Abstract
Free full text

Probiotic strains detect and suppress cholera in mice
Abstract
Microbiota-modulating interventions are an emerging strategy to promote gastrointestinal homeostasis. Yet, their use in the detection, prevention, and treatment of acute infections remains underexplored. We report the basis of a probiotic-based strategy to promote colonization resistance and point-of-need diagnosis of cholera, an acute diarrheal disease caused by the pathogen Vibrio cholerae. Oral administration of Lactococcus lactis, a common dietary fermentative bacterium, reduced intestinal V. cholerae burden and improved survival in infected infant mice through the production of lactic acid. Furthermore, we engineered an L. lactis strain that specifically detects quorum-sensing signals of V. cholerae in the gut and triggers expression of an enzymatic reporter that is readily detected in fecal samples. We postulate that preventive dietary interventions with fermented foods containing natural and engineered L. lactis strains may hinder cholera progression and improve disease surveillance in populations at risk of cholera outbreaks.
INTRODUCTION
Cholera is a life-threatening gastrointestinal infection that principally affects populations where basic sanitation and healthcare are insufficient (1). Severe dehydration from cholera can lead to death within hours if treatment is not provided promptly. Strengthening surveillance and early identification of cholera cases are top priori-ties of the World Health Organization for the prevention of cholera outbreaks. Therefore, implementation of cost-effective measures that prevent disease progression—from initial infection to active disease and dissemination—is urgently needed. Microbiota interventions have been used to treat or alleviate a variety of gastrointestinal dis-orders (2, 3). In the case of cholera, it has been demonstrated that natural members of the human microbiome can reduce colonization of Vibrio cholerae in gnotobiotic mice through interference of the pathogen’s quorum-sensing signaling (4). Furthermore, engineered probiotics have been used to interfere with bacterial communication to suppress virulence (5), or to compete with intestinal receptor binding to the cholera toxins (6). Laboratory strains of Escherichia coli have also been engineered for the detection of V. cholerae signals under culture conditions (7). However, the demonstration of the in vivo functionality of engineered V. cholerae detection systems in safely consumed, gut-relevant bacteria is lacking.
Food-associated bacteria play a central role in the function and health of the gastrointestinal tract (8). High doses of ingested, live bacteria can create a microbiota shift that allows transient modulation of the human intestinal microenvironment (8), a phenomenon that can be leveraged to reduce pathogen colonization (9) and enable in situ diagnosis at an early stage of infection. We hypothesized that dietary-associated bacteria with natural infection-antagonizing properties could be identified, applied, and further enabled with engineered diagnostic functions to ameliorate the impact of cholera and facilitate disease surveillance in populations at risk of infection.
RESULTS
Promoting colonization resistance against V. cholerae with Lactococcus lactis
L. lactis is a food-associated lactic acid bacterium that has been safely consumed for millennia as part of fermented milk products (10). This bacterium displays strong acidification capabilities that stem from its high carbon flux metabolism, which is optimized for the conversion of simple carbohydrates into lactic acid (11, 12). Given that V. cholerae is known to be particularly sensitive to acidic conditions (13) and that both L. lactis and V. cholerae find temporary residence in the small intestine (14, 15), we sought to determine whether L. lactis could be used to promote intestinal colonization resistance against V. cholerae through the production of lactic acid. Initial in vitro agar diffusion and liquid coculture assays showed that L. lactis could effectively antagonize V. cholerae growth (Fig. 1, ,AA and andB,B, and figs. S1 and S2). The L. lactis antibacterial effect on V. cholerae was abolished when lactic acid production was impaired by mutation of the lactate dehydrogenase (ldh) gene or when the pH of the coculture was strongly buffered (Fig. 1, ,AA and andB).B). These results indicate that lactic acid, a major product of L. lactis metabolism, is a V. cholerae–antagonizing agent (figs. S2 and S3).

Lactic acid–dependent antibacterial effect of L. lactis against V. cholera in vitro. (A) Top: Agar diffusion assay of wild-type (WT), lactate dehydrogenase mutant (Δldh), and plasmid-complemented lactate dehydrogenase mutant Δldh (pLDH) strains of L. lactis grown on a lawn of V. cholerae. Bottom: Agar diffusion assay of L. lactis strains in minimally buffered GM17 agar plates containing pH indicator bromocresol purple, which turns yellow when pH drops below 5.2. Inhibition zones correlate with the acidification range of L. lactis colonies. (B) Acidification dynamics and V. cholerae cell density in coculture with L. lactis in minimally buffered media and strongly buffered media (+buffer). For detailed media conditions, see Materials and Methods and fig. S2. Bars represent range of technical duplicates.
