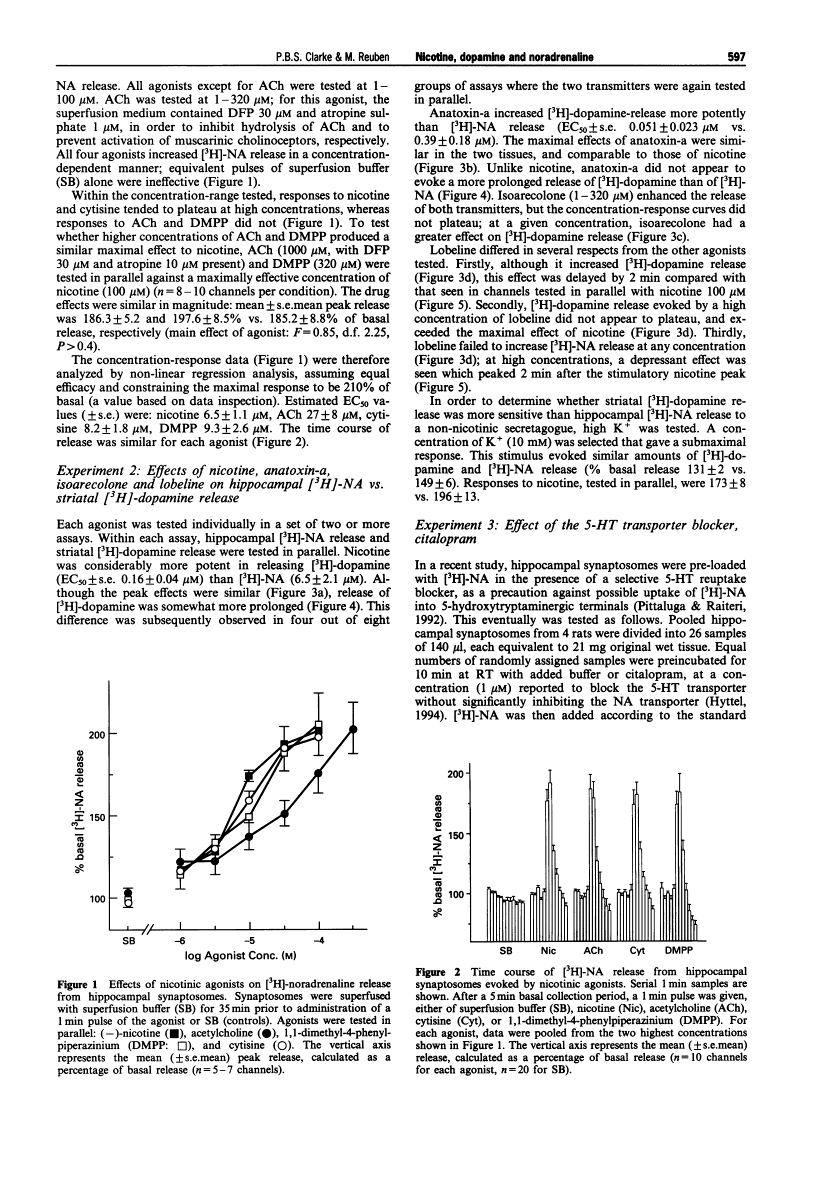
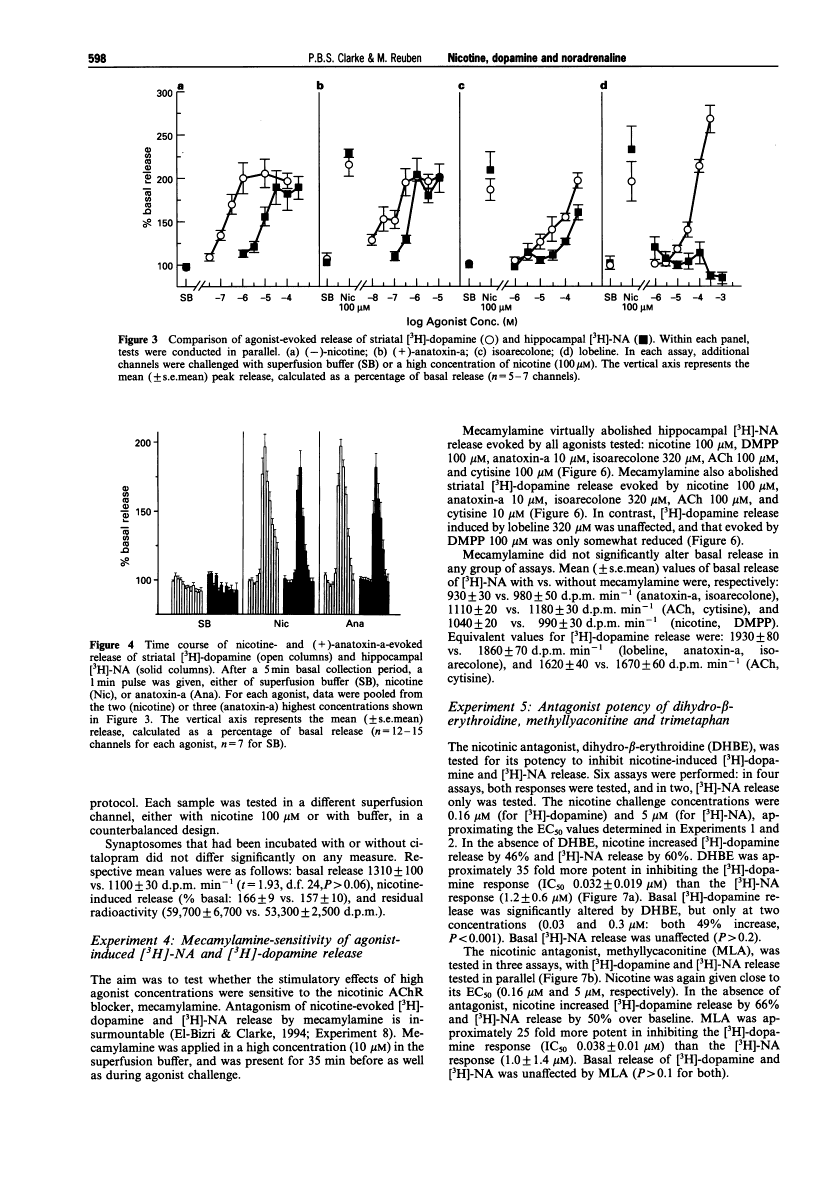
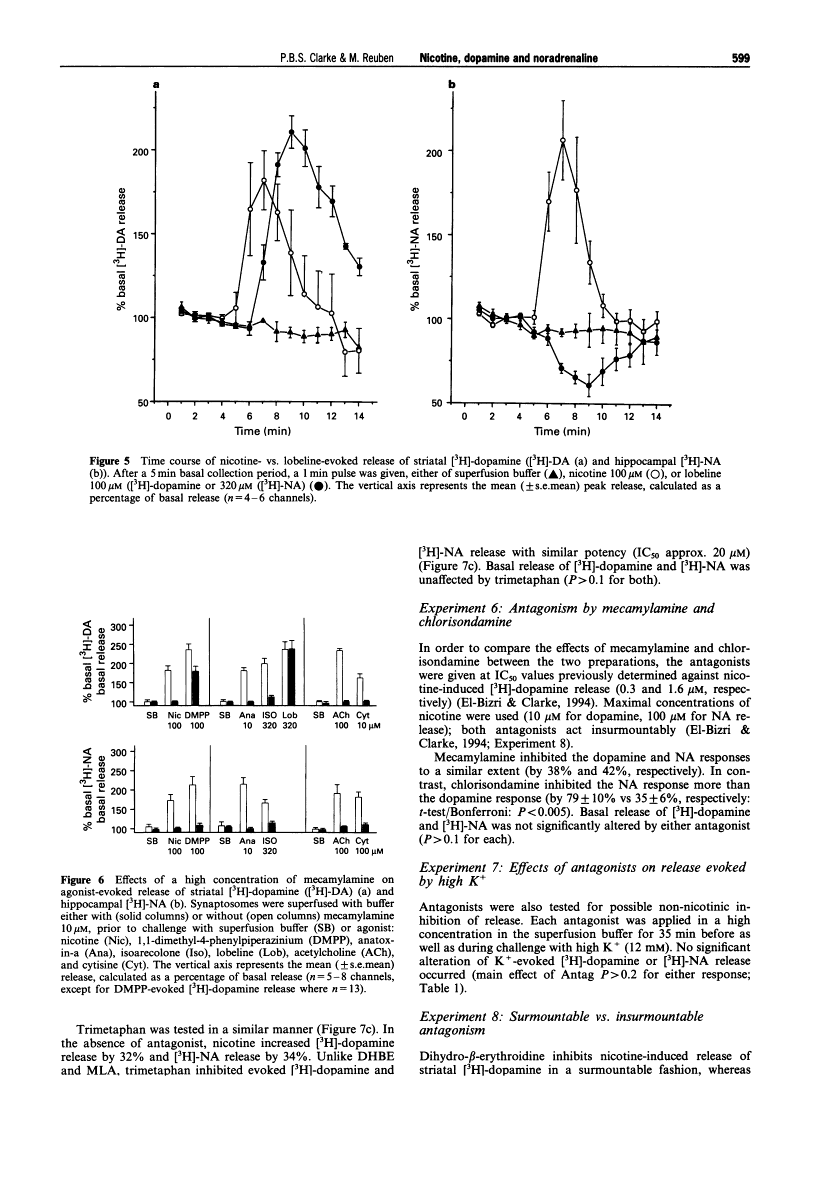
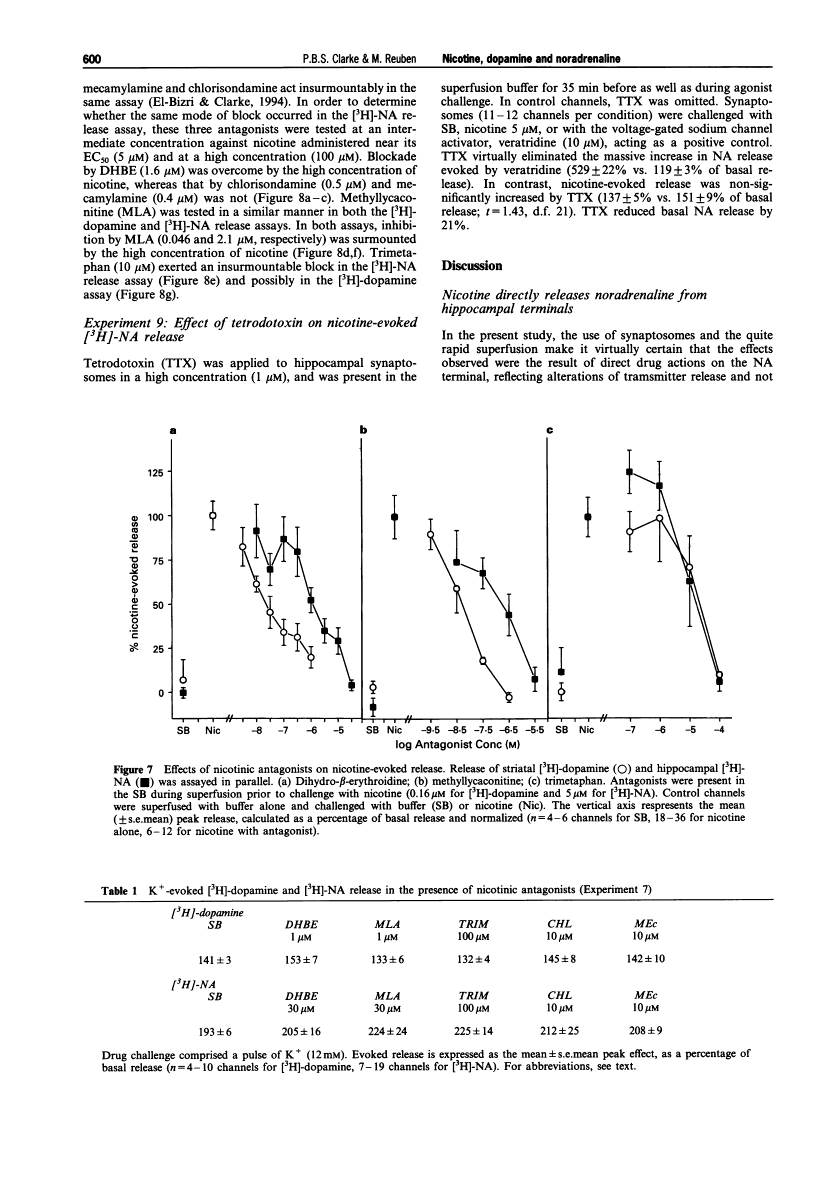
Abstract
Free full text

Release of [3H]-noradrenaline from rat hippocampal synaptosomes by nicotine: mediation by different nicotinic receptor subtypes from striatal [3H]-dopamine release.
Abstract
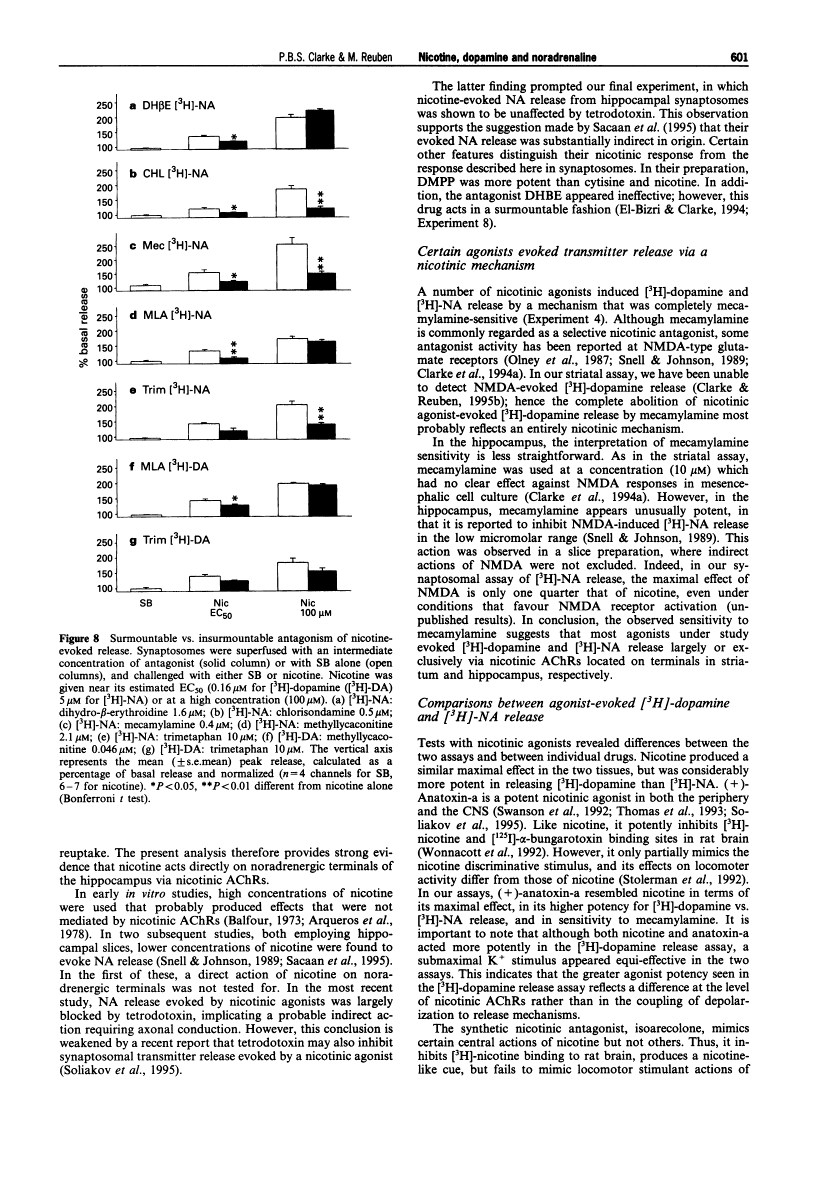
1. The aim of the present experiment was to characterize nicotine-evoked [3H]-noradrenaline ([3H]-NA) release from rat superfused hippocampal synaptosomes, using striatal [3H]-dopamine release for comparison. 2. (-)-Nicotine, cytisine, DMPP and acetylcholine (ACh) (with esterase inhibitor and muscarinic receptor blocker) increased NA release in a concentration-dependent manner (EC50 6.5 microM, 8.2 microM, 9.3 microM, and 27 microM, respectively) with similar efficacy. 3. Nicotine released striatal dopamine more potently than hippocampal NA (EC50 0.16 microM vs. 6.5 microM). (+)-Anatoxin-a also increased dopamine more potently than NA (EC50 0.05 microM vs. 0.39 microM), and maximal effects were similar to those of nicotine. Isoarecolone (10-320 microM) released dopamine more effectively than NA but a maximal effect was not reached. (-)-Lobeline (10-320 microM) evoked dopamine release, but the effect was large and delayed with respect to nicotine; NA release was not increased but rather depressed at high concentrations of lobeline. High K+ (10 mM) released and NA to similar extents. 4. Addition of the 5-hydroxytryptamine (5-HT) reuptake blocker, citalopram (1 microM) to hippocampal synaptosomes affected neither basal NA release nor nicotine-evoked release. 5. The nicotinic antagonist, mecamylamine (10 microM), virtually abolished NA and dopamine release evoked by high concentrations of nicotine, ACh, cytisine, isoarecolone, and anatoxin-a. Although NA release evoked by DMPP (100 microM) was entirely mecamylamine-sensitive, DMPP-evoked dopamine release was only partially blocked. Dopamine release evoked by lobeline (320 microM) was completely mecamylamine-insensitive. 6. The nicotinic antagonists dihydro-beta-erythroidine and methyllycaconitine inhibited nicotine-evoked dopamine release approximately 30 fold more potently than NA release. In contrast, the antagonist chlorisondamine, displayed a reverse sensitivity, whereas trimetaphan and mecamylamine did not preferentially block either response. None of these antagonists, given at a high concentration, significantly altered release evoked by high K+. 7. Blockade of nicotine-evoked transmitter release by methyllycaconitine and dihydro-beta-erythroidine was surmounted by a high concentration of nicotine (100 microM), but blockade by mecamylamine, chlorisondamine, and trimetaphan was insurmountable. 8. Nicotine-evoked NA release was unaffected by tetrodotoxin, whereas veratridine-evoked NA release was virtually abolished. 9. We conclude that presynaptic nicotinic receptors associated with striatal dopamine and hippocampal NA terminals differ pharmacologically. In situ hybridization studies suggest that nigrostriatal dopaminergic neurones express mainly alpha 4, alpha 5, and beta 2 nicotinic cholinoceptor subunits, whereas hippocampal-projecting noradrenaline (NA) neurones express alpha 3, beta 2 and beta 4 subunits. Pharmacological comparisons of recombinant receptors suggest that release of hippocampal NA may be modulated by receptors containing alpha 3 and beta 4 subunits.
