Abstract
Free full text

Tertiary lymphoid organs in infection and autoimmunity
Abstract
The lymph nodes (LNs) and spleen have an optimal structure that allows the interaction between T cells, B cells and antigen-presenting dendritic cells (DCs) on a matrix made up by stromal cells. Such a highly organized structure can also be formed in tertiary lymphoid organs (TLOs) at sites of infection or chronic immune stimulation. This review focuses on the molecular mechanisms of TLO formation and maintenance, the controversies surrounding the nature of the inducing events, and the functions of these structures in infection, transplantation and autoimmunity.
Definition and structure of tertiary lymphoid organs
The immune system has evolved in a way that optimizes the chance of encounter between the rare antigen-specific T and B cells of the adaptive immune system with antigen-presenting cells (APCs) of the innate immune system in organized lymphoid tissues, such as the spleen, lymph nodes (LNs) and Peyer's patches. These so-called secondary lymphoid organs (SLOs) have three unique features. First, they filter lymph or blood and sample the antigens in these fluids. Second, they allow entry of antigen-loaded dendritic cells (DCs) and other innate immune cells. Third, they allow recirculating naive T and B cells that have encountered antigen to extravasate, arrest temporarily, proliferate and differentiate [1]. To achieve this goal, the LNs and spleen are connected during embryogenesis to the lymph and bloodstream and are organized into well-defined areas rich in T or B cells. These areas are supported by a network of mesenchymal cells that provide the chemokine cues and extracellular matrix on which cells can meaningfully migrate and find each other in such a complex structure 2, 3. This highly efficient structural organization of antigen encounter and lymphocyte migration and activation can also be recapitulated after birth, in the form of TLOs; particularly when there is continued need for extravasation of leukocytes and a persistent source of antigen, such as seen in zones of infection, transplant rejection and auto-immune attack (see Table 1 for an overview).
Table 1
Diseases and experimental models in which TLOs have been found.
| Disease | Location | Species | Antigen specific | References |
|---|---|---|---|---|
| Microbial causes | ||||
| Influenza | iBALT | Mouse | Yes | 23, 37 |
| Vaccinia virus Ankara | Lung | Mouse | Naive T cell activation | [46] |
| Intestinal microbiota | ILFs in intestinal wall | Mouse, human | ? | 31, 72, 73 |
| Helicobacter pylori | Gastric wall | Mouse, human | Yes | [22] |
| Helicobacter hepaticus | Liver | Mouse | ? | [56] |
| Borrelia burgdorferi | Multiple sites Lyme's disease | Human | ? | [83] |
| Chronic LPS exposure | iBALT | Neonatal mouse | LPS? | [30] |
| Autoimmune disease | ||||
| RA | Synovial space | Human, mouse | Yes | 20, 74 |
| Lung interstitium | Human | Yes | [55] | |
| Primary biliary cirrhosis | Liver | Human | No: absence of any clonal T or B cell proliferation | [75] |
| Hashimoto's thyroiditis | Thyroid gland | Human | Thyroglobulin, Thyroperoxidase | [66] |
| Diabetes | Periductally in pancreatic parenchyma | NOD mouse | Yes, anti-insulin antibody responses | 41, 60 |
| Secondary progressive multiple sclerosis | Meninges at deep cerebral sulci | Mouse (EAE), human | Myelin and neuronal antigens: suggested | 62, 68, 76 |
| Myasthenia gravis | Thymus | Human | acetyl choline receptor: correlation of autoantibodies with infiltrates | [77] |
| Autoimmune gastritis | Gastric lamina propria | Mouse | Parietal cells | [78] |
| SLE | Tubulointerstitial nephritis | Human | Yes (clonal restriction and mutated antibodies) | 45, 67 |
| Chronic transplant rejection | ||||
| Heart, lung, kidney | Mouse, human | Yes, alloantigens | 64, 65, 79 | |
| Degenerative/environmental causes | ||||
| Cigarette smoke | End-stage COPD lung, iBALT and interstitial | Human, mouse | Autoimmune or latent infection | 44, 50, 58 |
| Inhaled organic dust | Interstitial TLO in hypersensitivity pneumonitis | Human | Fungal spores in moldy hay | [55] |
| Diesel particles | iBALT | Mouse | ? | [80] |
| Hypercholesterolemia | Atherosclerotic plaques | Mouse | ? | [19] |
| Metal-on-metal prosthetic joints | Soft tissues around joints | Human | Co or Ni? | [81] |