We next used an infant mouse model of cholera infection to test the ability of L. lactis to enhance colonization resistance against V. cholerae (5, 16). We evaluated the intestinal transit dynamics of L. lactis in the infant mouse model and determined that dosing 109 colony-forming units (CFU) of L. lactis cells every 10 hours ensures its sustained presence in the mouse small intestine (fig. S4). Four-day-old mice were inoculated with 107 CFU of V. cholerae either at the same time (coadministration regimen) or 5 hours after the first L. lactis dose (preadministration regimen), and host survival was monitored for the next 42 hours (Fig. 2A). We found that V. cholerae– infected mice were substantially more likely to survive when they were coinoculated with L. lactis (84.6%) compared to when they were mock-fed (45.7%) (Fig. 2B). Three independent trials showed that the improved survival is not affected by litter variation (fig. S5). Mouse survival was comparable for the coadministration and pre-administration regimens, indicating that the L. lactis dietary intervention provides protection against V. cholerae infection within the 10-hour window between the probiotic doses (Fig. 2B). Furthermore, in the coadministration regimen, the V. cholerae burden in L. lactis– fed mice was reduced by about 100-fold compared to that in mock-treated mice at 26 hours after infection (Fig. 2C). We found that the V. cholerae load correlates with the infection outcome (Fig. 2C). Regardless of the probiotic intervention, mice that died from cholera have V. cholerae developed to 109 CFU in the gut; the ones that survived until the end of study had lower than 108 CFU. This correlation be-tween reduced V. cholerae burden and mouse survival includes mice that were not fed L. lactis (Fig. 2C), further suggesting that L. lactis assists in mouse survival through reduced V. cholerae colonization. These findings indicate that a regular feeding regimen of L. lactis can prevent the aggressive development of cholera by limiting the bacterial burden to nonlethal concentrations in infant mice.
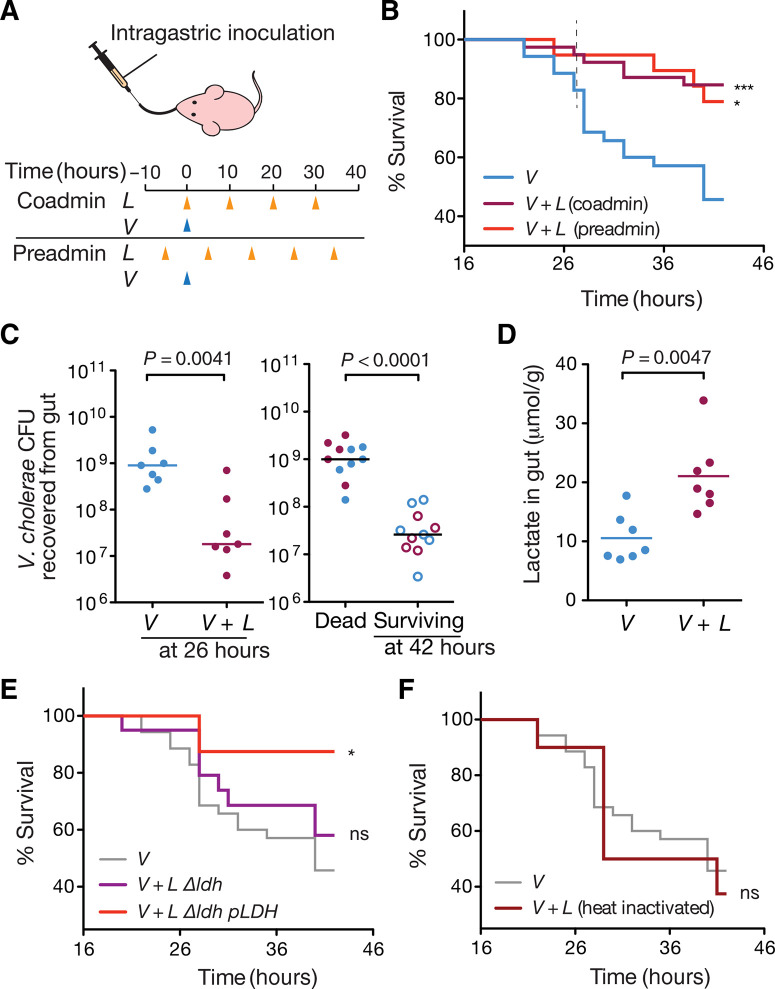
L. lactis–derived lactic acid antagonizes V. cholerae infection in mice. (A) Intragastric feeding regimens of L. lactis and inoculation time of V. cholerae to infant mice. Mock-fed mice were administered GM17 medium instead of L. lactis. (B) Effects of L. lactis intervention on infant mouse survival to cholera infection under both feeding regimens. Coadministration with V. cholerae, ***P = 0.0005; preadministration, *P = 0.0187, log-rank test against mock-fed (V). V: n = 37, N = 6; V + L, coadmin: n = 39, N = 6; V + L, preadmin: n = 19, N = 6. (C) Effect of L. lactis intervention on V. cholerae colonization in the infant mouse gut. (C) Left: V. cholerae intestinal colonization at 26 hours after infection. Right: V. cholerae burden in mice that died from cholera and in mice that survived the infection by 42 hours after infection. Each data point represents an individual mouse. Horizontal lines are medians. P values from Mann-Whitney test (n = 7, N = 7 for nontreated/treated; n = 11, N = 7 for dead versus survived, respectively). Data correspond to coadministration regimen. (D) Intestinal lactate concentration in L. lactis–treated and mock-treated infant mice at 26 hours after infection. Mann-Whitney test (n = 7, N = 7). Data correspond to coadministration regimen. (E) Effects of lactate dehydrogenase mutant (Δldh) and plasmid-complemented lactate dehydrogenase (Δldh pLDH) L. lactis strains on infant mouse survival to cholera infection. Functional knockout of lactate dehydrogenase compromises the protective effect against cholera (n = 20, N = 6). ns, not significant by log-rank test compared to mock treatment (V). Complementation with a plasmid-based ldh restores the protective effect (*P = 0.0349, log-rank test; n = 9, N = 3). (F) Effect of treatment with heat-inactivated L. lactis on infant mice survival to cholera infection. n = 10, N = 3. n, number of individual mice; N, number of litters covered in each group.