Full text
Full text is available as a scanned copy of the original print version. Get a printable copy (PDF file) of the complete article (2.5M), or click on a page image below to browse page by page. Links to PubMed are also available for Selected References.
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Aizenman E, Tang LH, Reynolds IJ. Effects of nicotinic agonists on the NMDA receptor. Brain Res. 1991 Jun 14;551(1-2):355–357. [Abstract] [Google Scholar]
- Alkondon M, Albuquerque EX. Diversity of nicotinic acetylcholine receptors in rat hippocampal neurons. I. Pharmacological and functional evidence for distinct structural subtypes. J Pharmacol Exp Ther. 1993 Jun;265(3):1455–1473. [Abstract] [Google Scholar]
- Alkondon M, Pereira EF, Wonnacott S, Albuquerque EX. Blockade of nicotinic currents in hippocampal neurons defines methyllycaconitine as a potent and specific receptor antagonist. Mol Pharmacol. 1992 Apr;41(4):802–808. [Abstract] [Google Scholar]
- Arqueros L, Naquira D, Zunino E. Nicotine-induced release of catecholamines from rat hippocampus and striatum. Biochem Pharmacol. 1978;27(23):2667–2674. [Abstract] [Google Scholar]
- Balfour DJ. Effects of nicotine on the uptake and retention of 14C-noradrenaline and 14C-5-hydroxytryptamine by rat brain homogenates. Eur J Pharmacol. 1973 Jul;23(1):19–26. [Abstract] [Google Scholar]
- Bhat RV, Turner SL, Selvaag SR, Marks MJ, Collins AC. Regulation of brain nicotinic receptors by chronic agonist infusion. J Neurochem. 1991 Jun;56(6):1932–1939. [Abstract] [Google Scholar]
- Buccafusco JJ, Jackson WJ, Gattu M, Terry AV., Jr Isoarecolone-induced enhancement of delayed matching to sample performance in monkeys: role of nicotinic receptors. Neuroreport. 1995 May 30;6(8):1223–1227. [Abstract] [Google Scholar]
- Cachelin AB, Jaggi R. Beta subunits determine the time course of desensitization in rat alpha 3 neuronal nicotinic acetylcholine receptors. Pflugers Arch. 1991 Dec;419(6):579–582. [Abstract] [Google Scholar]
- Cachelin AB, Rust G. Unusual pharmacology of (+)-tubocurarine with rat neuronal nicotinic acetylcholine receptors containing beta 4 subunits. Mol Pharmacol. 1994 Dec;46(6):1168–1174. [Abstract] [Google Scholar]
- Cachelin AB, Rust G. Beta-subunits co-determine the sensitivity of rat neuronal nicotinic receptors to antagonists. Pflugers Arch. 1995 Jan;429(3):449–451. [Abstract] [Google Scholar]
- Caulfield MP, Higgins GA. Mediation of nicotine-induced convulsions by central nicotinic receptors of the 'C6' type. Neuropharmacology. 1983 Mar;22(3):347–351. [Abstract] [Google Scholar]
