Abstract
Free full text

L-Fucose-terminated glycoconjugates are recognized by pinocytosis receptors on macrophages.
Abstract
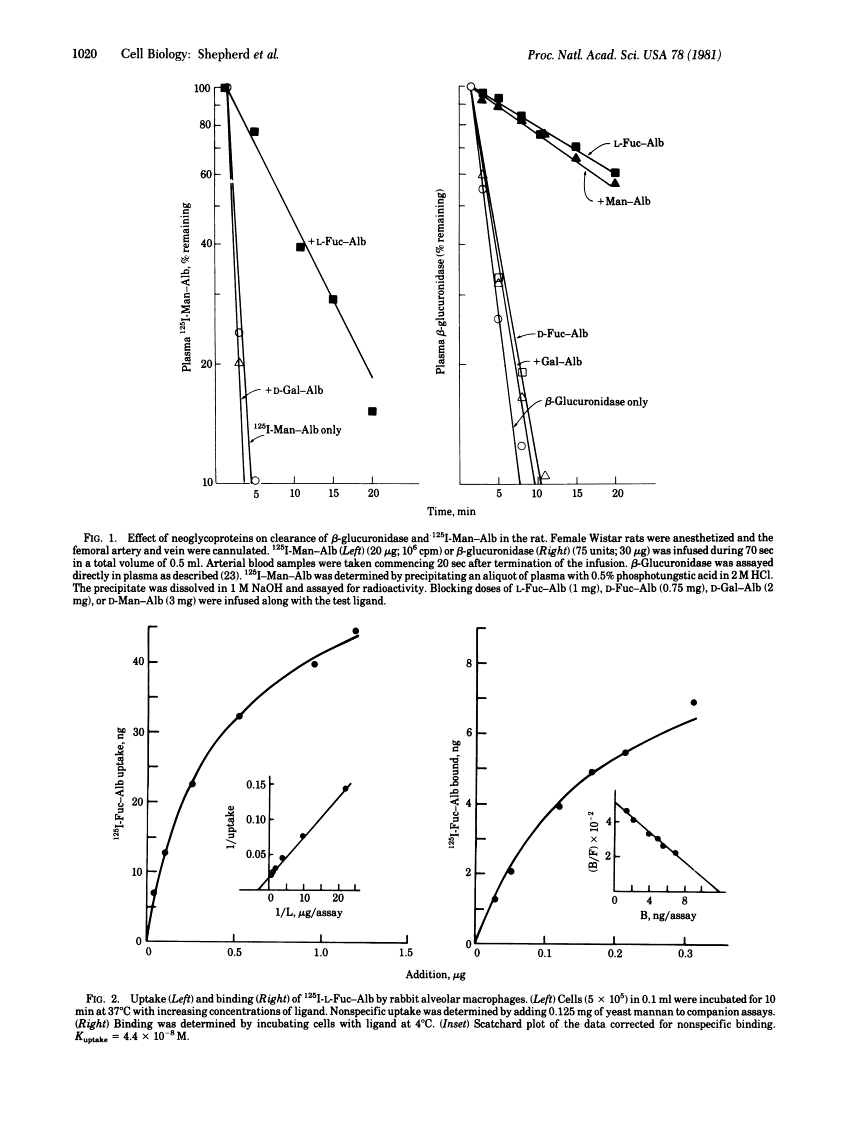
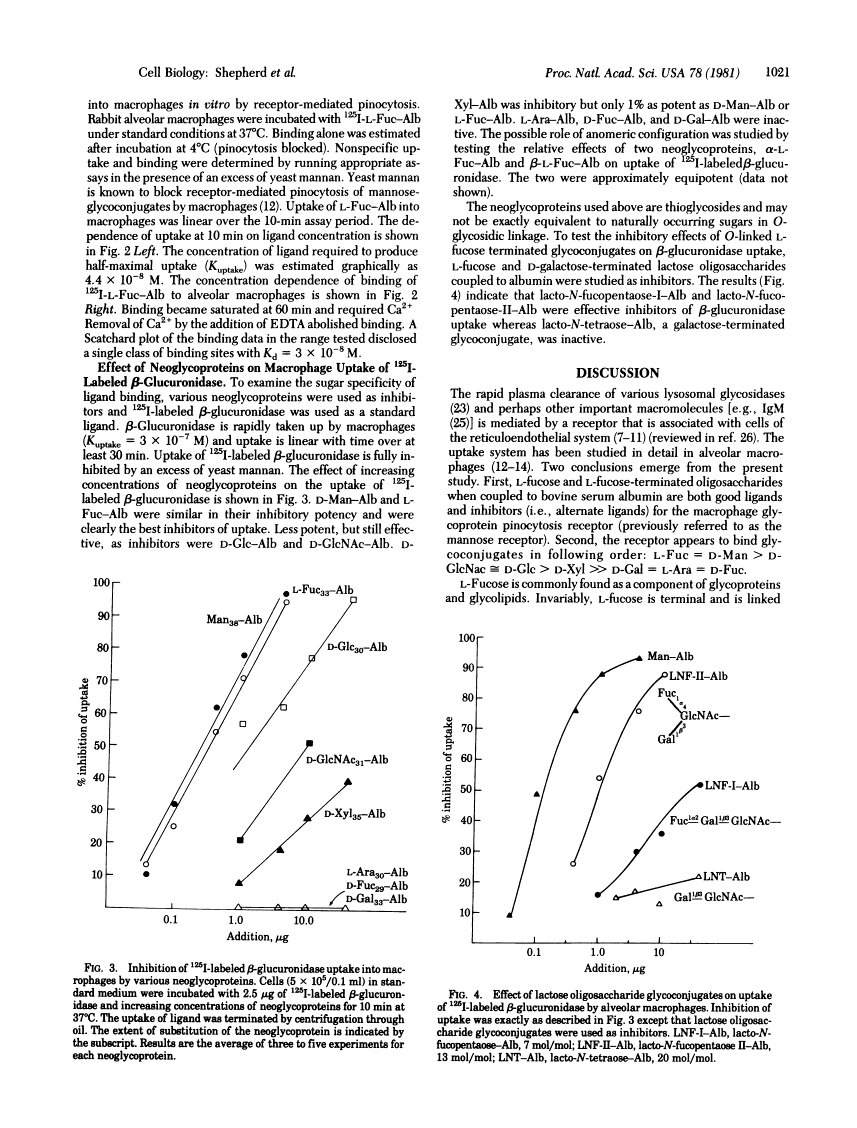
125I-Labeled L-fucose-albumin complex and rat preputial beta-glucuronidase are rapidly cleared from plasma after intravenous infusion. L-Fucose-albumin retards the plasma clearance of beta-glucuronidase whereas D-fucose-albumin is inactive. In vitro, 125I-labeled L-fucose-albumin is taken up into rat or rabbit alveolar macrophages by receptor-mediated pinocytosis. Uptake (37 degrees C) is time-dependent, is saturable with increasing ligand concentration (Kuptake = 4.4 X 10(-8) M), and requires Ca2+. 125I-labeled D-fucose-albumin is poorly taken up. Binding (4 degrees C) is saturable and Ca2+ dependent. Binding and uptake are fully inhibited by yeast mannan. A series of neoglycoproteins, including L-fucose-albumin, were tested as inhibitors of uptake of 125I-labeled beta-glucuronidase into macrophages. The following order of potency was observed: L-Fuc = D-Man greater than GlcNAc approximately D-Glc greater than D-Xyl much greater than than D-Gal = L-Ara = D-Fuc. L-Fucose-terminated oligosaccharides coupled to bovine serum albumin also block 125I-labeled beta-glucuronidase uptake into macrophages.
Full text
Full text is available as a scanned copy of the original print version. Get a printable copy (PDF file) of the complete article (704K), or click on a page image below to browse page by page. Links to PubMed are also available for Selected References.