To further investigate the involvement of L. lactis lactic acid production in the reduction of V. cholerae burden in vivo, we measured the intestinal lactate levels during V. cholerae mouse infections. At 26 hours after infection, mice that were fed L. lactis exhibited in-creased levels of lactate in the gut compared to mice that had been mock inoculated (Fig. 2D). When the lactate dehydrogenase mutant strain of L. lactis was used, the protective effect against V. cholera infection disappeared (Fig. 2E). Abolishing lactate dehydrogenase function reroutes carbon flux toward mixed acid fermentation path ways and results in a slower growth rate (fig. S6), an effect that might also contribute to its reduced protective effect in vivo. Genetic complementation with plasmid-encoded ldh restored both the strain’s growth rate and acidification capabilities, and consequently its protective function in vivo (Fig. 2E and fig. S6). As expected, when heat-inactivated cells were used instead of live L. lactis, the survival rate of the treated mice was similar to the mock-fed group, indicating that metabolically active L. lactis are needed for effective protection (Fig. 2F). These findings suggest that in situ production of lactic acid by metabolically active bacteria inhibits V. cholera colonization and is essential for L. lactis to provide the observed protective effect.
To investigate the effect of direct intake of lactic acid on V. cholera intestinal colonization, we performed oral gavage of lactic acid into V. cholerae–infected infant mice and observed a reduction of pathogen load similar to the treatment with L. lactis in 62.5% of the mice, whereas the rest of the litter showed a colonization akin to the mock-treated control (fig. S7). These results indicate that although direct administration of lactic acid to the gastrointestinal tract can be sufficient to recapitulate the effect of L. lactis in the reduction of V. cholerae colonization, it is not as effective as the dietary intervention with live bacteria. Orally administered lactic acid is susceptible to neutralization during translocation from the stomach into the upper small intestine; in contrast, in situ production of lactic acid by transiting L. lactis cells allows a direct supply of acid that is independent of upstream gastrointestinal pH changes.
Higher gastrointestinal pH increases the susceptibility to V. cholerae infection in humans, especially due to the use of acid-suppressing drugs such as proton pump inhibitors (17). We evaluated the effect of the L. lactis intervention on V. cholerae–infected infant mice when oral gavage is performed using mediabuffered to pH 7.4 (fig. S8). We found that under acid-neutralizing conditions, the survival of mock-treated mice was reduced to 11%, whereas the survival of L. lactis–treated mice was 50.8%, which suggests that probiotic intervention might ameliorate the impact of the disease even under conditions of increased susceptibility of infection.
Engineering cholera-sensing functions into L. lactis
Next, we sought to engineer L. lactis to specifically detect and report on V. cholera presence in the intestinal environment. V. cholerae produces the quorum-sensing molecules autoinducer-2 (AI-2) and cholera autoinducer 1 (CAI-1), and uses the two-component histidine kinase receptors LuxQ and CqsS to specifically detect each molecule, respectively (18). Unlike AI-2, CAI-1 is unique to the genus Vibrio; therefore, we sought to develop a receptor in L. lactis to detect CAI-1 from V. cholerae. We took advantage of CqsS’s specificity to CAI-1 and the modularity of histidine kinase receptors to design an L. lactis hybrid receptor (HR) that combines the transmembrane ligand binding domain of CqsS with the signal transduction domain of NisK, a two-component receptor in L. lactis that regulates the production of the lantibiotic nisin (Fig. 3A) (19). We designed 10 HR variants with different CqsS-NisK junction points (fig. S9A). Because of the known effect of altered NisK expression levels on signaling (20), we cloned a randomized ribosome binding site (RBS) sequence upstream of each HR variant and screened the resulting HR libraries for CAI-1–dependent activity (fig. S9B). To create a convenient screening platform, we cloned the fluorescent reporter gene encoding mCherry downstream of the NisR-responsive nisA promoter and assayed each HR variant in a high-throughput coculture system with both wild-type V. cholerae and a V. cholerae cqsA deletion strain that is unable to produce CAI-1 (Fig. 3B).