| PCNs | iBALT | Mouse | [70] | |
| Pristane adjuvant (2,6,10,14-tetramethyl-pentadecane) | Peritoneal cavity (milky spots) | Mouse | [45] | |
| Idiopathic disorders | ||||
| Usual interstitial pneumonia, idiopathic lung fibrosis | Lung interstitium | Human | Autoimmune component? | 51, 55 |
| IPAH | Perivascular lesions of the lung | Human | Probably autoimmune component | [26] |
| Cancer | Lung | Human | Tumor specific | [82] |
It is well known that when inflammation becomes chronic, plasma cells and lymphocytes gradually increase in number, lymphangiogenesis is induced, and blood vessels acquire characteristics of high endothelial venules (HEVs) specialized in allowing extravasation of lymphocytes. In some conditions, the chronic infiltrate organizes into structured lesions, such as those seen in chronic granulomatous inflammation in tuberculosis, Crohn's disease, and sarcoidosis. Sometimes, the chronic infiltrate organizes into distinct T and B cell-rich aggregates. Various terms, such as ectopic lymphoid tissue, lymphoid tissue neogenesis and TLOs, have been used to describe the occurrence of these organized structures. These structures are also named according to their anatomical site, such as inducible bronchus associated lymphoid tissue (iBALT) and vascular associated lymphoid tissue (VALT). Although in most disease models the structures do not fully cover the proposed TLO definition (Box 1 ), we use the term TLO throughout this paper because we believe the functional resemblance with SLOs is much more relevant than the structural resemblance.
Recent years have seen a surge in the number of papers dealing with the instructive signals for TLO induction and maintenance, yet it is not clear how lymphoid tissue inducer (LTi) cells, T helper cells and DCs contribute to this. It is becoming clear that these structures could have similar yet distinct functions in the induction and maintenance of adaptive immune responses. In this review, we first describe the molecular and cellular signals that lead to development of TLO structures in several disease models. Second, the functional importance of TLO structures is discussed in infection and autoimmunity.
Induction of tertiary lymphoid structures
Development of organized lymphoid tissues before birth
Before discussing the mechanisms controlling TLO development during chronic inflammation, it is important to summarize organogenesis of SLOs (reviewed in detail [2] and summarized in Figure 1 ). SLOs develop in predefined areas in the embryo, often at the crossroads of lymphatics. The lymph node anlagen are first identified in mice on embryonic day 14.5 by the presence of retinoic-acid receptor-related orphan receptor (ROR)γ+ LTi cells [4]. During SLO development [2], it is crucial that CD3–CD4+CD45+ LTi cells interact with stromal lymphoid tissue organizer (LTo) cells in a process involving lymphotoxin (LT)α1β2 signaling to the LTβ receptor (LTβR). When these cells interact, LTo cells express several adhesion molecules [vascular cell adhesion molecule (VCAM)1, intercellular adhesion molecule (ICAM)1, mucosal addressin cell adhesion molecule (MADCAM)1] and homeostatic chemokines chemokine CC ligand (CCL)19 and CCL21 for T cells and DCs; chemokine CXC ligand (CXCL)13 for attraction of B cells]. These molecules act as the driving force for recruitment of lymphocytes. In addition, production of lymphangiogenetic growth factors, such as vascular endothelial growth factor (VEGF)-C, VEGF-D, fibroblast growth factor (FGF)-2 and hepatocyte growth factor (HGF), leads to formation of Lyve-1+ lymphatic vessels 5, 6. LTo cells develop further into follicular DCs (FDCs) and fibroblast reticular cells (FRCs) that provide the conduit framework on which T and B cells migrate and interact with each other. Before LTi cells can cluster, an LTα1β2-independent instructive signal is given to local fibroblasts to start producing the initial CXCL13 to attract LTi cells. Recent insights indicate that signals derived from the nervous system could provide this first signal for LN development 7, 8. Together, these studies have highlighted the importance of interactions between hematopoietic cells and stromal cells, and between LTα1β2 signaling and homeostatic chemokines in the complex organization of SLOs.
