Abstract
Free full text

Does serotonin deficiency lead to anosmia, ageusia, dysfunctional chemesthesis and increased severity of illness in COVID-19?
Abstract
Different mechanisms forwarded to understand anosmia and ageusia in coronavirus patients are not adequate to explain reversible anosmia and ageusia, which are resolved quickly. In addition, the reason behind the impaired chemesthetic sensations in some coronavirus patients remains unknown. In the present paper it is proposed that SARS-CoV-2 patients suffer from depletion of tryptophan, as ACE2, a key element in the process of absorption of tryptophan from the food, is significantly reduced in the patients as coronavirus uses ACE2 as the receptor to enter the host cells. The tryptophan depletion leads to a deficit of serotonin (5-HT) in SARS-COV-2 patients because tryptophan is the precursor in the synthesis of 5-HT. Such 5-HT deficiency can explain anosmia, ageusia and dysfunctional chemesthesis in COVID-19, given the fact that 5-HT is an important neuromodulator in the olfactory neurons, taste receptor cells and transient receptor potential channels (TRP channels) involved in chemesthesis. In addition, 5-HT deficiency worsens silent hypoxemia and depresses hypoxic pulmonary vasoconstriction leading to increased severity of the disease. Also, the levels of anti-inflammatory melatonin (synthesized from 5-HT) and nicotinamide adenine dinucleotide (NAD+, produced from niacin whose precursor is the tryptophan) might decrease in coronavirus patients resulting in the aggravation of the disease. Interestingly, selective serotonin reuptake inhibitors (SSRIs) may not be of much help in correcting the 5-HT deficiency in COVID-19 patients, as their efficacy goes down significantly when there is depletion of tryptophan in the system. Hence, tryptophan supplementation may herald a radical change in the treatment of COVID-19 and accordingly, clinical trials (therapeutic / prophylactic) should be conducted on coronavirus patients to find out how tryptophan supplementation (oral or parenteral, the latter in severe cases where there is hardly any absorption of tryptophan from the food) helps in curing, relieving or preventing the olfactory, gustatory and chemesthetic dysfunctions and in lessening the severity of the disease.
Introduction
Anosmia (complete loss of ability to smell) or hyposmia/microsmia (reduced ability to smell) has been found to be a telltale symptom of COVID-19 and probably, is the early marker of the coronavirus disease [1], [2]. Such olfactory dysfunction, in many cases, is associated with gustatory dysfunction like ageusia (complete loss of ability to taste) or hypogeusia (reduced ability to taste) and multiple cross-sectional studies [2], [3], [4], [5], [6], [7], [8], [9] have demonstrated a wide variation of the incidence rate (depending on the gender, country and methodologies like self-report or testing etc.) of olfactory (5–95%) and gustatory dysfunction (38–89%) in coronavirus patients with female predominance. Many patients regain their sense of smell and taste within a week or so (especially in mild and moderate cases), however, in a minority of patients the problems linger for months and may be associated with other symptoms (long COVID) [10], [11].
Different mechanisms have been proposed to explain the olfactory dysfunctions of coronavirus patients. First, though many respiratory viruses can lead to the loss of smell owing to nasal congestion, COVID-19 anosmia is unusual because it often happens without any accompanying congestion [8], [11], [12], [13]. Hence, nasal congestion as the cause of anosmia in coronavirus patients can be ruled out.
Second, does anosmia then result from coronavirus induced damage of the olfactory neurons? It is known [14], [15] that the entry of coronavirus into host cells depends on the presence of cell receptor ACE2 as well as transmembrane protease serine 2 (TMPRSS2) and, incidentally, olfactory neurons do not have ACE2 receptors and cell surface TMPRSS2. However, it has been found that the supporting cells (non-neuronal cells in the olfactory epithelium), particularly sustentacular cells (and other support like horizontal basal cells, Bowman’s gland, and microvillar cells), which express both ACE2 and TMPRSS2, can get infected with coronavirus leading to collateral damage and death of olfactory neurons [13], [14], [15], [16]. However, in addition to ACE2 and TMPRSS2, neuropilin-1 (NRP1), a transmembrane receptor which exhibits high expression in the olfactory (and respiratory) epithelium can facilitate SARS-CoV-2 entry [17], [18], but its consequence on neuronal damage is yet to be studied. Hence, one of the reasons of anosmia (especially in severe cases) can be coronavirus induced collateral olfactory nerve damage.