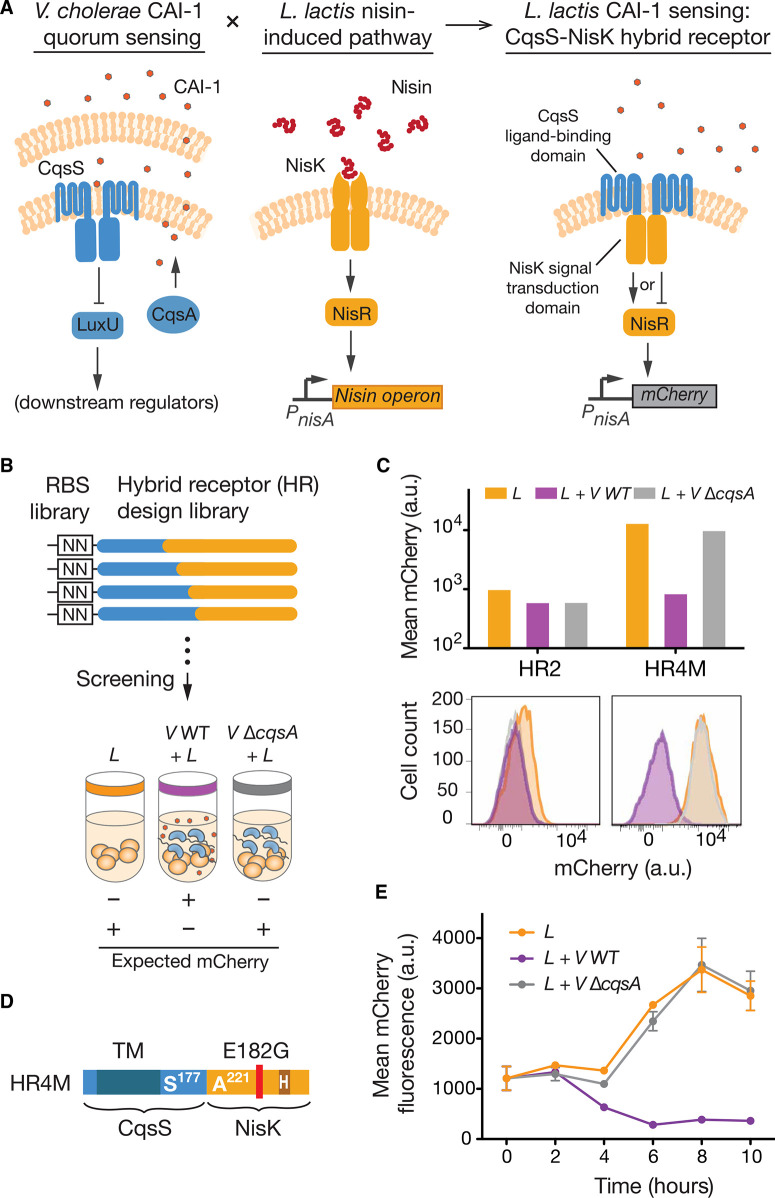
Construction of HR for V. cholerae sensing. (A) Creation of a CAI-1–sensing function in L. lactis requires the fusion of two distantly related quorum-sensing signaling pathways. The proposed HR fuses the CAI-1–binding domain of the CqsS receptor with the histidine kinase domain of NisK to achieve CAI-1–dependent signaling in L. lactis. The design of the HR cannot predict whether the regulatory outcome of CAI-1 binding will be activation (arrow-headed line) or inactivation of NisR (bar-headed line). (B) Functional screen for HR variants. CqsS-NisK fusion variants with randomized RBS strengths (fig. S9) were screened for their ability to modulate output gene expression in response to CAI-1. CAI-1–deficient V. cholerae (V ΔcqsA) serves as a negative control. (C) mCherry fluorescence output of HR variants in response to CAI-1. Hybrid receptor 4 mutant (HR4M) is a functional variant. Hybrid receptor 2 (HR2) is an example of a nonfunctional variant. (D) Primary sequence map of HR4M. S177 indicates the last residue of N-terminal part of CqsS, and A221 indicates the first residue of the C-terminal part of NisK. The HR contains a spontaneous Glu-to-Gly mutation at residue 182. (E) Reporter gene expression dynamics in L. lactis with HR4M in response to CAI-1. Error bars are SEM of three technical replicates. a.u., arbitrary units.
The native nisRK two-component system and the majority of HR variants were not responsive to CAI-1 (fig. S9, C and D). However, a functional variant with a Glu-to-Gly mutation at residue 182 (HR4M) displayed robust repression of mCherry in the presence of CAI-1 (Fig. 3, ,CC and andD).D). Detailed characterization of the dynamic response of HR4M to CAI-1 showed that mCherry repression oc-curred within 4 hours of L. lactis–V. cholerae coculture initiation, and the transcriptional response was entirely dependent on CAI-1 production by V. cholerae (Fig. 3E). Nisin binding to NisRK is known to cause a conformational shift that activates autophosphor-ylation and induces transcription of the target gene (18). However, the transcriptional repression observed suggested that HR4M pre-serves the phosphorelay properties of CqsS, wherein constitutive autophosphorylation is inhibited upon CAI-1 binding, leading to reduced transcription of target genes.