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Stahl P, Six H, Rodman JS, Schlesinger P, Tulsiani DR, Touster O. Evidence for specific recognition sites mediating clearance of lysosomal enzymes in vivo. Proc Natl Acad Sci U S A. 1976 Nov;73(11):4045–4049. [Europe PMC free article] [Abstract] [Google Scholar]
- Stockert RJ, Morell AG, Scheinberg IH. The existence of a second route for the transfer of certain glycoproteins from the circulation into the liver. Biochem Biophys Res Commun. 1976 Feb 9;68(3):988–993. [Abstract] [Google Scholar]
- Achord DT, Brot FE, Sly WS. Inhibition of the rat clearance system for agalacto-orosomucoid by yeast mannans and by mannose. Biochem Biophys Res Commun. 1977 Jul 11;77(1):409–415. [Abstract] [Google Scholar]
- Baynes JW, Wold F. Effect of glycosylation on the in vivo circulating half-life of ribonuclease. J Biol Chem. 1976 Oct 10;251(19):6016–6024. [Abstract] [Google Scholar]
- Winkelhake JL, Nicolson GL. Aglycosylantibody. Effects of exoglycosidase treatments on autochthonous antibody survival time in the circulation. J Biol Chem. 1976 Feb 25;251(4):1074–1080. [Abstract] [Google Scholar]
- Schlesinger PH, Rodman JS, Doebber TW, Stahl PD, Lee YC, Stowell CP, Kuhlenschmidt TB. The role of extra-hepatic tissues in the receptor-mediated plasma clearance of glycoproteins terminated by mannose or N-acetylglucosamine. Biochem J. 1980 Nov 15;192(2):597–606. [Europe PMC free article] [Abstract] [Google Scholar]
- Schlesinger PH, Doebber TW, Mandell BF, White R, DeSchryver C, Rodman JS, Miller MJ, Stahl P. Plasma clearance of glycoproteins with terminal mannose and N-acetylglucosamine by liver non-parenchymal cells. Studies with beta-glucuronidase, N-acetyl-beta-D-glucosaminidase, ribonuclease B and agalacto-orosomucoid. Biochem J. 1978 Oct 15;176(1):103–109. [Europe PMC free article] [Abstract] [Google Scholar]
- Achord DT, Brot FE, Bell CE, Sly WS. Human beta-glucuronidase: in vivo clearance and in vitro uptake by a glycoprotein recognition system on reticuloendothelial cells. Cell. 1978 Sep;15(1):269–278. [Abstract] [Google Scholar]
- Brown TL, Henderson LA, Thorpe SR, Baynes JW. The effect of alpha-mannose-terminal oligosaccharides on the survival of glycoproteins in the circulation. Rapid uptake and catabolism of bovine pancreatic ribonuclease B by nonparenchymal cells of rat liver. Arch Biochem Biophys. 1978 Jun;188(2):418–428. [Abstract] [Google Scholar]
- Steer CJ, Clarenburg R. Unique distribution of glycoprotein receptors on parenchymal and sinusoidal cells of rat liver. J Biol Chem. 1979 Jun 10;254(11):4457–4461. [Abstract] [Google Scholar]
- Hubbard AL, Wilson G, Ashwell G, Stukenbrok H. An electron microscope autoradiographic study of the carbohydrate recognition systems in rat liver. I. Distribution of 125I-ligands among the liver cell types. J Cell Biol. 1979 Oct;83(1):47–64. [Europe PMC free article] [Abstract] [Google Scholar]
- Stahl PD, Rodman JS, Miller MJ, Schlesinger PH. Evidence for receptor-mediated binding of glycoproteins, glycoconjugates, and lysosomal glycosidases by alveolar macrophages. Proc Natl Acad Sci U S A. 1978 Mar;75(3):1399–1403. [Europe PMC free article] [Abstract] [Google Scholar]
- Stahl P, Schlesinger PH, Sigardson E, Rodman JS, Lee YC. Receptor-mediated pinocytosis of mannose glycoconjugates by macrophages: characterization and evidence for receptor recycling. Cell. 1980 Jan;19(1):207–215. [Abstract] [Google Scholar]