- Clarke PB, Chaudieu I, el-Bizri H, Boksa P, Quik M, Esplin BA, Capek R. The pharmacology of the nicotinic antagonist, chlorisondamine, investigated in rat brain and autonomic ganglion. Br J Pharmacol. 1994 Feb;111(2):397–405. [Europe PMC free article] [Abstract] [Google Scholar]
- Clarke PB, Pert A. Autoradiographic evidence for nicotine receptors on nigrostriatal and mesolimbic dopaminergic neurons. Brain Res. 1985 Dec 2;348(2):355–358. [Abstract] [Google Scholar]
- Clarke PB, Reuben M. Inhibition by dizocilpine (MK-801) of striatal dopamine release induced by MPTP and MPP+: possible action at the dopamine transporter. Br J Pharmacol. 1995 Jan;114(2):315–322. [Europe PMC free article] [Abstract] [Google Scholar]
- Clarke PB, Reuben M, el-Bizri H. Blockade of nicotinic responses by physostigmine, tacrine and other cholinesterase inhibitors in rat striatum. Br J Pharmacol. 1994 Mar;111(3):695–702. [Europe PMC free article] [Abstract] [Google Scholar]
- Collins AC, Evans CB, Miner LL, Marks MJ. Mecamylamine blockade of nicotine responses: evidence for two brain nicotinic receptors. Pharmacol Biochem Behav. 1986 Jun;24(6):1767–1773. [Abstract] [Google Scholar]
- Connolly J, Boulter J, Heinemann SF. Alpha 4-2 beta 2 and other nicotinic acetylcholine receptor subtypes as targets of psychoactive and addictive drugs. Br J Pharmacol. 1992 Mar;105(3):657–666. [Europe PMC free article] [Abstract] [Google Scholar]
- Conroy WG, Berg DK. Neurons can maintain multiple classes of nicotinic acetylcholine receptors distinguished by different subunit compositions. J Biol Chem. 1995 Mar 3;270(9):4424–4431. [Abstract] [Google Scholar]
- Court JA, Perry EK, Spurden D, Lloyd S, Gillespie JI, Whiting P, Barlow R. Comparison of the binding of nicotinic agonists to receptors from human and rat cerebral cortex and from chick brain (alpha 4 beta 2) transfected into mouse fibroblasts with ion channel activity. Brain Res. 1994 Dec 19;667(1):118–122. [Abstract] [Google Scholar]
- Covernton PJ, Kojima H, Sivilotti LG, Gibb AJ, Colquhoun D. Comparison of neuronal nicotinic receptors in rat sympathetic neurones with subunit pairs expressed in Xenopus oocytes. J Physiol. 1994 Nov 15;481(Pt 1):27–34. [Abstract] [Google Scholar]
- Dar MS, Li C, Bowman ER. Central behavioral interactions between ethanol, (-)-nicotine, and (-)-cotinine in mice. Brain Res Bull. 1993;32(1):23–28. [Abstract] [Google Scholar]
- Dineley-Miller K, Patrick J. Gene transcripts for the nicotinic acetylcholine receptor subunit, beta4, are distributed in multiple areas of the rat central nervous system. Brain Res Mol Brain Res. 1992 Dec;16(3-4):339–344. [Abstract] [Google Scholar]
- el-Bizri H, Clarke PB. Blockade of nicotinic receptor-mediated release of dopamine from striatal synaptosomes by chlorisondamine and other nicotinic antagonists administered in vitro. Br J Pharmacol. 1994 Feb;111(2):406–413. [Europe PMC free article] [Abstract] [Google Scholar]
