Abstract
Free full text

Asymmetric localization of Numb in the chick somite and the influence of myogenic signals
Abstract
While Notch signaling is known to play an essential role in the formation of somites, its role during later stages of somite maturation is less well understood. Here we examine the signals and transcription factors that control the expression of the Notch antagonist, Numb, during somite maturation in the chick embryo. Numb mRNA is present in the epithelial somite and is increased in expression in the forming myotome. Numb protein displays a very specific sub-cellular localization and dynamic expression during somite maturation. Numb protein is asymmetrically localized in a cortical crescent on the basal side of dividing cells in the dorso-medial lip of the dermomyotome and is subsequently uniformly distributed throughout differentiated myotomal cells. Treatment of somites with either the combination of Wnt-3a and Shh, or ectodermal signals plus noggin, both of which induce somitic myogenesis, did not significantly affect Numb transcript levels but led to a dramatic increase in the levels of Numb protein which was uniformly distributed throughout the cytoplasm of the resultant myotubes. Forced expression of MyoD in somites similarly induced high levels of Numb protein throughout the cytoplasm, without affecting Numb mRNA levels. We also found that signals that promote somitic myogenesis or forced MyoD expression induced expression of the Notch ligand, Serrate-2. Our findings suggest that Notch signals are specifically repressed in the myotome and that asymmetric expression of Numb in dividing cells of the dorso-medial lip of the dermomyotome may modulate whether these cells continue to divide or differentiate into myotomal cells.
Introduction
In vertebrates, cells that eventually form the skeletal muscles of the trunk and limbs, differentiate from the dorsal part of the somite, the dermomyotome (Christ and Ordahl, 1995; Tajbakhsh and Buckingham, 2000; Gros et al., 2005; Kassar-Duchossoy et al., 2005; Relaix et al., 2005). Cells of the dermomyotome proliferate and form a pool of undifferentiated muscle progenitor cells. Cells located within the dorso-medial lip of the dermomyotome are the first to differentiate into skeletal muscle and migrate underneath the dermomyotome to form the medial region of the myotome (Denetclaw et al., 2001; Ordahl et al., 2001; Kahane et al., 2002; Venters and Ordahl, 2002; Gros et al., 2004); subsequently cells from all four borders of the dermomyotome give rise to myotomal cells (Gros et al., 2004). Each step of the muscle differentiation program can be distinguished by the expression of specific transcription factors. The dermomyotome expresses the paired box transcription factors, Pax-3 and Pax-7 (Goulding et al., 1991; Jostes et al., 1991). As cells enter the myotome and differentiate into skeletal muscle, the expression of these factors diminishes, while expression of the myogenic bHLH transcription factors Myf-5 and MyoD are up-regulated (Sassoon et al., 1989; Hirsinger et al., 2001; Kiefer and Hauschka, 2001). Pax-3 has been documented to be upstream of MyoD expression in the trunk (Tajbakhsh et al., 1997), and forced expression of Pax-3 in somitic cells is able to induce MyoD expression and skeletal muscle differentiation in tissue culture (Maroto et al., 1997). MyoD and Myf-5 in turn are required for the differentiation of skeletal muscle throughout the body (Rudnicki et al., 1993), and forced expression of these myogenic bHLH transcription factors can induce skeletal muscle differentiation in a variety of cell types (Weintraub et al., 1989).
Many extrinsic signals have been documented to control the patterning, differentiation, expansion and survival of skeletal muscle cells in the somite. While intrinsic Wnt signals within the somite have been found to be necessary for the induction of MyoD (Linker et al., 2003), Sonic hedgehog (Shh) secreted by the notochord and ventral neural tube acts in concert with Wnt-1 and Wnt-3a secreted by the dorsal neural tube to promote myotome formation (Munsterberg et al., 1995; Stern et al., 1995; Fan et al., 1997; Ikeya and Takada, 1998; Borycki et al., 1999; Gustafsson et al., 2002). In addition, Shh promotes the survival and expansion of skeletal muscle precursors (Duprez et al., 1998; Teillet et al., 1998; Marcelle et al., 1999; Kruger et al., 2001), and is necessary for the maintained expression of myogenic bHLH genes in the myotome (Chiang et al., 1996; Coutelle et al., 2001; Teboul et al., 2003). Also, signals from the surface ectoderm, which can be mimicked by Wnt-4 or Wnt-6, can induce dermomyotome formation (Fan et al., 1997) and expression of MyoD (Tajbakhsh et al., 1998). Bone morphogenetic protein (BMP), which is secreted by the lateral plate mesoderm can act as an inhibitor of myogenesis and can be counteracted by the BMP antagonist, Noggin, which is expressed in the dorso-medial lip of the dermomyotome (Hirsinger et al., 1997; Marcelle et al., 1997; Reshef et al., 1998; Tonegawa and Takahashi, 1998). Interestingly, Shh and Wnt signals induce Noggin expression in the dorso-medial lip of the dermomyotome, which in turn stimulates dermomyotomal cells to enter the myotomal compartment (Hirsinger et al., 1997; Marcelle et al., 1997; Reshef et al., 1998).