In situ detection of V. cholerae infection
We next incorporated a transcriptional inverter circuit that repro-grams HR4M signaling into an activation response upon pathogen detection. As shown in Fig. 4A, we placed the gene encoding the transcriptional repressor TetR downstream of the HR4M-controlled nisA promoter to enable constitutive repression of an engineered Bacillus subtilis xylA-tetO promoter (21) in the absence of CAI-1.
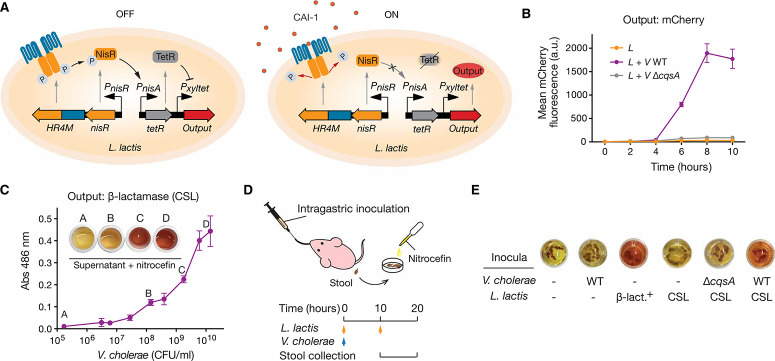
Cholera sensing and reporting by engineered L. lactis in vitro and in vivo. (A) Engineered CAI-1–dependent signaling in L. lactis. The HR4M-NisR two-component system sensing module and a TetR/Pxyltet signal-processing module constitute the complete V. cholerae–sensing circuit. In the absence of CAI-1, constitutive expression of TetR prevents the expression of the output gene. In the presence of CAI-1, the hybrid two-component system stops its phosphorelay, halting TetR expression and leading to activation of the output gene. (B) Activation dynamics of the cholera-sensing circuit. Error bars are SEM of three technical replicates. (C) Colorimetric reporting of V. cholerae sensing. The L. lactis CSL strain secretes β-lactamase, which hydrolyzes nitrocefin and produces a visible color change from yellow to red (maximum absorbance at 486 nm). Bars represent range of absorbance for technical duplicates. (D) Schematic of the infant mouse model for in vivo testing of the cholera-sensing strain. Cholera-infected mice were fed with L. lactis CSL, and stools were collected between 10 and 20 hours after infection for nitrocefin-based β-lactamase assay. (E) Performance of the living diagnostic and β-lactamase assay for the detection of cholera. Color change in nitrocefin with fecal samples indicates β-lactamase activity. V. cholerae WT, CAI-1–deficient V. cholerae (V ΔcqsA), and L. lactis cholera-sensing Lactococcus (CSL) strain. Constitutive β-lactamase expression L. lactis (β-lact.+).
Upon CAI-1 binding, HR4M repression of TetR expression enabled transcription from the xylA-tetO promoter (fig. S10A). We screened a library of tetR RBS sequences to identify an optimal TetR translation rate that would provide a suitable dynamic range of reporter gene expression upon CAI-1 induction (fig. S10B). Detection circuit characterization in coculture conditions indicates that mCherry expression increased 60-fold in response to CAI-1 from V. cholerae with minimal signal background (Fig. 4B). Although CAI-1–based quorum-sensing systems are present in most Vibrio spp., CAI-1 moieties might display different acyl chain lengths and modifications depending on the bacterial species (22). We tested the specificity of HR4M to other CAI-1–producing and nonproducing bacterial species and found that the detection circuit did not respond to most of the environmental Vibrio strains tested or to non– CAI-1–producing bacteria (fig. S11). However, Vibrio spp. producing CAI-1 moieties that are compatible with CqsS from V. cholerae such as Vibrio alginolyticus (22) were detected by HR4M (fig. S11), suggesting that this system might be used for the detection of other pathogenic Vibrio spp. that produce CqsS-compatible quorum-sensing molecules, such as Vibrio parahaemolyticus, Vibrio anguillarum, and Vibrio furnissii (22).