Third, the next question is whether SARS-COV-2 can reach the brain and damage the olfactory bulb and associated structure [19], [20] resulting in anosmia. It has been demonstrated [21] from the tissue biopsies of deceased old patients that transmucosal neuroinvasion can take place at the nasal neural–mucosal interface followed by the transport of the virus along the olfactory tract of the CNS to the brain. The researchers found viral RNA at the mucus of the nasal cavity and virus in the blood vessels of the brain as well as spike proteins in the brain along with neuron marker cells. However, the studies precluded such organ damage in mild or moderate cases.
Fourth, some workers proposed that inflammation markers (cytokine storm) like interleukin-6 [22], CXCL-10 [23], and TNF-α [24] can damage the olfactory neurons. Even bradykinin (a peptide that dilates blood vessels and makes them leaky) storm [25] has been suggested to explain the neural and non-neural damages by coronavirus probably leading to anosmia.
Surprisingly, the recovery after anosmia is often quicker (especially in mild cases) than the time it takes for neuron replacement, cilia maturation, and the growth of the new axons from the olfactory epithelium to form synapses in the olfactory bulb [26], [27]. From the brain imaging studies (CT, MRI, PET-MRI [28], [29], [30]), olfactory bulb/cleft abnormalities have been observed in severe cases of anosmia only (mostly persistent anosmia). For mild infections where anosmia resolved within a week or so, cytokine/bradykinin storm may also be ruled out as the cause of anosmia. Hence, the above explanations may not be satisfactory for fast resolved reversible anosmia in mild / moderate cases.
Like olfactory neurons, taste receptor cells, which identify chemicals in the saliva and delivers signals to the brain, do not contain ACE2, and thus, they probably are not infected by coronavirus [16], [31], [32]. But other support cells in the tongue, which carry the receptors can get infected and can inflict collateral damage on the taste receptor cells [16], [31], [32]. Also, cytokine induced taste receptor cell damage [22] can result in ageusia/hypogeusia. Though taste may partially disappear if olfaction is disrupted, a lot of COVID-19 patients develop true ageusia and cannot identify even a salty or sweet taste and the explanation still eludes the researchers [31].
In addition, it remains unexplained why many coronavirus patients lose the ability to ‘taste’ the burn of chili peppers or the cold feeling of mint where such sensations (chemesthesis, chemically induced sensations of the skin and mucus membranes, where taste receptor cells are not involved), are conveyed to brainstem by nociceptive (sensing pain from noxious stimuli) trigeminal neurons which do not express ACE2 or TRMPSS2 [16], [31], [32], [33].
Hypothesis: Formation of serotonin deficiency state in coronavirus patients
Interestingly, all the above apparently intriguing aspects of COVID-19 can be explained by considering the role of the ‘happy hormone” serotonin (5-HT) in coronavirus patients. 5-HT, also known as 5-hydroxytryptamine, is a neurotransmitter, a signaling molecule that affects all the major organ systems including the cardiovascular, pulmonary, gastrointestinal, genitourinary, and on top of these, the central nervous system [34], [35], [36]. 5-HT and 5-HT receptors are important in the regulation of virtually all brain functions and there are at least fifteen 5-HT receptor types, which are grouped into seven families based on their signaling mechanisms. The majority of 5-HT synthesis (up to 90%) takes place in the gastrointestinal enterochromaffin cells (and stored in platelets) and the peripheral 5-HT primarily regulates intestinal motility, metabolism and hemodynamics. In the central nervous system (CNS), 5-HT is mostly produced in the neurons of the brainstem raphe nuclei and released into synapses to act as a neuromodulator of many brain circuits. 5-HT in mammalian organism exists in two functionally independent compartments, central and peripheral, which are separated by the blood–brain barrier (BBB).