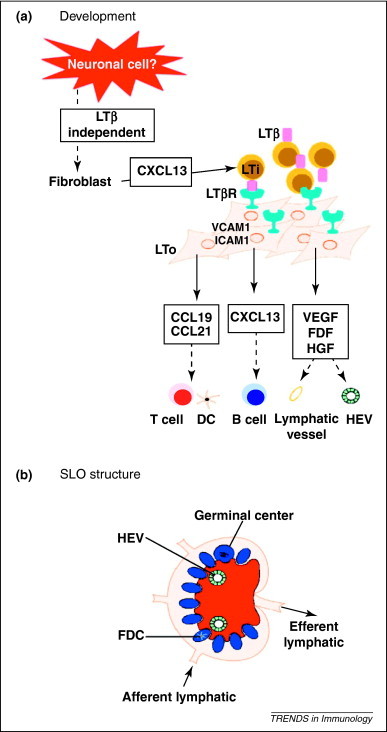
Development and structure of secondary lymphoid structures. (a) During SLO development, the earliest instructive signal is from a neuronal cell that induces the local fibroblasts to upregulate CXCL13 and thus attract LTi cells. These cells express LTβ and instruct the local fibroblasts to become LTo cells that start producing chemokines for B cells (CXCL13), T cells and DCs (CCL19 and CCL21). At the same time, the fibroblasts upregulate cell adhesion molecules to allow LTis and recruited T and B cells to stick together. Local angiogenic growth factors are also made to allow the development of high endothelial venules (HEV), as well as afferent and efferent lymph vessels. (b) As a result, a well-organized structure with a separate B cell (blue) and T cell zone (red) is formed. This allows cell–cell contact at the appropriate time; and entry and exit of lymphocytes and antigen via the HEVs and lymphatics.
Which cytokines and chemokines are involved in TLO formation?
Formation of TLOs at sites of chronic inflammation follows many of the pathways also used by lymphoid organogenesis before birth (schematically summarized in Figure 2 ). Indeed, when LTα, CCL21 or CXCL13 are overexpressed from a tissue-specific promoter (such as the rat insulin promoter; RIP), ectopic lymphoid tissues are induced before or soon after birth 9, 10, 11, 12, 13, 14, 15, 16. Conversely, in experiments in which either CXCL13, its receptor CXCR5, CCR7 or LTα was genetically absent or neutralized, TLO structures did not develop in various infectious and autoimmune models 17, 18, 19, 20, 21, 22. As in SLO formation, LTα seems to instruct stromal cells to develop into FDCs and HEVs, whereas CCL19 and CCL21 acting via CCR7 control the organization of the T cell zones of the TLO 17, 20, 23, 24.
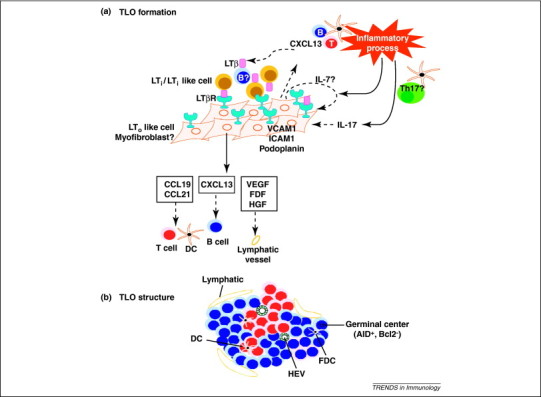
Development and structure of tertiary lymphoid structures. (a) During chronic immune responses or transplant rejection, DCs continuously present antigens to T and B cells. Activated B cells express LTβ and can act as potent LTi-like cells, to induce an LTo phenotype in local myofibroblasts. Alternatively, chronic antigen presentation by DCs might also lead to induction of a Th17 cell response that can also induce TLOs through unclear mechanisms. Th17 cells are held in place via interactions with podoplanin. (b) As in SLOs, TLOs are divided into discrete B and T cell areas. Although, fully formed TLO structures often contain only a single T cell area and a larger B cell area, in which GC reactions can be seen. These also contain DCs and FDCs. At the periphery, an elaborate network of lymphatics (Lyve1+ and Prox1+) is commonly found but it is currently unknown if these are afferent or efferent lymphatics.