In addition to Wnt, Shh and BMP signals, the Notch pathway has also been shown to influence myogenesis. Notch signaling is triggered by the interaction of the Notch receptor with one of its ligands, Delta or Serrate/Jagged (Mumm and Kopan, 2000). Following ligand binding, the Notch receptor undergoes a proteolytic cleavage that releases the Notch intracellular domain (NICD) to the cytoplasm. The NICD translocates to the nucleus, associates with members of the CSL (CBF1/RBP-Jκ, Suppressor of hairless (Su(H)), LAG-1) family, and modulates the transcriptional activity of these proteins. In the case of CBF1/RBP-Jκ, association with the NICD turns this transcriptional repressor into an activator (Kao et al., 1998). The NICD-CSL complex activates the expression of downstream targets including members of the Hairy/Enhancer-of-Split family of bHLH transcription factors. In vitro studies in mouse cell lines have shown that forced expression of either Notch ligands or activated Notch receptor inhibits muscle differentiation (Kopan et al., 1994; Shawber et al., 1996; Luo et al., 1997; Kuroda et al., 1999; Nofziger et al., 1999; Wilson-Rawls et al., 1999). This inhibition is mediated by at least two pathways: one pathway operates upstream of MyoD and involves transcriptional activation of CSL and the Hairy-related transcription factors HES-1 and/or Hey-1 (Kuroda et al., 1999; Nofziger et al., 1999; Sun et al., 2001). The second pathway appears to operate independent of CSL (Nofziger et al., 1999). Consistent with this, the NICD itself can directly bind to and inhibit the transcriptional activity of MEF-2C, a transcription factor, which acts downstream of myogenic bHLH proteins to promote skeletal muscle differentiation (Wilson-Rawls et al., 1999).
In vertebrate embryos, Notch receptors and ligands are widely expressed in the somite. Notch-1 RNA is expressed in the pre-somitic mesoderm and later is concentrated in the medial portions of the dermomyotome and myotome in both chick (Hirsinger et al., 2001) and mouse (Williams et al., 1995) embryos, while mouse Notch-2 and -3 are expressed in the dermomyotome of more mature somites (Williams et al., 1995). Chick Delta-1 is found in the caudal portion of each somite in both the dermomyotome and myotome, and chick Serrate-2 is expressed in the differentiating myotome (Hirsinger et al., 2001). Although mouse mutants bearing mutations in Notch-1 or Delta-1 develop abnormalities in somite formation, they do not possess obvious defects in skeletal muscle differentiation (Conlon et al., 1995; de la Pompa et al., 1997; Hrabe de Angelis et al., 1997). These results suggest that various Notch ligands and receptors may display functional redundancy during skeletal muscle formation. However, mice mutant for CBF1/RBP-Jκ lack somitic expression of the myogenic factor, myogenin (Oka et al., 1995), supporting a role for the Notch pathway in modulating muscle cell differentiation in vivo. Furthermore, forced expression of Delta-1 in chick embryos inhibits expression of MyoD and blocks skeletal muscle differentiation in both the myotome and developing wing bud (Delfini et al., 2000; Hirsinger et al., 2001).