Incidentally, 5-HT is synthesized from tryptophan, an essential amino acid, which is obtained from the food and in the process of absorption of tryptophan, ACE2 plays a key role and its deficiency leads to a substantial reduction of 5-HT in the body [37], [38]. Neutral amino acids like tryptophan are transported by B0AT1(broad neutral (0) amino acid transporter 1), a protein that mediates transfer of amino acids into and out of cells in the intestine, where ACE2 performs a crucial job as a chaperone (a protein that helps other proteins in folding or unfolding) for B0AT1 [39], [40]. Though ACE2 is abundantly present in the epithelial cells of the lungs where SARS-CoV-2 predominantly docks, intestinal infection with coronavirus is also very common [41] as ACE2 is present significantly in the enterocytes of small intestine. Around 70% of COVID-19 patients show GI symptoms and the actual percentage of the patients with gastrointestinal involvement should be much higher as coronavirus RNA has been found from the anal/rectal swabs of many asymptomatic patients [41]. Hence, SARS-CoV-2 induced downregulation of intestinal ACE2 should lead to a very low uptake of tryptophan from the food with consequent 5-HT deficiency state in COVID-19 patients. As stated earlier, 5-HT cannot cross the BBB and the central 5-HT is synthesized in the brainstem only from the absorbed tryptophan, which reaches the brainstem by crossing the BBB as free tryptophan [42], given the fact that tryptophan circulates as bound tryptophan (loosely bound to albumin) which dissociates from the albumin near the BBB. Fig. 1 schematically depicts how, as per the proposed hypothesis, serotonin deficiency state arises in coronavirus patients.
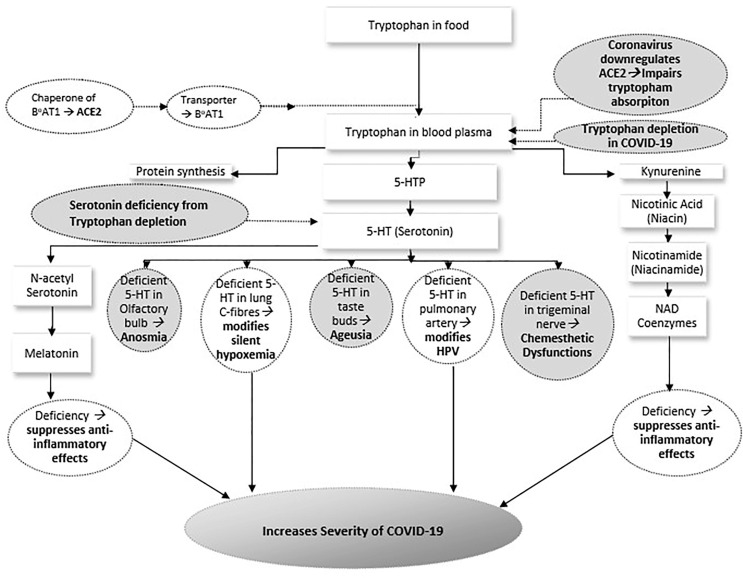
Schematic outline of the proposed hypothesis depicting how in coronavirus patients serotonin deficiency state arises, and how it explains anosmia, ageusia, chemesthetic dysfunctions and increased severity of illness in COVID-19 [B0AT1 → Broad neutral (0) Amino acid Transporter 1, ACE2 → Angiotensin-Converting Enzyme 2, TRP → Transient Receptor Potential, 5-HTP → 5-Hydroxytryptophan, 5-HT → 5-Hydroxytryptamine (Serotonin), NAD → Nicotinamide Adenine Dinucleotide, HPV → Hypoxic Pulmonary Vasoconstriction]
Implications and importance of the hypothesis
Evidence of serotonin deficiency in coronavirus patients
Interestingly, the hypothesis of 5-HT deficiency state in COVID-19 has been vindicated in a recent study [43], where it has been found that the 5-HT levels in coronavirus patients are indeed reduced and the researchers suggested [43] that the reduction of 5-HT is probably due to the alteration in enterochromaffin cell functions or increased 5-HT degradation by monoamine oxidase (MAO, a family of enzymes where MAO-A is specifically involved in the process of breaking down of 5-HT to 5-hydroxyindoleacetic acid, 5-HIAA). In another study [44], an apparent increase in plasma 5-HT level in a cohort has been attributed to the possibility of delayed or insufficient breakdown of 5-HT to 5-HIAA in the liver, although, in such cases, the brain 5-HT level (central 5-HT) should remain unaffected as 5-HT cannot cross the BBB. However, none of the studies considered the possibility of 5-HT deficiency state arising from tryptophan depletion as suggested in the present hypothesis.