The homeostatic cytokine interleukin (IL)-7 deserves separate mentioning. Il7 –/– mice lack LNs and Peyer's patches, whereas overexpression of IL-7 induces development of additional LNs. IL-7 is involved in stimulating LTi cells and in maintaining survival of T lymphocytes. IL-7 expression is augmented in the synovial tissue of rheumatoid arthritis (RA) patients, and there is a strong correlation between IL-7 expression and LTβ expression [25]. In the perivascular TLOs of patients with idiopathic pulmonary hypertension (IPAH), strong immunoreactivity for IL-7 very close to ER–TR7+ FRCs was found [26].
Together these data illustrate that homeostatic chemokines and cytokines are sufficient and necessary for the induction of TLOs. Given this, it has been suggested that inflammation-induced TLOs might have developed during evolution before SLOs did.
Are LTi cells necessary for inducing TLO formation?
In contrast to the involvement of CD3–CD4+CD45+ LTi cells in the formation of LNs, their necessity for TLO induction is controversial [2]. The differentiation of LTi cells from lymphoid progenitors involves the transcription factors Id2 and ROR-γt. Mice lacking these factors do not develop most LNs (except nasal associated mucosal tissues, NALT, and tear duct associated lymphoid tissue, TALT) 4, 27, 28, 29. When Id2 –/– or Rorc –/– mice, which are lacking LTi cells, are infected with influenza virus, they develop normal iBALT structures in the lung 23, 30, 31. Other phenomena that do not depend on LTi cells are postnatal development of omental milky spots (these are organized lymphomyelopoietic tissue in the peritoneal cavity), isolated lymphoid follicles and TLOs in the intestine caused by dextran sodium sulfate (DSS)-induced colitis, and TLOs in autoimmune thyroiditis or insulitis 12, 31, 32, 33. The controversy surrounding the role of LTi cells stems from three observations. First, adoptive transfer studies provide evidence that injection of LTi cells into normal skin is sufficient to induce TLO structures [34]. Second, LTi cells have been observed in models of spontaneous TLO development, such as the perivascular TLOs seen in hypercholesterolemic Apoe –/– mice and the iBALT structures seen in neonatal lungs exposed to lipopolysaccharide (LPS) [30]. Human TLOs, such as those seen in IPAH, also contain adult LTi-like cells that are CD4–, cKit+ and OX40L positive, and also express RORc and Id2 [26]. Third, overexpression of homeostatic chemokines under a tissue-specific promoter to induce TLOs (e.g. RIP or the intestine-specific villin promoter driving expression of CXCL13) and transgenic overexpression of IL-7 both result in increased numbers of LTi cells and dependence of TLO formation on LTi cells [33]. However, in these systems, lymphoid organogenesis starts before birth, which means that the requirement for LTi cells in induction of TLO formation could be overestimated. Nevertheless, most groups agree that LTα1β2 is necessary for TLO induction 17, 21.
Which other cells can act as LT inducers in chronic inflammation?
In the models in which LTi cells have been shown to be redundant, which other LTα1β2-expressing cell type could be providing the instructive signal to activate stromal organizer cells and initiate TLO formation? It has been proposed that B cells, T cells or DCs could substitute for LTi cells in their inductive function, especially when they are activated and express LTα1β2 on their surface 31, 35, 36, 37. Progression towards mature, fully structured TLOs is dependent on LTβ-sufficient B cells [24]. In a model of thyroid overexpression of CCL21, it has been shown that CD3+CD4+ activated T cells interact with DCs at sites of chronic inflammation, and subsequently the activated T cells act as LTi cells in the absence of Id2 activity [12]. In two recent studies on neonatal mice exposed to endotoxin inhalation and mice with experimental autoimmune encephalomyelitis (EAE), an activated T helper (Th)17 CD4 T cell population was found to be involved in inducing TLO structures 30, 38. RORC+ IL-17-producing cells have also been found inside TLOs of patients with IPAH. In humans, Th17 cells express the CCR6 receptor, and in the bloodstream of IPAH patients, circulating CCR6+ cells are fewer, whereas the ligand CCL20 is produced in the perivascular TLOs [26]. However, TLOs seem to develop normally in Ccr6 –/– mice [30]. The induction of TLOs by Th17 cells is dependent on expression of podoplanin, but why this is so remains to be studied. One possibility is that podoplanin is required for retention of Th17 cells at sites of TLO formation 30, 38. It remains to be seen whether all forms of TLO depend on IL-17 production, and whether IL-17A and/or IL-17F is involved. In this regard, iBALT induced by infection with modified vaccinia virus Ankara or influenza virus is not affected by deficiency of IL-17A 31, 39. LTβ signaling is not triggered only via LTα1β2; LIGHT (TNFSF14) can also bind the LTβ receptor. In a model of nonobese diabetes (NOD), LIGHT expressed on T cells has been shown to be important in the development and maintenance of TLO structures [40].