Because Notch and its ligands are expressed in the somite while myotome formation occurs, it seems plausible that Notch signaling may be actively repressed in this somitic domain. One mechanism for the inhibition of Notch signaling is through expression of the Notch inhibitor, Numb. Numb blocks Notch signaling by binding to the C-terminus of the NICD and has been reported to both prevent its nuclear translocation (Frise et al., 1996; Guo et al., 1996; Wakamatsu et al., 1999) and induce its endocytosis (Santolini et al., 2000; Berdnik et al., 2002). In dividing myoblasts and neuroblasts of the fly, Numb protein is asymmetrically localized in a cortical crescent on one side of a dividing cell and is required for specification of distinct muscle (Ruiz Gomez and Bate, 1997; Carmena et al., 1998) and neural (Frise et al., 1996; Spana and Doe, 1996) cell fates. Recently, a role for Numb in vertebrate myogenesis was shown in postnatally-derived muscle satellite cells (Conboy and Rando, 2002). Numb is localized asymmetrically in the daughters of these dividing muscle progenitor cells and its expression correlates with induction of myogenic regulatory factors, cell cycle withdrawal, and loss of Pax-3 expression (Conboy and Rando, 2002). Moreover, Numb over-expression promotes the differentiation of skeletal muscle progenitors (Conboy and Rando, 2002). Despite this new information regarding the role of Numb in postnatal vertebrate myogenesis, its role during muscle development in the embryo has not been ascertained.
Here we examine the dynamics of Numb RNA and protein expression during somitic myogenesis in chick embryos. Numb is asymmetrically localized to the basal surface of dividing cells in the dorso-medial lip of the dermomyotome which contains precursors for both the dermomyotome and myotome, In addition, Numb is expressed throughout the cytoplasm of differentiated muscle cells in the myotome. We find that signals and transcription factors that promote skeletal muscle formation induce the accumulation of Numb protein (by possibly a post-transcriptional mechanism) and simultaneously up-regulate the expression of the Notch ligand, Serrate-2. Together, these findings indicate that dividing cells within the dorso-medial lip of the dermomyotome display asymmetric accumulation of Numb protein and that induction of the myotome by skeletal muscle inducing signals results in the high uniform expression of Numb protein throughout the cytoplasm and the simultaneous expression of Serrate-2 in this tissue. The coordinated expression of both a Notch antagonist (i.e., Numb) and ligand (Serrate-2) within the myotome ensures that Notch signaling is selectively inhibited within this somitic domain and may be promoted in the surrounding dermomyotome and sclerotome.
Results
Numb mRNA is expressed in somites
We initially examined Numb RNA expression during somite differentiation in the chick embryo and compared it with that of MyoD and its upsteam activator, Pax3. RT-PCR analysis indicated that Numb, Notch-1, and Pax3 transcripts were expressed throughout the presomitic mesoderm as well as in newly formed somites I-XII isolated from stage 14 chick embryos, while transcripts for MyoD were most highly expressed in the anterior somites (Fig. 1A). To further examine the localization of Numb transcripts within the somites we performed in situ hybridization for Numb transcripts on transverse sections through either somite 10 from a stage 17 embryo or through a trunk level somite from a stage 25 embryo. While Numb RNA was uniformly expressed within the neural tube, it appears to be up-regulated within the developing myotome and expressed at lower levels in the sclerotome and dermomyotome (Figs. 1B and 1C).