As a proof of concept of a living diagnostic tool with an easy, point-of-need readout, we replaced mCherry with a secreted β-lactamase reporter whose activity is readily visualized by a colorimetric shift from yellow to red after hydrolysis of the chromogenic substrate nitrocefin (23, 24). Evaluation by in vitro coculture showed that the reporter strain, termed CSL, could generate a visible color change within 30 min when the coculture had reached a density of 108 CFU/ml of V. cholerae (Fig. 4C). When the reporter assay was tested in the infant mouse model (Fig. 4D), only the fecal pellets of mice dosed with a constitutive producer of β-lactamase displayed a positive signal, demonstrating both the orthogonality and stability of the β-lactamase reporter in the mouse intestinal environment (Fig. 4E). Also, fecal pellets of mice dosed only with the L. lactis CSL strain did not display a color change, which demonstrates a tight regulation of the engineered detection circuit under in vivo conditions (Fig. 4E). Finally, to determine whether L. lactis CSL could detect cholera infection in the infant mouse model, we administered two doses of the engineered strain, the first concurrent with V. cholera and the second 10 hours after infection, to mice and collected stool samples for colorimetric assays (Fig. 4D). The fecal samples of cholera-infected mice treated with L. lactis CSL showed positive signals after overnight incubation with nitrocefin, indicating circuit activation and β-lactamase secretion in the gut (Fig. 4E). In contrast, stool samples from uninfected mice and mice infected with the cqsA mutant of V. cholerae did not show colorimetric change, demonstrating that the circuit is CAI-1–specific.
Probiotic intervention with natural and engineered L. lactis
We found that, compared to wild-type L. lactis, the L. lactis CSL strain exhibited a reduced protective effect in our infant mouse model for cholera infection (Fig. 5A). To understand this phenomenon, we studied the strain’s growth and acidification dynamics in monoculture and coculture under anaerobic conditions (figs. S12 and S13). When assayed in the infant mouse model, the engineered L. lactis CSL strain had a lower median of recovered cells from the gut than the wild-type strain; however, no significant difference was detected between strains (fig. S12A). Under liquid culture conditions, L. lactis CSL acidifies medium slower than the wild-type strain (fig. S12B). Furthermore, the viability of the L. lactis CSL strain decreases under extended coculture condition, and the steady-state pH is higher than the coculture with wild-type L. lactis (fig. S12B). Also, given the observed defect in acidification, the V. cholerae killing by L. lactis CSL is delayed by 4 hours compared to wild-type L. lactis (fig. S12C). The results we obtained indicate that circuit activation and β-lactamase secretion in the engineered L. lactis strain create a metabolic burden that reduces the bacteria’s natural acidification capacities and its antagonistic effect on V. cholerae, further supporting the hypothesis that lactic acid production in the gut environment plays a key role in the protective effect. To achieve a combined protective and diagnostic function, we introduced a mixed population of wild-type and CSL L. lactis into the feeding regimen (Fig. 5A). The combined dose of natural and engineered L. lactis successfully restored the protective effect and provided an effective diagnostic readout in stools (Fig. 5A, and andBB).
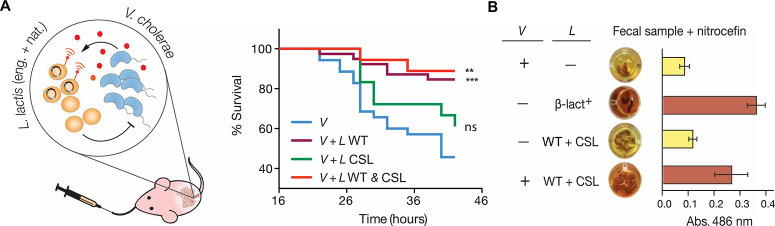
L. lactis intervention for combined cholera protection and detection. (A) Left: Strategy of mixed population of natural and engineered L. lactis for cholera protection and detection. The natural lactic acid bacteria acidify the intestinal environment to antagonize the proliferation of the pathogen. The engineered probiotics detect the quorum-sensing molecule of V. cholerae and produce an easy-to-read reporter. Right: Infant mouse survival to V. cholerae infection after the intervention with single or combined L. lactis strains. n = 18, N = 4 for CSL and WT + CSL, respectively. V and V + L WT curves are overlays from Fig. 2B. Log-rank test against the mock-fed group (V): ***P = 0.005, WT coadministration; **P = 0.0034, WT and CSL; ns, CSL. (B) In vivo detection of V. cholerae using single or combined doses of L. lactis strains. Dosing scheme was performed as described in Fig. 4D. Absorbance at 486 nm was measured on three independent samples in each group. Images on the left column correspond to one representative sample. Error bars represent SEM.
Discussion
Here, we present evidence indicating that the common lactic acid bacterium L. lactis, through its natural acidification function, can be used to inhibit V. cholerae infection in a mouse model. Our experiments demonstrate that dietary interventions with metabolically active probiotic bacteria can be used as an effective strategy to induce modifications of the intestinal microenvironment that in turn lead to the enhancement of colonization resistance against a major intestinal pathogen. The infection model used in this work allows infant mice to feed ad libitum from the mother, a situation that provides them with a constant source of lactose, favoring the L. lactis–mediated production of lactic acid in the intestinal environment. The beneficial effects of ingesting metabolically active L. lactis have been observed in other animal models of cholera infection—Zamri et al. (25) noted that oral vaccination of adult rabbits with wild-type and recombinant L. lactis expressing V. cholerae membrane proteins abolished the diarrheal symptoms of the cholera infection. Although antimicrobial properties of lactic acid have been widely applied as a food preservation method (26), our findings uncover that regular ingestion of metabolically active L. lactis represents an effective means to deliver lactic acid to the intestinal environment and create inhospitable conditions for V. cholerae proliferation. Future implementation of probiotic interventions with L. lactis in humans will require a formulation system that ensures the delivery of sufficient quantities of probiotic bacteria and key metabolic substrates so that lactic acid production occurs regardless of the individual’s diet or intestinal conditions. The use of microbiota-modulating interventions represents an emerging strategy that might confer rapid protection against fast-developing infections like cholera. Notably, Hubbard et al. (27) created a live cholera vaccine candidate that provides colonization resistance in a probiotic-like fashion, conferring cholera protection within 1 day in an infant rabbit model.