In almost all TLO structures that have been described, the T cell area contained antigen-presenting DCs 26, 37. DCs activate T cells, therefore, it has been suggested that DCs are sufficient for TLO induction [41]. Repeated injection of DCs into the lungs of mice is sufficient for induction of iBALT structures accompanied by induction of myofibroblast differentiation 37, 42. During formation of Peyer's patches, a CD11c+ cell type expressing LTαβ accumulates at the LN anlagen and is necessary for stromal instruction [8]. DCs might also directly instruct stromal cells irrespective of their effects on T cells. In TLO structures induced in the thymus, DCs are specifically necessary for induction of lymphangiogenesis from stromal cells [43]. How DCs induce TLOs is less clear. In virus-induced iBALT, mainly CD11b+ DCs accumulate; these cells express instructive LTα1β2 and at the same time produce the homeostatic chemokines CXCL13 and CCL19/CCL21 [37]. However, in some models, mostly pDCs accumulate, suggesting a functional role for type I interferons. This is the case in TLOs found in end-stage chronic obstructive pulmonary disease (COPD) patients and in a murine systemic lupus erythematosus (SLE) model 44, 45. Three studies have shown that depletion of DCs leads to disappearance of existing TLO structures, suggesting that DCs are necessary for structural organization and maintenance of TLOs; probably through transpresentation of chemokines, or by provision of a continuous source of antigen presentation to T cells 37, 43, 46.
Overall, there is no strong evidence pointing towards one specialized LTi cell type in TLO induction. LTi cells, B cells, T cells and DCs remain candidate cell types, with an emerging role for Th17 cells.
What is the nature of the stromal organizer cell?
Stromal cells, such as FDCs of the B cell area, FRCs of the T cell area, lymphatic endothelial cells and HEVs, play an important role in the functioning of SLOs [47]. It has become clear that TLOs have the same complicated network of stromal cells and the conduits they form, although their function remains undetermined [33]. Upon LTβR triggering by LTi or inflammatory cells, LTo cells express cell adhesion molecules and produce chemokines that attract B and T cells, produce cytokines that maintain lymphocyte viability (e.g. IL-7), and differentiate into FRCs and FDCs. LTβ receptor signaling from host cells is involved in formation of lymphatic vessels in thyroid TLOs 21, 43 and formation of the reticular network [33]. Several studies have suggested that LTo and FRC cells of the T cell zone, like myofibroblasts, stain positive for α-smooth muscle actin and produce desmin [48]. It has been elegantly demonstrated that inflammation can induce this program of myofibroblast differentiation in tissue-resident fibroblasts, while at the same time inducing the production of the homeostatic chemokines CXCL13 and CCL21, and lymphangiogenic cytokines. This programming does not require adaptive immunity, innate lymphoid cells, LTi cells, inflammasomes, or pattern recognition receptors, but is dependent on a myeloid cell population; probably neutrophils. The molecular signal released by these myeloid inflammatory cells is not known [6]. In a model of atherosclerosis, it has been shown that mouse aorta smooth muscle cells can differentiate into a cell type that resembles the LTo cell 19, 49. Myofibroblasts are typically seen at sites of scarring and fibrosis, which might explain the occurrence of TLOs in patients with COPD, scleroderma, lung fibrosis and primary biliary cirrhosis 50, 51, 52.
In several instances where TLOs have been found in humans, they were also closely associated with c-Kit+ mast cells 25, 26. Mast cells produce cytokines (e.g. tumor necrosis factor (TNF)α and express surface molecules (CD40L) that could help in structuring TLOs. They are also an important source of VEGF-C, which induces lymphangiogenesis.
