Abstract
Free full text

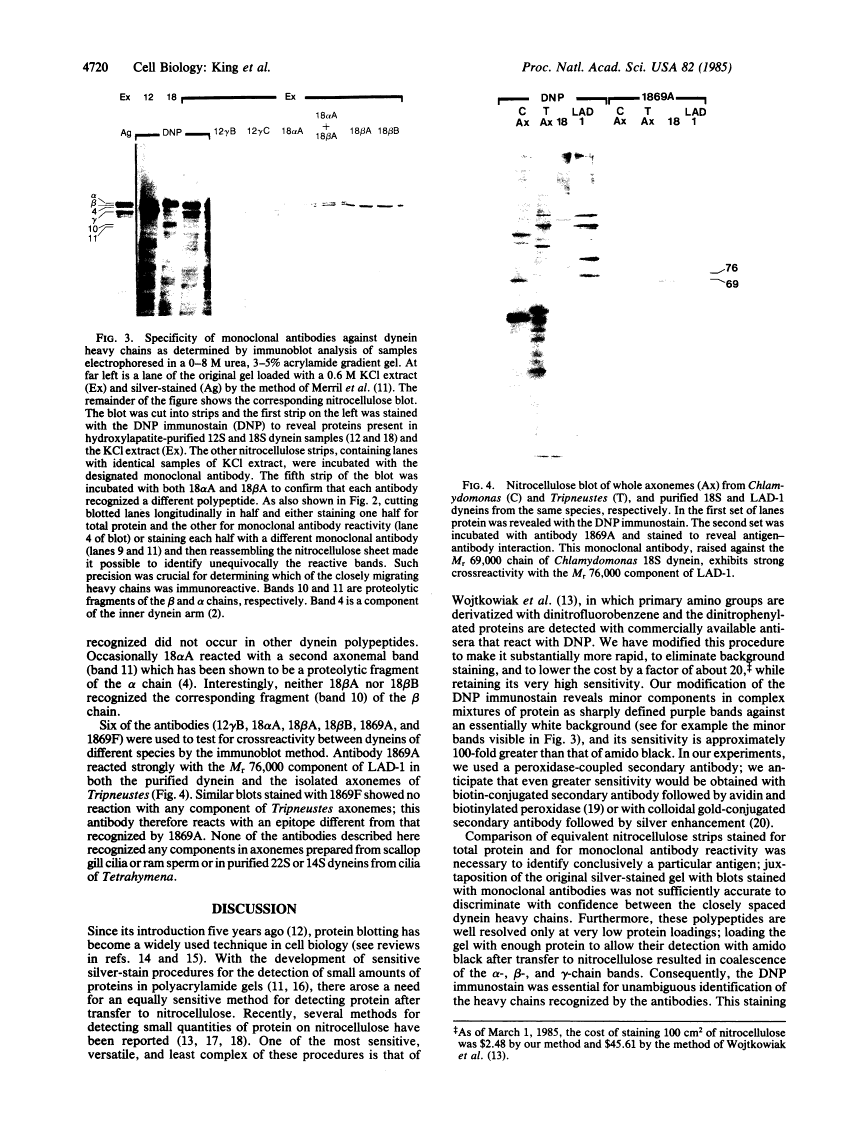
Characterization of monoclonal antibodies against Chlamydomonas flagellar dyneins by high-resolution protein blotting.
Abstract
Monoclonal antibodies that recognize individual polypeptides of the outer arm dyneins of Chlamydomonas flagella were obtained and used to study the structural relationships between the various polypeptides. Immunoblot analysis showed that the gamma heavy chain of 12S dynein and the alpha and beta heavy chains and Mr 69,000 intermediate chain of 18S dynein each contain immunoreactive sites not found in the other dynein chains or in any other axonemal protein. We also used these antibodies to investigate possible structural similarities between dynein polypeptides from Chlamydomonas and phylogenetically distant species. No crossreactivity was observed with antibodies against either the alpha, beta, or gamma heavy chains, demonstrating that each Chlamydomonas heavy chain has structural features distinct from those present in dyneins from the other species tested. However, one antibody against the Mr 69,000 polypeptide recognized an intermediate chain (Mr 76,000) of latent-activity dynein-1 from the sea urchin Tripneustes gratilla. This result provides further evidence that 18S dynein and latent-activity dynein-1 are related. In the course of the above studies, we modified existing procedures to achieve efficient transfer of high molecular weight proteins from NaDodSO4/polyacrylamide gels to nitrocellulose sheets, and to detect small quantities of protein on nitrocellulose. Our modified procedure for staining total protein on nitrocellulose is rapid, inexpensive, and as sensitive as silver-staining of polyacrylamide gels. These methods should be useful to investigators working with small amounts of protein or requiring resolution of closely migrating polypeptides after transfer to nitrocellulose.