- Kaplan J, Nielsen M. Pinocytic activity of rabbit alveolar macrophages in vitro. J Reticuloendothel Soc. 1978 Dec;24(6):673–685. [Abstract] [Google Scholar]
- Kawasaki T, Etoh R, Yamashina I. Isolation and characterization of a mannan-binding protein from rabbit liver. Biochem Biophys Res Commun. 1978 Apr 14;81(3):1018–1024. [Abstract] [Google Scholar]
- Townsend R, Stahl P. Isolation and characterization of a mannose/N-acetylglucosamine/fucose-binding protein from rat liver. Biochem J. 1981 Jan 15;194(1):209–214. [Europe PMC free article] [Abstract] [Google Scholar]
- Lee YC, Stowell CP, Krantz MJ. 2-Imino-2-methoxyethyl 1-thioglycosides: new reagents for attaching sugars to proteins. Biochemistry. 1976 Sep 7;15(18):3956–3963. [Abstract] [Google Scholar]
- Stowell CP, Lee YC. Preparation of some new neoglycoproteins by amidination of bovine serum albumin using 2-imino-2-methoxyethyl 1-thioglycosides. Biochemistry. 1980 Oct 14;19(21):4899–4904. [Abstract] [Google Scholar]
- Zopf DA, Tsai CM, Ginsburg V. Antibodies against oligosaccharides coupled to proteins: characterization of carbohydrate specificity by radioimmune assay. Arch Biochem Biophys. 1978 Jan 15;185(1):61–71. [Abstract] [Google Scholar]
- Himeno M, Ohara H, Arakawa Y. Beta-glucuronidase of rat preputial gland. Crystallization, properties, carbohydrate composition, and subunits. J Biochem. 1975 Feb;77(2):427–438. [Abstract] [Google Scholar]
- Stahl PD, Touster O. Beta-glucuronidase of rat liver lysosomes. Purification, properties, subunits. J Biol Chem. 1971 Sep 10;246(17):5398–5406. [Abstract] [Google Scholar]
- GREENWOOD FC, HUNTER WM, GLOVER JS. THE PREPARATION OF I-131-LABELLED HUMAN GROWTH HORMONE OF HIGH SPECIFIC RADIOACTIVITY. Biochem J. 1963 Oct;89:114–123. [Europe PMC free article] [Abstract] [Google Scholar]
- Stahl P, Rodman JS, Schlesinger P. Clearance of lysosomal hydrolases following intravenous infusion. Kinetic and competition experiments with beta-glucuronidase and N-acetyl-beta-D-glucosaminidase. Arch Biochem Biophys. 1976 Dec;177(2):594–605. [Abstract] [Google Scholar]
- Leake ES, Myrvik QN. Changes in morphology and in lysozyme content of free alveolar cells after the intravenous injection of killed BCG in oil. J Reticuloendothel Soc. 1968 Feb;5(1):33–53. [Abstract] [Google Scholar]
- Day JF, Thornburg RW, Thorpe SR, Baynes JW. Carbohydrate-mediated clearance of antibody . antigen complexes from the circulation. The role of high mannose oligosaccharides in the hepatic uptake of IgM . antigen complexes. J Biol Chem. 1980 Mar 25;255(6):2360–2365. [Abstract] [Google Scholar]
- Kornfeld R, Kornfeld S. Comparative aspects of glycoprotein structure. Annu Rev Biochem. 1976;45:217–237. [Abstract] [Google Scholar]
- Watkins WM. Blood-group substances. Science. 1966 Apr 8;152(3719):172–181. [Abstract] [Google Scholar]
- Remold HG. Requirement for alpha-L-fucose on the macrophage membrane receptor for MIF. J Exp Med. 1973 Nov 1;138(5):1065–1076. [Europe PMC free article] [Abstract] [Google Scholar]
- Prieels JP, Pizzo SV, Glasgow LR, Paulson JC, Hill RL. Hepatic receptor that specifically binds oligosaccharides containing fucosyl alpha1 leads to 3 N-acetylglucosamine linkages. Proc Natl Acad Sci U S A. 1978 May;75(5):2215–2219. [Europe PMC free article] [Abstract] [Google Scholar]
- Grant DA, Magee AI, Meeks D, Regan C, Bainbridge DR, Hermon-Taylor J. Identification of a defence mechanism in vivo against the leakage of enterokinase into the blood. Biochem J. 1979 Dec 15;184(3):619–626. [Europe PMC free article] [Abstract] [Google Scholar]