In conclusion, TLO formation follows the same basic set of rules also implicated in formation of LNs and Peyer's patches (Figure 2). Controversies remain on the precise nature of the LTi cell, and how it communicates with LTo cells.
Function of TLOs
TLOs develop in areas of chronic immune stimulation. However, although different infectious and immune or inflammatory triggers exhibit the same degree of chronic inflammation and inflict similar damage, their capacity to induce TLOs can vary widely. Moreover, tertiary lymphoid tissues develop more easily in neonates [30]. There is accumulating evidence that TLOs represent an adaptation of the body to increased demand for a localized immune response. The evolutionary pressure could be the constant equilibrium between commensals and the mucosal immune system, such as occurs typically in the gut and the lung. Isolated lymphoid follicles (ILFs) are a specialized type of TLO that forms postnatally in the intestinal wall, through epithelial recognition of nucleotide-binding oligomerization domain protein 1 (NOD1) ligands derived from commensals [53]. Although the deeper lung was once considered sterile, there is now evidence that it is not; particularly following viral infection or chronic cigarette smoking, typical triggers of TLO formation in the lung [54]. Here again, commensals could be a driving force for TLO development.
TLOs as immune inductive sites for protective immunity to (re-)infection
Similar to SLOs that bring together naive T cells, B cells and DCs, TLOs have been shown to allow the activation of recirculating naive T cells and the activation of B cells within germinal centers (GCs). One caveat is that these studies were performed in mice lacking LNs, which could have led to overestimation of the potential of TLOs [23]. However, recent work has suggested that TLOs can be immune inductive even in mice with normal LN anatomy and a fully functional immune system. DCs in TLOs often have an activated phenotype, suggesting that they are the predominant APCs stimulating immunity [45]. The function of DCs in TLO biology has been studied mainly in iBALT structures induced by modified Ankara vaccine virus or influenza virus 37, 46. Preformed iBALT structures collect DCs that are injected into the trachea, but it is unclear how these DCs reach the T cell area of iBALT [46]. One possibility is that iBALT structures are connected to afferent lymphatics located immediately underneath the epithelial basement membrane [52]. In many instances, the TLOs are found immediately next to lymphatics, but no study so far has been able to determine if these lymphatics are afferent (bringing antigen to the TLO) or efferent (exporting lymphocytes). It is also possible that the remodeling around blood vessels and the subepithelial areas leads to formation of a conduit system that provides a path for the encountered antigens to reach the TLO. In support of this possibility is the close resemblance between the type of extracellular matrix found in atherosclerosis-associated or IPAH-associated TLOs and the one found in the FRC network and conduit system of LNs 19, 26. Whatever the mechanism, TLO structures do allow the extravasation of naive T cells and their differentiation into effector T cells upon contact with DCs [46].
A major constituent of TLOs are the B cells that accumulate on the FDC network. Almost all TLOs described exhibit signs of B cell class switching, as exemplified by the presence of high amounts of the Activation-induced cytidine deaminase (AID) enzyme and the presence of GC reactions 26, 37. Such B cells provide an important source of memory B cells that are activated upon reinfection 37, 55. Plasma cells are found in the immediate vicinity of the TLOs and secrete antibodies specific for the pathogen that induces the TLO, such as influenza virus or Helicobacter species 37, 56. Although some of the B cells generated in the TLO also seem to reach the bone marrow, where they can reside as long lived plasma cells, the iBALT system that forms after influenza virus infection and the ILFs that form in response to intestinal commensals mainly serve to produce local mucosal IgA 37, 57, 58.
It is not known why some infections leave behind a TLO structure whereas others do not. In a model of influenza, for T cell immunity to persist, there is continued dependency on lung DCs that take viral antigens from the lung to the mediastinal LNs, even long after the virus has been cleared, for presentation to memory T cells. CD11b+ DCs can be found mainly in iBALT in the lung long after virus is cleared, therefore, it is possible that they capture retained viral antigens from the FDC network [59].