Role of serotonin deficiency in anosmia
It is worth noting that 5-HT (specifically, central 5-HT) is intricately associated with olfaction and it increases the synaptic activity in olfactory bulb glomeruli (globular tangle of axons in the olfactory epithelium) [45]. The olfactory bulb receives dense serotonergic innervation particularly from the median raphe nucleus in brainstem to the glomeruli. It has been found that 5-HT plays an important role in exciting tufted cells and depolarizing some mitral cells, given the fact that the mitral cells and tufted cells are the principal neurons in the olfactory bulb and they receive the olfactory sensory inputs, process the signals in the glomeruli and finally send the sensory information to the olfactory cortex [46], [47]. In the olfactory bulb 5-HT is mainly involved in adjusting the sensory gain and given the presence of a variety of 5-HT receptor types in the olfactory bulb, the role of 5-HT in odour detection and differentiation is a bit complex in the sense that the mitral cells can be in excitatory, inhibitory or in a mixed mode (inhibition followed by excitation) [47], [48], [49]. The overall effect of 5-HT on mitral cells is generally bidirectional (excitatory and inhibitory) and the apparent inhibitory effect probably increases the discriminability between odors or favor the perception of some odors over others [50] by gating out the sensory information that might otherwise compete for attention [51] and/or by blocking the spontaneous firing of olfactory cortical neurons (sparing the sensory-driven firing) resulting in improved signal-to-noise ratio [52], [53]. Hence, 5-HT has got an important role in olfaction.
Incidentally, in the literature, studies on the role of 5-HT in modulating the olfactory functions in humans are scarcely available. Depression, a low 5-HT state, is often associated with reduced olfactory function [54], [55] and fluoxetine [54], a selective serotonin reuptake inhibitor (SSRI), and venlafaxine [56], a serotonin-norepinephrine reuptake inhibitor (SNRI), have been reported to regenerate the lost olfactory acuity in depressed patients along with the improvement in depression. Patients of obstructive sleep apnea (also a low 5-HT state) show reduced olfactory function which improves after treatment with SSRIs [57], [58], [59]. An interesting observation is the improvement in olfactory sensitivity during fasting [60] and, incidentally, 5-HT in fasting state is significantly increased [61]. Another interesting observation is the disruption of olfactory (and also gustatory) functions at high altitude [62], [63] and it is known that low oxygen at high altitude lowers 5-HT as the synthesis of 5-HT from tryptophan is hindered because the enzyme tryptophan hydroxylase involved in the synthesis of 5-HT requires molecular oxygen [64], [65]. Also, anosmia or hyposmia is more frequent in migraineurs [66] who have chronic low 5-HT level (the 5-HT level only rises during the attack) [67]. Hence, as argued above, it can be suggested that in COVID-19, 5-HT deficiency due to tryptophan depletion is mainly responsible for the olfactory deficit though the possibility of neuronal damage and dysfunctional olfactory bulb (and associated structure) should be taken into consideration for severe cases when the patients suffer more prolonged olfactory disorder.