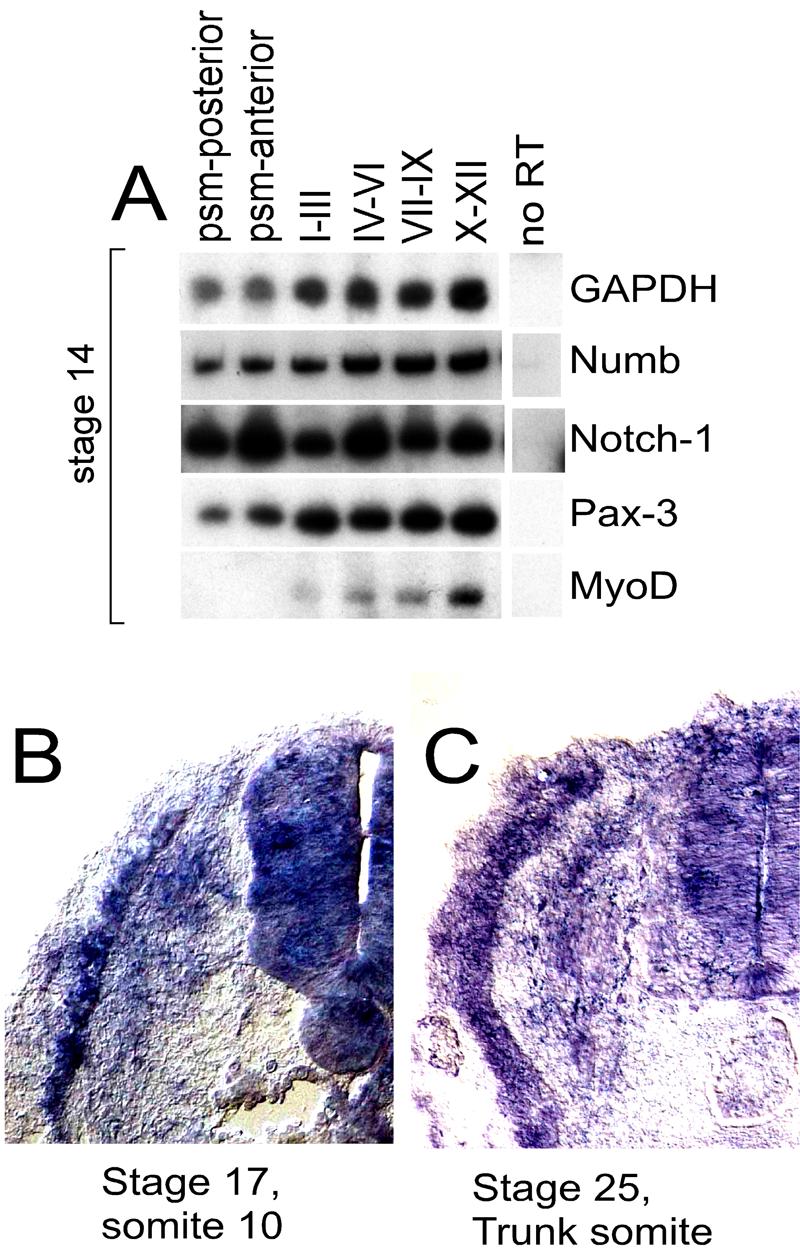
(A) RT-PCR analysis of gene expression in paraxial mesoderm isolated from differing axial levels of a stage 14 chicken embryo. (B) and (C) Section in situ hybridization for Numb expression in transverse sections through somite 10 from a stage 17 chicken embryo (B) or through a trunk level somite from a stage 25 chicken embryo (C).
Numb protein is asymmetrically localized during somitic myogenesis
To evaluate Numb protein distribution during somitic myogenesis, we performed immunofluorescent staining with antisera made against chick Numb and observed the localization of Numb protein using confocal microscopy (Wakamatsu et al., 1999). As this antisera was generated against a region of Numb which is also conserved in the mouse homolog of a related protein, Numblike (Zhong et al., 1997), it is possible that this antisera recognizes both Numb and Numblike in the chick. In anterior somites of a 3-day embryo (stage 17), Numb protein was present in the myotome (Fig. 2 A,B). At the trunk level, where the myotome is less mature, Numb is present in only a few cells (Fig. 2 E,F). In immature epithelial somites in the caudal regions of the embryo, we did not detect Numb protein (Fig. 2 H,I). Therefore, despite the presence of Numb RNA at all somite levels, accumulation of Numb protein appears to be restricted to the myotome, where it initially accumulates along the ventral edge of myotomal cells adjacent to the sclerotome (Fig. 2A).