Here, we have also demonstrated the rational design and development of a synthetic gene circuit that enables L. lactis to detect and respond to V. cholerae both in vitro and in the intestinal environment. We further coupled the circuit to a proof-of-concept enzyme-based assay that reports the in situ detection of the pathogen through a simple colorimetric assay on stools, demonstrating the potential of engineered probiotics as point-of-need diagnostic tools. Our results indicate that in situ biosensing and reporting of the pathogen presence is feasible through the use of synthetic gene networks and stable, orthogonal reporter enzymes that can survive the intestinal environment. Future application of such living diagnostics in the field will entail optimization of the response time to readable output to obtain diagnostic results within clinically relevant time scales. Furthermore, there are additional technological and regulatory limitations that need to be addressed before in vivo diagnostic systems are suitable for deployment in cholera endemic areas. The β-lactamase–based enzymatic reporter used here as signal output is suitable for laboratory experiments; however, its use in the field is restricted due to potential horizontal gene transfer of the β-lactamase gene to pathogenic bacteria and cost-effectiveness of the use of its substrate, nitrocefin. Therefore, there is a need for the development of a food-grade enzymatic reporter that is orthogonal to the human intestine and whose cognate substrate can be produced cost-effectively and distributed en masse in low-resource settings.
Our work sets the stage for dietary interventions with natural and engineered probiotics as an alternative strategy to combat the spread of cholera in vulnerable populations. Household contact with infected individuals is among the highest known risk factors for subsequent infection and dehydration (28). Regular consumption of affordable, home-fermented food products containing live L. lactis may not only diminish the severity of the disease through colonization resistance, but if used in combination with a living di-agnostic L. lactis strain, it may also provide an opportune alert for patients to seek further medical intervention and a timely warning for close contacts to apply sanitary measures to prevent transmission. The use of biologics-based prophylaxis, such as the probiotic intervention presented in this study or the recently proposed phage-based prophylaxis (29), could serve as an alternative to convention-al chemoprophylaxis, limiting the emergence of antibiotic resistance in the population (28). The implementation of engineered probiotics, such as the L. lactis CSL strain, has the potential to provide community-level surveillance of cholera cases and could help to elucidate the contribution of asymptomatic carriage of V. cholerae to disease transmission (30). The development of living diagnostic bacteria (31–35) represents a promising approach to achieve near real-time surveillance of multiple pathological conditions. Our work advances this endeavor by demonstrating the design and construction of a hybrid sensing and reporting system, which provides a molecular architecture strategy for generalizable “sense-and-respond” bioengineering, expanding the possible targets of detection and the variety of engineerable sensor bacteria.
Materials And Methods
Study design
The overall objective of this work was to demonstrate that microbiota-modulating interventions with natural and engineered probiotics might represent a plausible strategy to achieve two high-priority goals in the fight against cholera, namely, enhancement of colonization resistance and point-of-need diagnostics for disease surveil-lance. We hypothesized that the transient colocalization of ingested L. lactis and V. cholerae could be leveraged to promote colonization resistance through the in situ production of lactic acid and to detect the presence of the pathogen at an early stage of the infection. We used a synthetic biology approach to construct a hybrid-signaling pathway in L. lactis that enables the detection of quorum-sensing signals that are indicative of V. cholerae infection. After in vitro validation of these hypotheses, we used the infant mouse model of cholera infection to evaluate the efficacy of the proposed dietary intervention using survival, intestinal colonization, and production of diagnostic enzymatic reporters as the principal outcomes. To optimize the number of mice needed in each trial without compromising the power of the study, we performed the survival studies in a randomized cross-litter manner (except for the three whole-litter studies in fig. S5). Each trial was performed with at least three litters. Each litter was randomly divided into two to three groups with a random number generator. Each group was assigned to a different treatment. Overall, each treatment covered individual mice from at least three litters, and data were collected over two or more independent trials [apart from the heat-inactivated (Fig. 2F) and ldh-complemented (Fig. 2E) L. lactis tests, each from one independent trial]. No blinding was performed in either experiments or data analysis. Sample sizes were determined by power analysis to reach a statistical power of 0.8 or higher using the pwr package in R.
All mice in this study were treated in accordance with protocol number IS00000852, approved by Harvard Medical School Institutional Animal Care and Use Committee and the Committee on Microbiological Safety. Primary data are reported in table S1. A detailed description of all experimental procedures performed in this study is included in the Supplementary Materials and Methods.