Role of serotonin deficiency in ageusia
A little is known about the mechanism behind the gustatory dysfunctions of SARS-COV-2 patients as ACE2 is only expressed in the epithelial cells of the tongue but not in the taste buds (groups of taste receptor cells) [16], [32], [68]. Incidentally, 5-HT is also an important neurotransmitter / neuromodulator in taste buds [69], [70] and hence, 5-HT is expected to modulate the gustatory functions too. It has been reported that dysgeusia (altered sense of taste) in a depressed patient is relieved by sertraline (an SSRI) [71]. There are at least two reports [72], [73] where enhancing 5-HT by prescribing SSRIs significantly improved the gustatory sensitivity of the volunteers towards bitter and sweet tastes. It is worth mentioning that many COVID-19 patients lose the ability to specifically identify bitter and sweet tastes [74], and as mentioned above, the sensitivity towards the bitter and sweet tastes is expected to degrade when the 5-HT level is low [72], [73]. This interesting correlation strongly supports the proposition that COVID-19 leads to a 5-HT deficiency state and ageusia also arises from the deficit of 5-HT in coronavirus patients.
Another interesting finding is the wide variation of the incidence rate of anosmia and ageusia amongst the patients [2], [3], [4], [5], [6], [7], [8], [9] and female predominance [2], [75]. Incidentally, the 5-HT level in human varies widely (normal range 65–250 ng/ml in whole blood and 5.6–23.9 ng/ml in plasma [76]) and hence, those patients whose 5-HT levels are near the lower limit of the range should be more susceptible to getting anosmia and/or ageusia than those who have high serotonin levels. In addition, as discussed earlier, although intestinal infection is very common in COVID-19, a small percentage of the patients probably remains free from intestinal infection and consequently, they are protected from tryptophan depletion and, hence, may not suffer from anosmia and ageusia. The female predominance of anosmia/ageusia can be explained by considering the fact that the brain 5-HT levels of the females are generally lower than those of the males [77].
Role of serotonin deficiency in chemesthetic dysfunctions
The hitherto unexplained disruption of chemesthetic sensations of coronavirus patients [78] can also be understood by considering the deficiency of 5-HT in coronavirus patients. Chemesthetic sensations of hot pepper and cool mint activate transient receptor potential channels (TRP channels), a family of cation permeable channels (polymodal sensors), which are expressed in the taste buds, nerve fibers, and keratinocytes in the oronasal cavity [79], [80], [81]. The signals from the irritants in pepper or mint are communicated from the oral mucosa to the brain by the trigeminal nerve [79], [80], [81], [82]. Capsaicin of peppers activates TRPV1, a member of the TRP family that is involved in chemosensation (and thermosensation) [83] and another member of the TRP family, TRPM8 is activated by menthol [84]. As stated earlier nociceptive trigeminal neurons do not express ACE2 or TRMPSS2 [16], [31], [32], [33] and are not expected to be attacked by coronavirus. However, it is known [85], [86], [87], [88] that 5-HT stimulates nociceptor activity of the TRP channels by increasing calcium influx in the nociceptive sensory neurons and enhancing proinflammatory peptide release from the sensory neurons and hence, low 5-HT level in COVID-19 patients is expected to degrade chemesthetic sensations. Also, the oft-reported symptoms of brain fog (cognitive impairment) and depression [89], [90] of many coronavirus patients can be understood by considering the role of 5-HT deficiency in such patients.