- Flores CM, Rogers SW, Pabreza LA, Wolfe BB, Kellar KJ. A subtype of nicotinic cholinergic receptor in rat brain is composed of alpha 4 and beta 2 subunits and is up-regulated by chronic nicotine treatment. Mol Pharmacol. 1992 Jan;41(1):31–37. [Abstract] [Google Scholar]
- Fudala PJ, Iwamoto ET. Further studies on nicotine-induced conditioned place preference in the rat. Pharmacol Biochem Behav. 1986 Nov;25(5):1041–1049. [Abstract] [Google Scholar]
- Grady SR, Marks MJ, Collins AC. Desensitization of nicotine-stimulated [3H]dopamine release from mouse striatal synaptosomes. J Neurochem. 1994 Apr;62(4):1390–1398. [Abstract] [Google Scholar]
- Grady S, Marks MJ, Wonnacott S, Collins AC. Characterization of nicotinic receptor-mediated [3H]dopamine release from synaptosomes prepared from mouse striatum. J Neurochem. 1992 Sep;59(3):848–856. [Abstract] [Google Scholar]
- Holbach HJ, Lindmar R, Löffelholz K. DMPP and the adrenergic nerve terminal: mechanisms of noradrenaline release from vesicular and extravesicular compartments. Naunyn Schmiedebergs Arch Pharmacol. 1977 Nov;300(2):131–138. [Abstract] [Google Scholar]
- Hyttel J. Pharmacological characterization of selective serotonin reuptake inhibitors (SSRIs). Int Clin Psychopharmacol. 1994 Mar;9 (Suppl 1):19–26. [Abstract] [Google Scholar]
- Izenwasser S, Jacocks HM, Rosenberger JG, Cox BM. Nicotine indirectly inhibits [3H]dopamine uptake at concentrations that do not directly promote [3H]dopamine release in rat striatum. J Neurochem. 1991 Feb;56(2):603–610. [Abstract] [Google Scholar]
- Khan IM, Taylor P, Yaksh TL. Cardiovascular and behavioral responses to nicotinic agents administered intrathecally. J Pharmacol Exp Ther. 1994 Jul;270(1):150–158. [Abstract] [Google Scholar]
- Loiacono R, Stephenson J, Stevenson J, Mitchelson F. Multiple binding sites for nicotine receptor antagonists in inhibiting [3H](-)-nicotine binding in rat cortex. Neuropharmacology. 1993 Sep;32(9):847–853. [Abstract] [Google Scholar]
- Luetje CW, Patrick J. Both alpha- and beta-subunits contribute to the agonist sensitivity of neuronal nicotinic acetylcholine receptors. J Neurosci. 1991 Mar;11(3):837–845. [Europe PMC free article] [Abstract] [Google Scholar]
- Luetje CW, Piattoni M, Patrick J. Mapping of ligand binding sites of neuronal nicotinic acetylcholine receptors using chimeric alpha subunits. Mol Pharmacol. 1993 Sep;44(3):657–666. [Abstract] [Google Scholar]
- Luetje CW, Wada K, Rogers S, Abramson SN, Tsuji K, Heinemann S, Patrick J. Neurotoxins distinguish between different neuronal nicotinic acetylcholine receptor subunit combinations. J Neurochem. 1990 Aug;55(2):632–640. [Abstract] [Google Scholar]
- Mandelzys A, Pié B, Deneris ES, Cooper E. The developmental increase in ACh current densities on rat sympathetic neurons correlates with changes in nicotinic ACh receptor alpha-subunit gene expression and occurs independent of innervation. J Neurosci. 1994 Apr;14(4):2357–2364. [Europe PMC free article] [Abstract] [Google Scholar]
- Marks MJ, Collins AC. Characterization of nicotine binding in mouse brain and comparison with the binding of alpha-bungarotoxin and quinuclidinyl benzilate. Mol Pharmacol. 1982 Nov;22(3):554–564. [Abstract] [Google Scholar]