Statistical analysis
Statistics were performed with GraphPad Prism 5.0c. Log-rank tests were performed for all survival studies, assuming the mice in each group were independent and the hazard ratio was consistent over the study period. Each treatment group was predefined randomly cross litters at the beginning of the study (details in the Supplementary Materials and Methods). Mann-Whitney test was used where each data point was independent from each other, and no Gaussian distribution was assumed. P values reported in the figure legends are two-tailed. Specific sample size, P value, and other details for each test are described in the figure legends.
Supplementary Material
Acknowledgments:
We are grateful to W. Zhao (Mekalanos Lab, Harvard Medical School) and A. Graveline (Wyss Institute) for help on the animal protocol setup. We thank F. Hussain and D. VanInsberghe (Polz Lab, Massachusetts Institute of Technology) for providing us with environmental Vibrio spp. strains. We also thank X. Tan, M. Takahashi, and S. Slomovic for the helpful discussions.
Funding:
This work was supported by funding from the Defense Threat Reduction Agency grant HDTRA1-14-1-0006 (to J.J.C.), the Gates Foundation #OPP1058951 (to D.E.C), and the Paul G. Allen Frontiers Group (to J.J.C.).
Author contributions:
J.J.C., D.E.C., A.C.-R., and N.M. conceived the project. N.M. and A.C.-R. designed and performed the colonization resistance studies. N.M. and D.E.C. designed and performed the diagnostic circuit experiments. N.M., D.E.C., and A.C.-R. designed the animal experiments. N.M. performed the animal experiments and analyzed the data. All the authors wrote and edited the manuscript.
Competing interests:
J.J.C. is the scientific co-founder of Synlogic, a biotech startup focused on developing engineered probiotics to treat disease. Patent disclosure: Synthetic hybrid receptor and genetic circuit in probiotic bacteria to detect enteric pathogenic microorganisms. Inventors: D.E.C., N.M., and J. J.C. (U.S. provisional application no. 62/172,971; filed on 9 June 2015).
Data and materials availability:
Primary data for experiments presented here are in the Supplementary Materials. Correspondence and requests for materials should be addressed to J.J.C.
References And Notes
Full text links
Read article at publisher's site: https://doi.org/10.1126/scitranslmed.aao2586
Read article for free, from open access legal sources, via Unpaywall:
https://stm.sciencemag.org/content/scitransmed/10/445/eaao2586.full.pdf
Citations & impact
Impact metrics
Article citations
Bioengineered Probiotics for Clostridioides difficile Infection: An Overview of the Challenges and Potential for This New Treatment Approach.
Probiotics Antimicrob Proteins, 12 Nov 2024
Cited by: 0 articles | PMID: 39531149
Review
Bacterial live therapeutics for human diseases.
Mol Syst Biol, 23 Oct 2024
Cited by: 0 articles | PMID: 39443745
Review
Engineering bacterial theranostics: from logic gates to <i>in vivo</i> applications.
Front Bioeng Biotechnol, 12:1437301, 18 Sep 2024
Cited by: 0 articles | PMID: 39359265 | PMCID: PMC11444965
Review Free full text in Europe PMC
Accelerating Genetic Sensor Development, Scale-up, and Deployment Using Synthetic Biology.
Biodes Res, 6:0037, 25 Jun 2024
Cited by: 0 articles | PMID: 38919711 | PMCID: PMC11197468
Effects of Probiotics on Gut Microbiota: An Overview.
Int J Mol Sci, 25(11):6022, 30 May 2024
Cited by: 5 articles | PMID: 38892208 | PMCID: PMC11172883
Review Free full text in Europe PMC
Go to all (106) article citations
Data
Data behind the article
This data has been text mined from the article, or deposited into data resources.
BioStudies: supplemental material and supporting data
Similar Articles
To arrive at the top five similar articles we use a word-weighted algorithm to compare words from the Title and Abstract of each citation.
Anti-biofilm Properties of the Fecal Probiotic Lactobacilli Against Vibrio spp.
Front Cell Infect Microbiol, 8:120, 24 Apr 2018
Cited by: 57 articles | PMID: 29740541 | PMCID: PMC5928150
Members of the human gut microbiota involved in recovery from Vibrio cholerae infection.
Nature, 515(7527):423-426, 17 Sep 2014
Cited by: 208 articles | PMID: 25231861 | PMCID: PMC4353411
Effects of Lactococcus lactis subsp. lactis JCM5805 on colonization dynamics of gut microbiota and regulation of immunity in early ontogenetic stages of tilapia.
Fish Shellfish Immunol, 86:53-63, 11 Nov 2018
Cited by: 14 articles | PMID: 30428393
Curbing cholera.
Sci Transl Med, 10(445):eaat9483, 01 Jun 2018
Cited by: 4 articles | PMID: 29899025
Review
Funding
Funders who supported this work.
Bill and Melinda Gates Foundation
Defense Threat Reduction Agency (1)
Grant ID: HDTRA1-14-1-0006