Serotonin deficiency and severity of COVID-19
Role of serotonin in silent hypoxemia
5-HT is expected to play an important role in silent hypoxemia (insufficient oxygen in the blood) also called ‘happy’ hypoxia (insufficient oxygen in the tissue), an apparently intriguing clinical aspect of some COVID-19 patients, where there is a near-absence of shortness of breath or any discomfort in patients in spite of having a dangerously low level of oxygen in the body which seems to be incompatible with life [91], [92], [93], [94], [95], [96], [97], [98]. However, such cardiorespiratory compensation can fail precipitously (as a small change in alveolar PO2 can result in a significant change in oxygen saturation, given the fact that the patients’ gas exchange is on the steep portion of the oxyhemoglobin dissociation curve [91]) leading to a sudden worsening of the symptoms followed by organ damage and even death. Such silent hypoxemia in coronavirus patients is not a very unusual phenomenon as high-altitude hypoxemia [91], [99] may also remain silent and indeed, pilots [100] are trained so that they can detect insidious hypoxemia before there is any cognitive impairment.
Generally, as a compensatory response to hypoxemia, hyperventilation ensues and CO2 being much more diffusible than O2, there is a disproportional pulmonary exchange of CO2 and O2 leading to reduced CO2 in blood (hypocapnia) [96], [97], [101], [102], [103]. Incidentally, even if the blood oxygen level goes down, dyspnea may be well-nigh absent if, simultaneously, blood carbon dioxide level becomes low too (hypocapnia), as the ventilatory response to hypoxemia is blunted in presence of hypocapnia [91], [99], [100], [101], [102], [103], [104]. At the same time, mild hyperventilation may not be noticed by the patients (especially, when they are not ambulatory) though it can lead to significant hypocapnia and consequently, hypoxemia accompanied by hypocapnia may remain asymptomatic (silent hypoxemia) in coronavirus patients [91], [94], [105].
In this context, 5-HT has an important role too, as in addition to hypocapnia, low 5-HT is also expected to augment the asymptomatic manifestation of hypoxemia in COVID-19. It is known [106], [107] that the unpleasant sensory stimuli originating from the respiratory system, such as dyspnea or air hunger are detected by the lung afferent C-fibres (unmyelinated low conduction velocity nerve fibers), where TRPV1 and TRPM8 channels (the same TRP channels involved in dysfunctional chemesthesis in COVID-19 as discussed earlier) are significantly expressed. As 5-HT enhances nociceptor activity of TRP channels [80], [81], [82], [83], [84], [85], low 5-HT is expected to reduce the subjective sensation of breathlessness and can aggravate the consequences of silent hypoxemia. Hence, restoring the 5-HT level in SARS-CoV-2 patients is expected to convert the silent hypoxemia to symptomatic hypoxemia at an early stage before the cardiorespiratory compensation abruptly fails and the patients get time to seek medical treatment.
Role of serotonin in hypoxic pulmonary vasoconstriction
Serotonin is also partly involved in the severity of COVID-19 via its effect on hypoxic pulmonary vasoconstriction. Incidentally, to understand the reason(s) behind the hypoxia in coronavirus patients, different mechanisms [91], [92], [93], [94], [95], [96], [97], [98] like i) inability of the infected lungs to restrict blood flow to the damaged tissues of the lungs which cannot collect oxygen, ii) formation of intravascular microthrombi restricting blood flow, iii) pulmonary shunt (no gas exchange, i.e., entry of deoxygenated blood into the arterial system bypassing the alveolar air because of airspace collapse or fluid filled alveolar spaces) iv) ventilation-perfusion (VQ) mismatch (gas exchange unimpeded in some areas and restricted/prohibited in other areas, i.e., a mismatch between the alveolar ventilation and the alveolar blood flow) and v) increased dead space (volume of inhaled air that does not participate in gas exchange) have been forwarded. Amongst them, the first factor is related to the well-known hypoxic pulmonary vasoconstriction [108], [109], which is an important protective physiological mechanism that diverts blood flow away from the hypoxic areas of the lungs to the areas with better ventilation so as to improve the pulmonary gas exchange efficiency. Incidentally, 5-HT, which is released from pulmonary neuroendocrine cells during hypoxia, is a pulmonary vasoconstrictor [110] and hence, 5-HT deficiency is expected to disrupt pulmonary vasoconstriction and consequently, worsen COVID-19 hypoxia.