- Marks MJ, Farnham DA, Grady SR, Collins AC. Nicotinic receptor function determined by stimulation of rubidium efflux from mouse brain synaptosomes. J Pharmacol Exp Ther. 1993 Feb;264(2):542–552. [Abstract] [Google Scholar]
- Mitchell SN. Role of the locus coeruleus in the noradrenergic response to a systemic administration of nicotine. Neuropharmacology. 1993 Oct;32(10):937–949. [Abstract] [Google Scholar]
- Mulle C, Vidal C, Benoit P, Changeux JP. Existence of different subtypes of nicotinic acetylcholine receptors in the rat habenulo-interpeduncular system. J Neurosci. 1991 Aug;11(8):2588–2597. [Europe PMC free article] [Abstract] [Google Scholar]
- Olney JW, Price MT, Labruyere J, Salles KS, Frierdich G, Mueller M, Silverman E. Anti-parkinsonian agents are phencyclidine agonists and N-methyl-aspartate antagonists. Eur J Pharmacol. 1987 Oct 13;142(2):319–320. [Abstract] [Google Scholar]
- Papke RL. The kinetic properties of neuronal nicotinic receptors: genetic basis of functional diversity. Prog Neurobiol. 1993 Oct;41(4):509–531. [Abstract] [Google Scholar]
- Papke RL, Duvoisin RM, Heinemann SF. The amino terminal half of the nicotinic beta-subunit extracellular domain regulates the kinetics of inhibition by neuronal bungarotoxin. Proc Biol Sci. 1993 May 22;252(1334):141–148. [Abstract] [Google Scholar]
- Papke RL, Heinemann SF. Partial agonist properties of cytisine on neuronal nicotinic receptors containing the beta 2 subunit. Mol Pharmacol. 1994 Jan;45(1):142–149. [Abstract] [Google Scholar]
- Pittaluga A, Raiteri M. N-methyl-D-aspartic acid (NMDA) and non-NMDA receptors regulating hippocampal norepinephrine release. I. Location on axon terminals and pharmacological characterization. J Pharmacol Exp Ther. 1992 Jan;260(1):232–237. [Abstract] [Google Scholar]
- Rapier C, Lunt GG, Wonnacott S. Stereoselective nicotine-induced release of dopamine from striatal synaptosomes: concentration dependence and repetitive stimulation. J Neurochem. 1988 Apr;50(4):1123–1130. [Abstract] [Google Scholar]
- Rapier C, Lunt GG, Wonnacott S. Nicotinic modulation of [3H]dopamine release from striatal synaptosomes: pharmacological characterisation. J Neurochem. 1990 Mar;54(3):937–945. [Abstract] [Google Scholar]
- Reavill C, Spivak CE, Stolerman IP, Waters JA. Isoarecolone can inhibit nicotine binding and produce nicotine-like discriminative stimulus effects in rats. Neuropharmacology. 1987 Jul;26(7A):789–792. [Abstract] [Google Scholar]
- Reavill C, Walther B, Stolerman IP, Testa B. Behavioural and pharmacokinetic studies on nicotine, cytisine and lobeline. Neuropharmacology. 1990 Jul;29(7):619–624. [Abstract] [Google Scholar]
- Reavill C, Waters JA, Stolerman IP, Garcha HS. Behavioural effects of the nicotinic agonists N-(3-pyridylmethyl)pyrrolidine and isoarecolone in rats. Psychopharmacology (Berl) 1990;102(4):521–528. [Abstract] [Google Scholar]