Full text
Full text is available as a scanned copy of the original print version. Get a printable copy (PDF file) of the complete article (1.5M), or click on a page image below to browse page by page. Links to PubMed are also available for Selected References.
Images in this article
Click on the image to see a larger version.
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Piperno G, Luck DJ. Axonemal adenosine triphosphatases from flagella of Chlamydomonas reinhardtii. Purification of two dyneins. J Biol Chem. 1979 Apr 25;254(8):3084–3090. [Abstract] [Google Scholar]
- Pfister KK, Fay RB, Witman GB. Purification and polypeptide composition of dynein ATPases from Chlamydomonas flagella. Cell Motil. 1982;2(6):525–547. [Abstract] [Google Scholar]
- Pfister KK, Haley BE, Witman GB. The photoaffinity probe 8-azidoadenosine 5'-triphosphate selectively labels the heavy chain of Chlamydomonas 12 S dynein. J Biol Chem. 1984 Jul 10;259(13):8499–8504. [Abstract] [Google Scholar]
- Pfister KK, Witman GB. Subfractionation of Chlamydomonas 18 S dynein into two unique subunits containing ATPase activity. J Biol Chem. 1984 Oct 10;259(19):12072–12080. [Abstract] [Google Scholar]
- Tash JS, Means AR. Regulation of protein phosphorylation and motility of sperm by cyclic adenosine monophosphate and calcium. Biol Reprod. 1982 May;26(4):745–763. [Abstract] [Google Scholar]
- Harrison RA. Glycolytic enzymes in mammalian spermatozoa. Activities and stabilities of hexokinase and phosphofructokinase in various fractions from sperm homogenates. Biochem J. 1971 Oct;124(4):741–750. [Europe PMC free article] [Abstract] [Google Scholar]
- Galfrè G, Milstein C. Preparation of monoclonal antibodies: strategies and procedures. Methods Enzymol. 1981;73(Pt B):3–46. [Abstract] [Google Scholar]
- Köhler G, Howe SC, Milstein C. Fusion between immunoglobulin-secreting and nonsecreting myeloma cell lines. Eur J Immunol. 1976 Apr;6(4):292–295. [Abstract] [Google Scholar]
- Laemmli UK, Favre M. Maturation of the head of bacteriophage T4. I. DNA packaging events. J Mol Biol. 1973 Nov 15;80(4):575–599. [Abstract] [Google Scholar]
- Merril CR, Goldman D, Sedman SA, Ebert MH. Ultrasensitive stain for proteins in polyacrylamide gels shows regional variation in cerebrospinal fluid proteins. Science. 1981 Mar 27;211(4489):1437–1438. [Abstract] [Google Scholar]
- Towbin H, Staehelin T, Gordon J. Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedure and some applications. Proc Natl Acad Sci U S A. 1979 Sep;76(9):4350–4354. [Europe PMC free article] [Abstract] [Google Scholar]
- Wojtkowiak Z, Briggs RC, Hnilica LS. A sensitive method for staining proteins transferred to nitrocellulose sheets. Anal Biochem. 1983 Mar;129(2):486–489. [Abstract] [Google Scholar]
- Gershoni JM, Palade GE. Protein blotting: principles and applications. Anal Biochem. 1983 May;131(1):1–15. [Abstract] [Google Scholar]
- Towbin H, Gordon J. Immunoblotting and dot immunobinding--current status and outlook. J Immunol Methods. 1984 Sep 4;72(2):313–340. [Abstract] [Google Scholar]
- Wray W, Boulikas T, Wray VP, Hancock R. Silver staining of proteins in polyacrylamide gels. Anal Biochem. 1981 Nov 15;118(1):197–203. [Abstract] [Google Scholar]
- Kittler JM, Meisler NT, Viceps-Madore D, Cidlowski JA, Thanassi JW. A general immunochemical method for detecting proteins on blots. Anal Biochem. 1984 Feb;137(1):210–216. [Abstract] [Google Scholar]
- Yuen KC, Johnson TK, Denell RE, Consigli RA. A silver-staining technique for detecting minute quantities of proteins on nitrocellulose paper: retention of antigenicity of stained proteins. Anal Biochem. 1982 Nov 1;126(2):398–402. [Abstract] [Google Scholar]
- Ogata K, Arakawa M, Kasahara T, Shioiri-Nakano K, Hiraoka K. Detection of toxoplasma membrane antigens transferred from SDS-polyacrylamide gel to nitrocellulose with monoclonal antibody and avidin-biotin, peroxidase anti-peroxidase and immunoperoxidase methods. J Immunol Methods. 1983 Dec 16;65(1-2):75–82. [Abstract] [Google Scholar]
