholar]
- Weiss AA, Hewlett EL, Myers GA, Falkow S. Tn5-induced mutations affecting virulence factors of Bordetella pertussis. Infect Immun. 1983 Oct;42(1):33–41. [Europe PMC free article] [Abstract] [Google Scholar]
Associated Data
Articles from Infection and Immunity are provided here courtesy of American Society for Microbiology (ASM)
Full text links
Read article at publisher's site: https://doi.org/10.1128/iai.58.9.2895-2905.1990
Read article for free, from open access legal sources, via Unpaywall:
https://iai.asm.org/content/iai/58/9/2895.full.pdf
Free after 4 months at iai.asm.org
http://iai.asm.org/cgi/reprint/58/9/2895
Free to read at iai.asm.org
http://iai.asm.org/cgi/content/abstract/58/9/2895
Citations & impact
Impact metrics
Citations of article over time
Alternative metrics

Discover the attention surrounding your research
https://www.altmetric.com/details/127840939
Article citations
Virus-like particles displaying the mature C-terminal domain of filamentous hemagglutinin are immunogenic and protective against <i>Bordetella pertussis</i> respiratory infection in mice.
Infect Immun, 92(8):e0027024, 18 Jul 2024
Cited by: 0 articles | PMID: 39023271 | PMCID: PMC11320929
Structural and biophysical characterization of the secreted, β-helical adhesin EtpA of Enterotoxigenic Escherichia coli.
PLoS One, 18(6):e0287100, 21 Jun 2023
Cited by: 3 articles | PMID: 37343026 | PMCID: PMC10284417
Filamentous Hemagglutinin of Bordetella pertussis Does Not Interact with the β2 Integrin CD11b/CD18.
Int J Mol Sci, 23(20):12598, 20 Oct 2022
Cited by: 1 article | PMID: 36293453 | PMCID: PMC9604300
Subcutaneous administration of a fusion protein composed of pertussis toxin and filamentous hemagglutinin from Bordetella pertussis induces mucosal and systemic immune responses.
Iran J Basic Med Sci, 21(7):753-759, 01 Jul 2018
Cited by: 1 article | PMID: 30140416 | PMCID: PMC6098962
Restricted antibody response to Bordetella pertussis filamentous hemagglutinin induced by whole-cell and acellular pertussis vaccines.
Infect Dis (Lond), 48(2):127-132, 06 Oct 2015
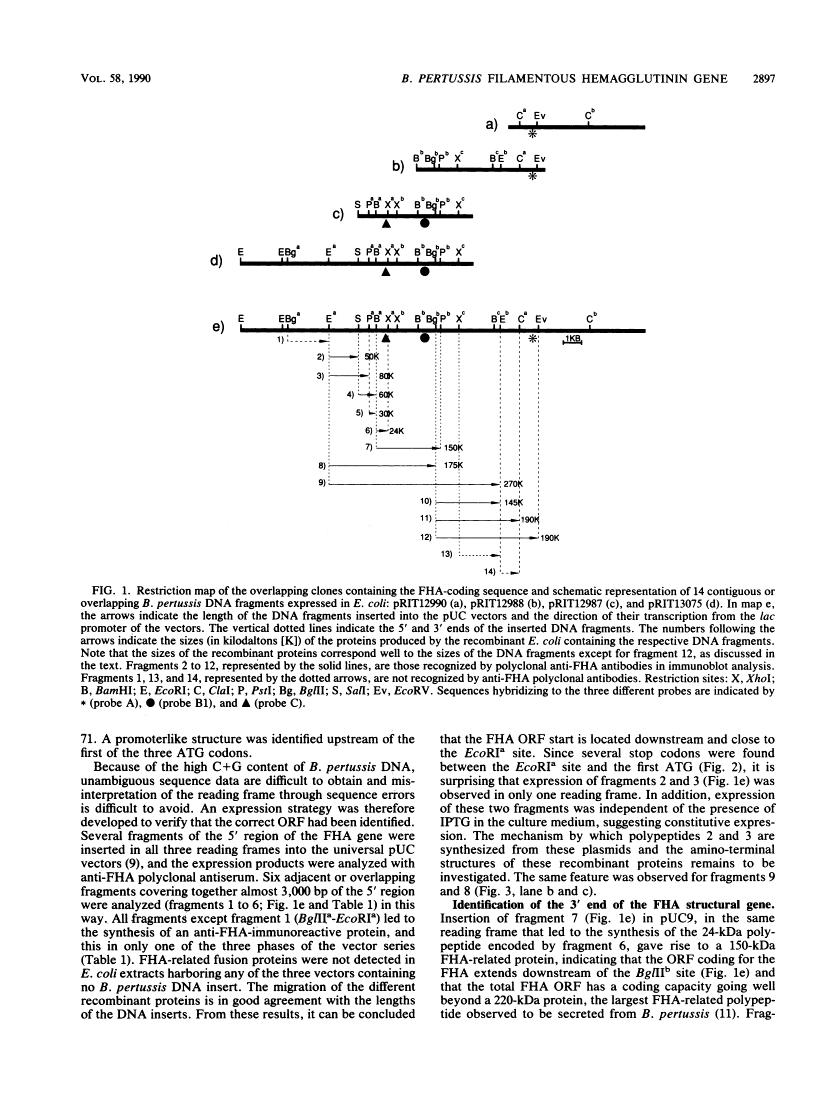
Cited by: 3 articles | PMID: 26439274
Go to all (55) article citations
Data
Data behind the article
This data has been text mined from the article, or deposited into data resources.
BioStudies: supplemental material and supporting data
Similar Articles
To arrive at the top five similar articles we use a word-weighted algorithm to compare words from the Title and Abstract of each citation.
Mutational analysis of the Bordetella pertussis fim/fha gene cluster: identification of a gene with sequence similarities to haemolysin accessory genes involved in export of FHA.
Mol Microbiol, 11(2):337-347, 01 Jan 1994
Cited by: 59 articles | PMID: 8170396
Antigenic analysis of Bordetella pertussis filamentous hemagglutinin with phage display libraries and rabbit anti-filamentous hemagglutinin polyclonal antibodies.
Infect Immun, 66(10):4884-4894, 01 Oct 1998
Cited by: 16 articles | PMID: 9746593 | PMCID: PMC108604
Lack of functional complementation between Bordetella pertussis filamentous hemagglutinin and Proteus mirabilis HpmA hemolysin secretion machineries.
J Bacteriol, 179(3):775-783, 01 Feb 1997
Cited by: 56 articles | PMID: 9006033 | PMCID: PMC178760
Direct expression of Bordetella pertussis filamentous hemagglutinin in Escherichia coli and Salmonella typhimurium aroA.
Infect Immun, 59(10):3787-3795, 01 Oct 1991
Cited by: 9 articles | PMID: 1716615 | PMCID: PMC258952