- Romano C. Nicotine action on rat colon. J Pharmacol Exp Ther. 1981 Jun;217(3):828–833. [Abstract] [Google Scholar]
- Romano C, Goldstein A, Jewell NP. Characterization of the receptor mediating the nicotine discriminative stimulus. Psychopharmacology (Berl) 1981;74(4):310–315. [Abstract] [Google Scholar]
- Rowell PP, Hillebrand JA. Characterization of nicotine-induced desensitization of evoked dopamine release from rat striatal synaptosomes. J Neurochem. 1994 Aug;63(2):561–569. [Abstract] [Google Scholar]
- Rowell PP, Wonnacott S. Evidence for functional activity of up-regulated nicotine binding sites in rat striatal synaptosomes. J Neurochem. 1990 Dec;55(6):2105–2110. [Abstract] [Google Scholar]
- Rust G, Burgunder JM, Lauterburg TE, Cachelin AB. Expression of neuronal nicotinic acetylcholine receptor subunit genes in the rat autonomic nervous system. Eur J Neurosci. 1994 Mar 1;6(3):478–485. [Abstract] [Google Scholar]
- Sacaan AI, Dunlop JL, Lloyd GK. Pharmacological characterization of neuronal acetylcholine gated ion channel receptor-mediated hippocampal norepinephrine and striatal dopamine release from rat brain slices. J Pharmacol Exp Ther. 1995 Jul;274(1):224–230. [Abstract] [Google Scholar]
- Sakurai Y, Takano Y, Kohjimoto Y, Honda K, Kamiya HO. Enhancement of [3H]dopamine release and its [3H]metabolites in rat striatum by nicotinic drugs. Brain Res. 1982 Jun 17;242(1):99–106. [Abstract] [Google Scholar]
- Schmidt J. Drug binding properties of an alpha-bungarotoxin-binding component from rat brain. Mol Pharmacol. 1977 Mar;13(2):283–290. [Abstract] [Google Scholar]
- Schulz DW, Zigmond RE. Neuronal bungarotoxin blocks the nicotinic stimulation of endogenous dopamine release from rat striatum. Neurosci Lett. 1989 Apr 10;98(3):310–316. [Abstract] [Google Scholar]
- Schwartz RD, Kellar KJ. [3H]acetylcholine binding sites in brain. Effect of disulfide bond modification. Mol Pharmacol. 1983 Nov;24(3):387–391. [Abstract] [Google Scholar]
- Schwartz RD, Lehmann J, Kellar KJ. Presynaptic nicotinic cholinergic receptors labeled by [3H]acetylcholine on catecholamine and serotonin axons in brain. J Neurochem. 1984 May;42(5):1495–1498. [Abstract] [Google Scholar]
- Séguéla P, Wadiche J, Dineley-Miller K, Dani JA, Patrick JW. Molecular cloning, functional properties, and distribution of rat brain alpha 7: a nicotinic cation channel highly permeable to calcium. J Neurosci. 1993 Feb;13(2):596–604. [Europe PMC free article] [Abstract] [Google Scholar]
- Snell LD, Johnson KM. Effects of nicotinic agonists and antagonists on N-methyl-D-aspartate-induced 3H-norepinephrine release and 3H-(1-[1-(2-thienyl)cyclohexyl]-piperidine) binding in rat hippocampus. Synapse. 1989;3(2):129–135. [Abstract] [Google Scholar]
- Stolerman IP, Albuquerque EX, Garcha HS. Behavioural effects of anatoxin, a potent nicotinic agonist, in rats. Neuropharmacology. 1992 Mar;31(3):311–314. [Abstract] [Google Scholar]
- Stolerman IP, Garcha HS, Mirza NR. Dissociations between the locomotor stimulant and depressant effects of nicotinic agonists in rats. Psychopharmacology (Berl) 1995 Feb;117(4):430–437. [Abstract] [Google Scholar]