Role of serotonin / tryptophan in the synthesis of melatonin and NAD
Incidentally, 5-HT is the precursor in the synthesis of pineal and extrapineal melatonin (Fig. 1), which is well-known as a chronobiotic, hypnotic, neuroprotective and cardioprotective agent, and, of late, melatonin has been touted as a potential adjuvant in the fight against COVID-19 because of its immunomodulatory, anti-inflammatory and anti-oxidative effects [111], [112]. Consequently, 5-HT deficiency should lead to a reduction of melatonin putting the coronavirus patients at a further disadvantage.
Niacin, an important B vitamin (B3), is produced from tryptophan (and also partly absorbed from niacin rich diet) through kynurenine pathway (Fig. 1) and acts as a building block of nicotinamide adenine dinucleotide (NAD), which is an important coenzyme in various metabolic pathways. The oxidized form of NAD (NAD+) is known to lower pro-inflammatory cytokines and ventilator induced lung injury and probably, reduces viral replication too [113]. Hence, any niacin deficiency arising from tryptophan depletion can increase the severity of COVID-19.
Treatment to correct serotonin deficiency in coronavirus patients
Apparently, an administration of SSRIs is expected to boost the 5-HT level and provide much relief to the patients. However, no clinical study has been done with a view to correcting the 5-HT deficiency of coronavirus patients as the possibility of such 5-HT deficiency has not been considered earlier. Nevertheless, on the basis of the existing knowledge that some SSRIs have anti-inflammatory, antiviral, immunoregulatory and neuroprotective properties, a few studies [114], [115], [116], [117], [118] have been carried out to explore the effect of SSRIs on the coronavirus patients, where SSRIs like fluvoxamine, fluoxetine and escitalopram have been found to somewhat decrease the severity of COVID-19. This observation is in line with the present hypothesis, though, as anticipated from the hypothesis, a significant improvement is lacking.
Interestingly, only SSRI administration is not going to provide a significant improvement in COVID-19, as the key to restore the 5-HT level of coronavirus patients is to correct the tryptophan depletion first, as SSRIs are of little help in raising the 5-HT level as their efficacy drops significantly when there is tryptophan depletion in the system [119], [120]. Hence, clinical trials (both therapeutic and prophylactic) of tryptophan supplementation (oral and parenteral, the latter in severe cases where there is almost no absorption of tryptophan from the food) should be conducted on coronavirus patients to ascertain how tryptophan works in mitigating or preventing the aforementioned dysfunctions. In addition, clinical studies of SSRIs should also be performed to find out their efficacy in mild cases and as prophylactic measures and such studies should be redesigned with a fresh perspective to ascertain their role in preventing or curing anosmia, ageusia, and chemesthetic dysfunctions apart from lessening the severity of the disease. The study should also take into account the fact that there are at least fifteen 5-HT receptor types [34], [35], [36], and if one particular SSRI does not show satisfactory results, others should be tried, given an interesting report that in depressed patients residing at high altitudes, SSRIs like fluoxetine, paroxetine and escitalopram show no efficacy but sertraline works [121]. Also, 5-HT1A partial agonist buspirone alone or together with an SSRI may also be studied in this context [122]. As a preventive measure, combinations of SSRI and tryptophan should preferably be avoided to guard against serotonin syndrome and the possibility of rare eosinophilia-myalgia syndrome from the intake of tryptophan in susceptible patients should also be kept in mind [123].