- Krantz MJ, Holtzman NA, Stowell CP, Lee YC. Attachment of thioglycosides to proteins: enhancement of liver membrane binding. Biochemistry. 1976 Sep 7;15(18):3963–3968. [Abstract] [Google Scholar]
- Stowell CP, Lee YC. The binding of d-glucosyl-neoglycoproteins to the hepatic asialoglycoprotein receptor. J Biol Chem. 1978 Sep 10;253(17):6107–6110. [Abstract] [Google Scholar]
- Stowell CP, Lee RT, Lee YC. Studies on the specificity of rabbit hepatic carbohydrate-binding protein using neoglycoproteins. Biochemistry. 1980 Oct 14;19(21):4904–4908. [Abstract] [Google Scholar]
Associated Data
Articles from Proceedings of the National Academy of Sciences of the United States of America are provided here courtesy of National Academy of Sciences
Full text links
Read article at publisher's site: https://doi.org/10.1073/pnas.78.2.1019
Read article for free, from open access legal sources, via Unpaywall:
https://www.pnas.org/content/pnas/78/2/1019.full.pdf
Citations & impact
Impact metrics
Citations of article over time
Smart citations by scite.ai
Explore citation contexts and check if this article has been
supported or disputed.
https://scite.ai/reports/10.1073/pnas.78.2.1019
Article citations
Mannose Ligands for Mannose Receptor Targeting.
Int J Mol Sci, 25(3):1370, 23 Jan 2024
Cited by: 5 articles | PMID: 38338648 | PMCID: PMC10855088
Review Free full text in Europe PMC
N-glycosylation of mannose receptor (CD206) regulates glycan binding by C-type lectin domains.
J Biol Chem, 298(12):102591, 13 Oct 2022
Cited by: 4 articles | PMID: 36244450 | PMCID: PMC9672410
The mannose receptor ligands and the macrophage glycome.
Curr Opin Struct Biol, 75:102394, 23 May 2022
Cited by: 19 articles | PMID: 35617912 | PMCID: PMC10243190
Review Free full text in Europe PMC
Macrophage targeted theranostic strategy for accurate detection and rapid stabilization of the inflamed high-risk plaque.
Theranostics, 11(18):8874-8893, 18 Aug 2021
Cited by: 9 articles | PMID: 34522216 | PMCID: PMC8419038
Tumor cells express pauci- and oligomannosidic N-glycans in glycoproteins recognized by the mannose receptor (CD206).
Cell Mol Life Sci, 78(14):5569-5585, 05 Jun 2021
Cited by: 9 articles | PMID: 34089345 | PMCID: PMC11072813
Go to all (70) article citations
Similar Articles
To arrive at the top five similar articles we use a word-weighted algorithm to compare words from the Title and Abstract of each citation.
Expression of a mannosyl-fucosyl receptor for endocytosis on cultured primary macrophages and their hybrids.
J Cell Biol, 93(1):49-56, 01 Apr 1982
Cited by: 115 articles | PMID: 6279673 | PMCID: PMC2112098
The binding of fucose-containing glycoproteins by hepatic lectins. Re-examination of the clearance from blood and the binding to membrane receptors and pure lectins.
J Biol Chem, 261(16):7412-7418, 01 Jun 1986
Cited by: 26 articles | PMID: 3011783
Metabolism of glycosylated human salivary amylase: in vivo plasma clearance by rat hepatic endothelial cells and in vitro receptor mediated pinocytosis by rat macrophages.
J Leukoc Biol, 36(3):307-320, 01 Sep 1984
Cited by: 12 articles | PMID: 6207253
Characterization of the mannose/fucose receptor on human mononuclear phagocytes.
J Reticuloendothel Soc, 32(6):423-431, 01 Dec 1982
Cited by: 83 articles | PMID: 6298410
Funding
Funders who supported this work.
NCI NIH HHS (1)
Grant ID: CA 12858
NIGMS NIH HHS (1)
Grant ID: GM 21096