TLOs in immunopathology
The presence of TLOs in models of chronic autoimmunity and in chronic transplant rejection and their association with tissue destruction (Table 1) has led to the suggestion that they are important inductive sites for self-reactive T lymphocytes and antibodies that contribute to pathology. However, the evidence for this is rather sparse. During progression of diabetes in NOD mice, there is progressive development of TLOs in the pancreas. In these TLOs, T lymphocytes are detected that destroy islets and plasma cells with specificity for insulin 40, 60. The expansion of B cells inside an individual TLO around islets is often oligoclonal, suggesting the presence of an autoantigen [61]. More severe pathology of the grey matter and earlier onset of disease has been noticed in cases of autoimmune encephalomyelitis in which ectopic B cell follicles are present [62].
In a model of DSS-induced colitis in mice lacking RORγT (and thus lacking LNs, LTi cells, Th17 cells and innate lymphoid cells), the induction of B-cell-rich TLOs was associated with more severe immunopathology, and the mice succumbed from a lethal wasting disease that could be prevented by treatment with LTβR immunoglobulin. Strikingly, the disease was also prevented by treatment with intravenous immunoglobulins (IVIGs) to saturate Fc receptors on effector cells. This finding shows that these TLOs contribute to immunopathology via a humoral mechanism [31]. Chronic alloimmune responses induce TLOs in mice and humans, and these TLOs could be involved in chronic rejection. Transplantation of skin harboring TLOs to mice of a different strain lacking all LNs and spleen leads to chronic rejection, whereas transplantation of skin lacking TLOs is not rejected [63].
Also in humans, TLO structures might contribute to disease progression in autoimmunity and transplant rejection 64, 65. In the lungs of patients with RA, B cells in the TLO produce rheumatoid factor (RF) and antibodies against citrullinated proteins; typical self antigens in this disease [52]. In human autoimmune thyroiditis, most B cells inside ectopic GCs bound the autoantigens thyroglobulin and thyroidal peroxidase [66]. A study on SLE patients has confirmed the restricted B cell repertoire, with formation of isotype-switched antibodies that are deposited in the glomerular basement membrane [67]. TLOs have also been found in the meninges of patients with secondary progressive multiple sclerosis; a severe feature of SLE. Notably, all meningeal B cell follicles are found in cerebral sulci adjacent to large subpial cortical lesions, suggesting that soluble factors diffusing from these structures have a pathogenic role [68].
However, it is less clear which antigens the lymphocytes might react to in more severely degenerative diseases such as COPD induced by cigarette smoke and atherosclerosis induced by western diet. Although it has been proposed that there is an autoimmune component in many chronic tissue-destructive diseases such as COPD, another explanation could be colonization of the lung by latent adenovirus or unsuspected commensals in the lung [50]. Until we know the antigen-specificity of the B and T cell responses in TLOs, we can only speculate on the nature of the antigens presented in diseases such as IPAH and primary biliary sclerosis. In any case, the extensive formation of myofibroblast-like cells that constitute part of the stromal organizing network might contribute to local fibrosis that could compromise organ function.
Therapeutic applications of TLOs
Exploiting the induction of TLOs by vaccines could be a valuable option in promoting long-lasting, local antimicrobial immunity in the lung and gut [69]. Mice pretreated with protein-caged nanoparticle (PCN) adjuvants in the absence of any specific viral antigens are protected against both sublethal and lethal doses of two different influenza viruses, a mouse-adapted SARS-coronavirus, and a mouse pneumovirus. Treatment with PCN significantly increases survival, enhances viral clearance, accelerates induction of virus-specific antibody production, and significantly decreases morbidity and lung damage; these changes have been attributed to prior development of iBALT [70]. A deeper understanding of the mechanism of action of this protection is needed before we can rationally design adjuvants that induce TLOs.
If TLOs are found in many chronic diseases with an autoimmune component and in chronic transplant rejection, could interfering with their induction or maintenance be developed into a novel therapeutic approach? One possibility is to interfere with the LTβ receptor. In a model of autoimmune diabetes in NOD mice, disrupting TLOs by antagonizing the action of LIGHT with LTβR inhibits formation of autoaggressive T cells and progression to diabetes [40]. Similarly, disruption of TLOs in the salivary glands that develop in the NOD model of Sjögren's disease also leads to partial restoration of salivary function [18]. Another approach could be to target crucial TLO chemokines or their receptors, such as CXCL13 or CXCR5. In a model of RA, Cxcr5 –/– mice have significantly reduced joint destruction [20]. In a model of diabetes in NOD mice, neutralization of CXCL13 leads to disorganized TLOs, yet there is no clear effect on diabetes incidence [71]. Clearly, more preclinical studies on these compounds are needed before we can envisage an intervention study in humans.