- Swanson KL, Aronstam RS, Wonnacott S, Rapoport H, Albuquerque EX. Nicotinic pharmacology of anatoxin analogs. I. Side chain structure-activity relationships at peripheral agonist and noncompetitive antagonist sites. J Pharmacol Exp Ther. 1991 Oct;259(1):377–386. [Abstract] [Google Scholar]
- Thomas P, Stephens M, Wilkie G, Amar M, Lunt GG, Whiting P, Gallagher T, Pereira E, Alkondon M, Albuquerque EX, et al. (+)-Anatoxin-a is a potent agonist at neuronal nicotinic acetylcholine receptors. J Neurochem. 1993 Jun;60(6):2308–2311. [Abstract] [Google Scholar]
- Vernallis AB, Conroy WG, Berg DK. Neurons assemble acetylcholine receptors with as many as three kinds of subunits while maintaining subunit segregation among receptor subtypes. Neuron. 1993 Mar;10(3):451–464. [Abstract] [Google Scholar]
- Vidal C, Changeux JP. Nicotinic and muscarinic modulations of excitatory synaptic transmission in the rat prefrontal cortex in vitro. Neuroscience. 1993 Sep;56(1):23–32. [Abstract] [Google Scholar]
- Wada E, McKinnon D, Heinemann S, Patrick J, Swanson LW. The distribution of mRNA encoded by a new member of the neuronal nicotinic acetylcholine receptor gene family (alpha 5) in the rat central nervous system. Brain Res. 1990 Aug 27;526(1):45–53. [Abstract] [Google Scholar]
- Wada E, Wada K, Boulter J, Deneris E, Heinemann S, Patrick J, Swanson LW. Distribution of alpha 2, alpha 3, alpha 4, and beta 2 neuronal nicotinic receptor subunit mRNAs in the central nervous system: a hybridization histochemical study in the rat. J Comp Neurol. 1989 Jun 8;284(2):314–335. [Abstract] [Google Scholar]
- Ward JM, Cockcroft VB, Lunt GG, Smillie FS, Wonnacott S. Methyllycaconitine: a selective probe for neuronal alpha-bungarotoxin binding sites. FEBS Lett. 1990 Sep 17;270(1-2):45–48. [Abstract] [Google Scholar]
- Weaver WR, Wolf KM, Chiappinelli VA. Functional heterogeneity of nicotinic receptors in the avian lateral spiriform nucleus detected with trimethaphan. Mol Pharmacol. 1994 Nov;46(5):993–1001. [Abstract] [Google Scholar]
- Whiting PJ, Lindstrom JM. Characterization of bovine and human neuronal nicotinic acetylcholine receptors using monoclonal antibodies. J Neurosci. 1988 Sep;8(9):3395–3404. [Europe PMC free article] [Abstract] [Google Scholar]
- Williams M, Robinson JL. Binding of the nicotinic cholinergic antagonist, dihydro-beta-erythroidine, to rat brain tissue. J Neurosci. 1984 Dec;4(12):2906–2911. [Europe PMC free article] [Abstract] [Google Scholar]
- Wong ET, Holstad SG, Mennerick SJ, Hong SE, Zorumski CF, Isenberg KE. Pharmacological and physiological properties of a putative ganglionic nicotinic receptor, alpha 3 beta 4, expressed in transfected eucaryotic cells. Brain Res Mol Brain Res. 1995 Jan;28(1):101–109. [Abstract] [Google Scholar]
- Wonnacott S, Jackman S, Swanson KL, Rapoport H, Albuquerque EX. Nicotinic pharmacology of anatoxin analogs. II. Side chain structure-activity relationships at neuronal nicotinic ligand binding sites. J Pharmacol Exp Ther. 1991 Oct;259(1):387–391. [Abstract] [Google Scholar]