- Glenney JR, Jr, Glenney P. Fodrin is the general spectrin-like protein found in most cells whereas spectrin and the TW protein have a restricted distribution. Cell. 1983 Sep;34(2):503–512. [Abstract] [Google Scholar]
- Gardner JM, Fambrough DM. Fibronectin expression during myogenesis. J Cell Biol. 1983 Feb;96(2):474–485. [Europe PMC free article] [Abstract] [Google Scholar]
- Bloom GS, Schoenfeld TA, Vallee RB. Widespread distribution of the major polypeptide component of MAP 1 (microtubule-associated protein 1) in the nervous system. J Cell Biol. 1984 Jan;98(1):320–330. [Europe PMC free article] [Abstract] [Google Scholar]
- Kiehart DP, Kaiser DA, Pollard TD. Monoclonal antibodies demonstrate limited structural homology between myosin isozymes from Acanthamoeba. J Cell Biol. 1984 Sep;99(3):1002–1014. [Europe PMC free article] [Abstract] [Google Scholar]
- Bell CW, Fronk E, Gibbons IR. Polypeptide subunits of dynein 1 from sea urchin sperm flagella. J Supramol Struct. 1979;11(3):311–317. [Abstract] [Google Scholar]
- Johnson KA, Porter ME, Shimizu T. Mechanism of force production for microtubule-dependent movements. J Cell Biol. 1984 Jul;99(1 Pt 2):132s–136s. [Europe PMC free article] [Abstract] [Google Scholar]
- Tang WJ, Bell CW, Sale WS, Gibbons IR. Structure of the dynein-1 outer arm in sea urchin sperm flagella. I. Analysis by separation of subunits. J Biol Chem. 1982 Jan 10;257(1):508–515. [Abstract] [Google Scholar]
- Goodenough U, Heuser J. Structural comparison of purified dynein proteins with in situ dynein arms. J Mol Biol. 1984 Dec 25;180(4):1083–1118. [Abstract] [Google Scholar]
- Goodenough UW, Heuser JE. Substructure of the outer dynein arm. J Cell Biol. 1982 Dec;95(3):798–815. [Europe PMC free article] [Abstract] [Google Scholar]
Associated Data
Articles from Proceedings of the National Academy of Sciences of the United States of America are provided here courtesy of National Academy of Sciences
Full text links
Read article at publisher's site: https://doi.org/10.1073/pnas.82.14.4717
Read article for free, from open access legal sources, via Unpaywall:
https://www.pnas.org/content/pnas/82/14/4717.full.pdf
Citations & impact
Impact metrics
Citations of article over time
Article citations
A dynein-associated photoreceptor protein prevents ciliary acclimation to blue light.
Sci Adv, 7(9):eabf3621, 26 Feb 2021
Cited by: 6 articles | PMID: 33637535 | PMCID: PMC7909887
Mutations in PIH proteins MOT48, TWI1 and PF13 define common and unique steps for preassembly of each, different ciliary dynein.
PLoS Genet, 16(11):e1009126, 03 Nov 2020
Cited by: 11 articles | PMID: 33141819 | PMCID: PMC7608865
WDR92 is required for axonemal dynein heavy chain stability in cytoplasm.
Mol Biol Cell, 30(15):1834-1845, 22 May 2019
Cited by: 18 articles | PMID: 31116681 | PMCID: PMC6727741
Proteome of the central apparatus of a ciliary axoneme.
J Cell Biol, 218(6):2051-2070, 15 May 2019
Cited by: 34 articles | PMID: 31092556 | PMCID: PMC6548120
Chlamydomonas DYX1C1/PF23 is essential for axonemal assembly and proper morphology of inner dynein arms.
PLoS Genet, 13(9):e1006996, 11 Sep 2017
Cited by: 25 articles | PMID: 28892495 | PMCID: PMC5608425
Go to all (65) article citations
Similar Articles
To arrive at the top five similar articles we use a word-weighted algorithm to compare words from the Title and Abstract of each citation.
Protein-protein interactions in the 18S ATPase of Chlamydomonas outer dynein arms.
Cell Motil Cytoskeleton, 6(5):510-520, 01 Jan 1986
Cited by: 39 articles | PMID: 2947699
Subfractionation of Chlamydomonas 18 S dynein into two unique subunits containing ATPase activity.
J Biol Chem, 259(19):12072-12080, 01 Oct 1984
Cited by: 55 articles | PMID: 6237107
Outer-arm dynein from trout spermatozoa: substructural organization.
Cell Motil Cytoskeleton, 16(4):266-278, 01 Jan 1990
Cited by: 17 articles | PMID: 1697510
Dynein motors of the Chlamydomonas flagellum.
Int Rev Cytol, 210:227-268, 01 Jan 2001
Cited by: 49 articles | PMID: 11580207
Review
Funding
Funders who supported this work.
NCI NIH HHS (1)
Grant ID: CA12708
NIGMS NIH HHS (2)
Grant ID: GM30626
Grant ID: GM08986