Concluding remarks
It is now clear that chronic inflammation induced by chronic infections, autoimmune reactions or chronic graft rejection is often accompanied by tertiary lymphoid structures that are tightly organized to allow interactions between recirculating T and B cells and APCs. The formation of these structures follows the same basic rules and involves the same cell types that also control the formation of SLOs. Given the fact that the function of TLOs is similar to that of SLOs, the basic question for the field is why exactly our defense system mounts this, at first sight, redundant response. Given the fact that TLOs often lead to formation of tissue-directed autoantibodies, targeting TLO formation could be beneficial is autoimmune diseases.
Acknowledgments
K.N. is supported by a grant from FWO Flanders. F.P. is supported by a grant from Fondation pour la Recherche Médicale (FRM), grant number DEQ20100318257, D.H.G. is supported by a ESCMID grant, H.H. is supported by grants from FWO Flanders, B.N.M. is supported by Odysseus and Project grants of FWO Flanders, by an ERC Consolidator grant, and by a Ghent University MRP Grant (Group-ID consortium).
References
Full text links
Read article at publisher's site: https://doi.org/10.1016/j.it.2012.04.006
Read article for free, from open access legal sources, via Unpaywall:
http://www.cell.com/article/S1471490612000713/pdf
Citations & impact
Impact metrics
Citations of article over time
Alternative metrics
Smart citations by scite.ai
Explore citation contexts and check if this article has been
supported or disputed.
https://scite.ai/reports/10.1016/j.it.2012.04.006
Article citations
Tertiary Lymphoid Structures in Microorganism-Related Cancer.
Cancers (Basel), 16(20):3464, 12 Oct 2024
Cited by: 0 articles | PMID: 39456558 | PMCID: PMC11505735
Review Free full text in Europe PMC
Podoplanin expressing macrophages and their involvement in tertiary lymphoid structures in mouse models of Sjögren's disease.
Front Immunol, 15:1455238, 17 Sep 2024
Cited by: 0 articles | PMID: 39355243 | PMCID: PMC11442383
The role of immune cells in the pathogenesis of connective tissue diseases-associated pulmonary arterial hypertension.
Front Immunol, 15:1464762, 17 Sep 2024
Cited by: 0 articles | PMID: 39355239 | PMCID: PMC11442293
Review Free full text in Europe PMC
Single-cell and spatial transcriptome analyses reveal tertiary lymphoid structures linked to tumour progression and immunotherapy response in nasopharyngeal carcinoma.
Nat Commun, 15(1):7713, 04 Sep 2024
Cited by: 1 article | PMID: 39231979 | PMCID: PMC11375053
Tertiary lymphoid structures in diseases: immune mechanisms and therapeutic advances.
Signal Transduct Target Ther, 9(1):225, 28 Aug 2024
Cited by: 0 articles | PMID: 39198425 | PMCID: PMC11358547
Review Free full text in Europe PMC
Go to all (229) article citations
Similar Articles
To arrive at the top five similar articles we use a word-weighted algorithm to compare words from the Title and Abstract of each citation.
High Endothelial Venules and Lymphatic Vessels in Tertiary Lymphoid Organs: Characteristics, Functions, and Regulation.
Front Immunol, 7:491, 09 Nov 2016
Cited by: 63 articles | PMID: 27881983 | PMCID: PMC5101196
Review Free full text in Europe PMC
Basics of Inducible Lymphoid Organs.
Curr Top Microbiol Immunol, 426:1-19, 01 Jan 2020
Cited by: 18 articles | PMID: 32588229
Review
Tertiary Lymphoid Structures Among the World of Noncanonical Ectopic Lymphoid Organizations.
Methods Mol Biol, 1845:1-15, 01 Jan 2018
Cited by: 4 articles | PMID: 30141004
Review
The role of dendritic cells in tertiary lymphoid structures: implications in cancer and autoimmune diseases.
Front Immunol, 15:1439413, 11 Oct 2024
Cited by: 0 articles | PMID: 39483484 | PMCID: PMC11526390
Review Free full text in Europe PMC