Conclusions
5-HT deficiency (primarily, central 5-HT) arising from tryptophan depletion in coronavirus patients can explain reversible anosmia and ageusia (in mild and moderate cases), a wide variation of incidence rate of anosmia and ageusia amongst the patients, female predominance of anosmia and ageusia, chemesthetic dysfunction as well as worsening of silent hypoxemia and near-absence of protective hypoxic pulmonary vasoconstriction leading to increased severity of the disease. In addition, melatonin, which can lessen the SARS-CoV-2 induced cytokine storm, is also reduced owing to the deficit of 5-HT. Also, NAD+ deficiency may develop because of tryptophan depletion resulting in exacerbation of the disease. Accordingly, clinical trials to evaluate the prophylactic and therapeutic effectiveness of tryptophan supplementation should be conducted to assess how they perform in getting rid of or improving the above dysfunctions and mitigating the severity of COVID-19 so as to save the patients from the serious consequences of the pandemic.
Declaration of Competing Interest
The author declares that he has no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
Citations & impact
Impact metrics
Citations of article over time
Alternative metrics

Discover the attention surrounding your research
https://www.altmetric.com/details/107821675
Smart citations by scite.ai
Explore citation contexts and check if this article has been
supported or disputed.
https://scite.ai/reports/10.1016/j.mehy.2021.110627
Article citations
SARS-CoV-2 propagation to the TPH2-positive neurons in the ventral tegmental area induces cell death via GSK3β-dependent accumulation of phosphorylated tau.
PLoS One, 19(10):e0312834, 30 Oct 2024
Cited by: 0 articles | PMID: 39475992 | PMCID: PMC11524480
Precision nutrition to reset virus-induced human metabolic reprogramming and dysregulation (HMRD) in long-COVID.
NPJ Sci Food, 8(1):19, 30 Mar 2024
Cited by: 2 articles | PMID: 38555403 | PMCID: PMC10981760
Review Free full text in Europe PMC
Treatment of 95 post-Covid patients with SSRIs.
Sci Rep, 13(1):18599, 02 Nov 2023
Cited by: 6 articles | PMID: 37919310 | PMCID: PMC10622561
Selective serotonin reuptake inhibitors: New hope in the fight against COVID-19.
Front Pharmacol, 13:1036093, 30 Nov 2022
Cited by: 1 article | PMID: 36532776 | PMCID: PMC9748354
Review Free full text in Europe PMC
Psychological and Biochemical Effects of an Online Pilates Intervention in Pregnant Women during COVID-19: A Randomized Pilot Study.
Int J Environ Res Public Health, 19(17):10931, 01 Sep 2022
Cited by: 8 articles | PMID: 36078648 | PMCID: PMC9517892
Go to all (15) article citations
Similar Articles
To arrive at the top five similar articles we use a word-weighted algorithm to compare words from the Title and Abstract of each citation.
In Reference to Anosmia and Ageusia: Common Findings in COVID-19 Patients.
Laryngoscope, 130(9):E504-E505, 30 Jun 2020
Cited by: 2 articles | PMID: 32603479 | PMCID: PMC7361812
Negative correlation between ACE2 gene expression levels and loss of taste in a cohort of COVID-19 hospitalized patients: New clues to long-term cognitive disorders.
Front Cell Infect Microbiol, 12:905757, 29 Sep 2022
Cited by: 7 articles | PMID: 36250059 | PMCID: PMC9556632
Olfactory and gustatory dysfunctions as a clinical presentation of mild-to-moderate forms of the coronavirus disease (COVID-19): a multicenter European study.
Eur Arch Otorhinolaryngol, 277(8):2251-2261, 06 Apr 2020
Cited by: 1449 articles | PMID: 32253535 | PMCID: PMC7134551
Potential pathogenesis of ageusia and anosmia in COVID-19 patients.
Int Forum Allergy Rhinol, 10(9):1103-1104, 15 Jun 2020
Cited by: 147 articles | PMID: 32342636 | PMCID: PMC7267531
Review Free full text in Europe PMC