- Zoli M, Le Novère N, Hill JA, Jr, Changeux JP. Developmental regulation of nicotinic ACh receptor subunit mRNAs in the rat central and peripheral nervous systems. J Neurosci. 1995 Mar;15(3 Pt 1):1912–1939. [Europe PMC free article] [Abstract] [Google Scholar]
Associated Data
Articles from British Journal of Pharmacology are provided here courtesy of The British Pharmacological Society
Full text links
Read article at publisher's site: https://doi.org/10.1111/j.1476-5381.1996.tb15232.x
Read article for free, from open access legal sources, via Unpaywall:
https://europepmc.org/articles/pmc1909326?pdf=render
Citations & impact
Impact metrics
Article citations
Dopamine and Norepinephrine Tissue Levels in the Developing Limbic Brain Are Impacted by the Human CHRNA6 3'-UTR Single-Nucleotide Polymorphism (rs2304297) in Rats.
Int J Mol Sci, 25(7):3676, 26 Mar 2024
Cited by: 1 article | PMID: 38612487 | PMCID: PMC11011259
The Anti-Aggregative Peptide KLVFF Mimics Aβ1-40 in the Modulation of Nicotinic Receptors: Implications for Peptide-Based Therapy.
Biomedicines, 10(9):2231, 08 Sep 2022
Cited by: 2 articles | PMID: 36140331 | PMCID: PMC9496455
Differentiating the Neuropharmacological Properties of Nicotinic Acetylcholine Receptor-Activating Alkaloids.
Front Pharmacol, 13:668065, 22 Mar 2022
Cited by: 1 article | PMID: 35392565 | PMCID: PMC8980233
Evidence for Alpha7 Nicotinic Receptor Activation During the Cough Suppressing Effects Induced by Nicotine and Identification of ATA-101 as a Potential Novel Therapy for the Treatment of Chronic Cough.
J Pharmacol Exp Ther, 380(2):94-103, 15 Nov 2021
Cited by: 2 articles | PMID: 34782407 | PMCID: PMC8969114
Cyanotoxins and the Nervous System.
Toxins (Basel), 13(9):660, 16 Sep 2021
Cited by: 11 articles | PMID: 34564664 | PMCID: PMC8472772
Review Free full text in Europe PMC
Go to all (165) article citations
Similar Articles
To arrive at the top five similar articles we use a word-weighted algorithm to compare words from the Title and Abstract of each citation.
Nicotine-evoked [3H]5-hydroxytryptamine release from rat striatal synaptosomes.
Neuropharmacology, 39(2):290-299, 01 Jan 2000
Cited by: 51 articles | PMID: 10670424
Persistent nicotinic blockade by chlorisondamine of noradrenergic neurons in rat brain and cultured PC12 cells.
Br J Pharmacol, 125(6):1218-1227, 01 Nov 1998
Cited by: 11 articles | PMID: 9863650 | PMCID: PMC1565714
Effects of lobeline and dimethylphenylpiperazinium iodide (DMPP) on N-methyl-D-aspartate (NMDA)-evoked acetylcholine release in vitro: evidence for a lack of involvement of classical neuronal nicotinic acetylcholine receptors.
Neuropharmacology, 36(1):39-50, 01 Jan 1997
Cited by: 13 articles | PMID: 9144640
Modulatory role of presynaptic nicotinic receptors in synaptic and non-synaptic chemical communication in the central nervous system.
Brain Res Brain Res Rev, 30(3):219-235, 01 Nov 1999
Cited by: 160 articles | PMID: 10567725
Review