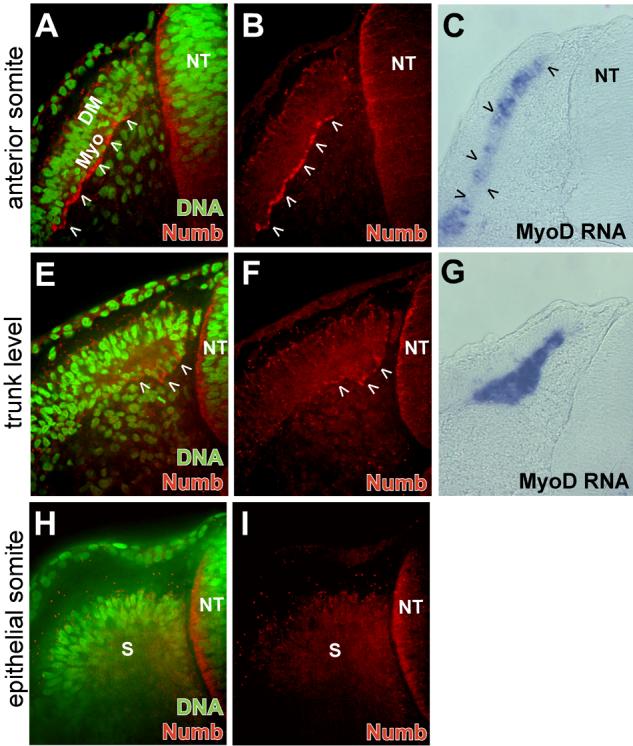
Confocal microscope images displaying Numb (red) and DNA (green) localization at differing axial levels of a stage 17 chicken embryo. (A and B) Numb protein is specifically localized on the ventral side of myotomal cells (arrowheads) of an anterior somite. (C) Section in situ hybridization staining for MyoD RNA (blue) at the same axial level displayed in (A and B) shows MyoD expression throughout the myotome (arrowheads). (E and F) At the trunk level, Numb protein is distributed in crescents in a few myotomal cells (arrowheads). (G) Section in situ hybridization staining for MyoD RNA (blue) at the same axial level displayed in (E and F). (H and I) In immature epithelial somites, Numb protein is not observed. Medial region of the somite adjacent to neural tube is on right side of each image. DM, dermomyotome; Myo, myotome; NT, neural tube; s, epithelial somite.
In anterior somites at stage 22, Numb protein continues to show an asymmetric distribution (Fig. 3 A,B). Crescents of Numb protein could be seen in cells of the dorso-medial lip of the dermomyotome, specifically on the basal side (i.e., on the outer surface of the dorso-medial lip). In contrast, within the myotome, Numb protein was distributed throughout the cytoplasm, while very little Numb was seen in the more lateral dermomyotome (Fig. 3 D,E). The region of high uniform cytoplasmic Numb protein expression corresponds to the MyoD and myosin heavy chain (MHC) expressing regions of the maturing myotome (Fig. 3 C,D). Within the myotome, Numb protein co-localizes with MHC except for a narrow band along the ventral edge of the myotome where Numb, but not MHC, is found (Fig. 3D).
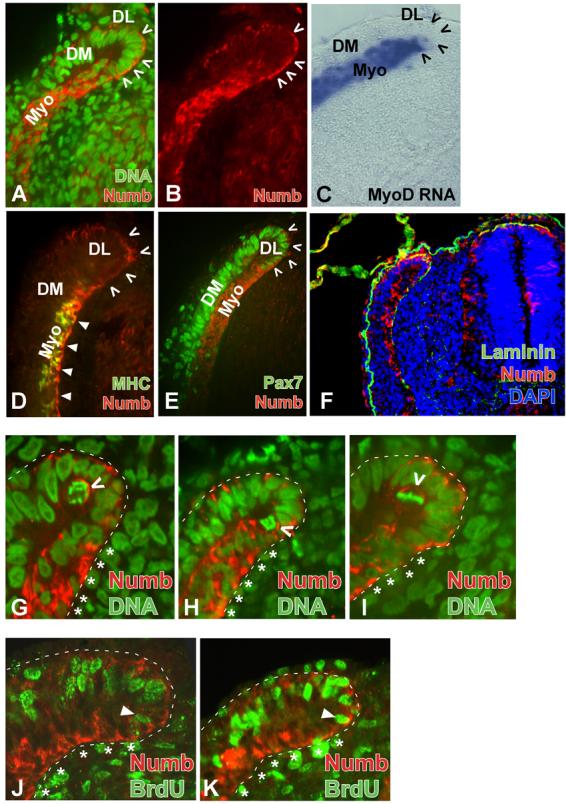
(A and B) Confocal microscope images displaying Numb (red) and DNA (green) localization in anterior somites from a stage 22 chicken embryo. Crescents of Numb protein (red) are located on the basal surface of the dorso-medial lip of the dermomyotome (DL, arrowheads). High levels of Numb protein are present throughout the cytoplasm in the myotome. DM, dermomyotome; Myo, myotome. (B) Same image as in A displaying only Numb protein. (C) Section in situ hybridization staining for MyoD RNA (blue) at the same axial level displayed in (A and B) shows MyoD expression throughout the myotome. Dorso-medial lip is delineated by open arrowheads. (D) Confocal microscope images displaying Numb (red) and myosin heavy chain (MHC; green) localization in anterior somites from a stage 22 chicken embryo. Ventral edge of the myotome that lacks MHC immunoreactivity retains Numb staining (solid arrowheads). DL delineated by open arrowheads. (E) Confocal microscope images displaying Numb (red) and Pax7 (green) localization in anterior somites from a stage 22 chicken embryo. In the DL of the dermomyotome, crescents of Numb protein (red, open arrowheads) are found in cells that express Pax7 (green). Cells of the myotome (Myo) that do not express Pax7 show uniform cytoplasmic Numb protein accumulation. (F) Numb accumulates in cells that lie adjacent to a basal lamina. Numb (red) and Laminin (green) expression in an anterior somite from a stage 25 chick embryo. Dapi staining (blue) identifies nuclei of cells. (G, H, I) Confocal image of the dorso-medial lip of the dermomyotome in anterior somites of a stage 22 chick embryo showing numb (red) accumulation on the basal side of mitotic cells. Dashed line outlines border of somite. Asterisks (*) align with the myotome. Nuclei stained with Sytox green. Open arrowheads indicate Numb crescents (red) on the basal surface of mitotic cells in which chromosomes are condensed. (J, K) Confocal image of the BrdU incorporation (green) and Numb (red) accumulation in the dorso-medial lip of the dermomyotome in anterior somites of a stage 22 chick embryo. Proliferating cells in the dermomyotome and in the dorso-medial lip of the dermomyotome incorporate BrdU (green) and display cortical crescents of Numb protein (red). High levels of Numb accumulation in post-mitotic myotomal cells (BrdU-negative cells, indicated with *) is observed throughout their cytoplasm. Closed arrowhead indicates last BrdU+ cell in dorso-medial lip of the DM that borders the myotome and has asymmetric Numb protein. Note that Numb staining in J and K is poor due to treatment required to reveal BrdU antigenicity. Medial region of the somite adjacent to neural tube is on right side of each image
Since Numb protein accumulates in a crescent pattern in cells located at the dorso-medial lip of the dermomyotome, where differentiated muscle cells first arise from the dermomyotome, we compared the expression of the dermomyotomal marker Pax-7 (Goulding et al., 1991) with that of Numb. Antibody staining for Pax-7 revealed nuclear localization in cells of the dermomyotome and in the dorso-medial lip of the dermomyotome (Fig. 3E). Only cells in the dorso-medial lip displayed crescents of Numb as well as nuclear Pax-7 (Fig. 3E, cells marked with arrowheads). We observed a sharp transition in Pax-7 expression at the boundary between the dorso-medial lip of the dermomyotome (Pax-7+) and the myotome (Pax-7−) (Fig. 3E). Interestingly, Numb protein distribution changed at this boundary from cortical crescents in the Pax-7-positive dorso-medial lip dermomyotomal cells to more uniform cytoplasmic expression in the Pax-7-negative myotomal cells (Fig. 3E). Numb expression in cells located in the dorso-medial lip of the dermomotome seemed to be greatest on the basal surface of these cells (see for example Figs. 3A and 3B). To ascertain if Numb expressing cells lie adjacent to a basal lamina, we evaluated the expression of Numb and Laminin in the anterior somites of stage 22 chick embryos. We observed that Numb expressing cells were indeed situated adjacent to a basal lamina in both the dorsal-medial lip of the dermomyotome and in the myotome proper (Fig. 3F).
In summary, whereas Numb mRNA expression is widespread in the somitic mesoderm, with a slight increase in the early myotome, Numb protein distribution is much more dynamic. Cortical crescents of Numb are present on the basal side of cells in the dorso-medial lip of the dermomyotome, while high amounts of Numb protein accumulate throughout the cytoplasm of differentiating myotomal cells.
Numb protein is localized to the basal side of dividing cells in the dorso-medial lip of the dermomyotome
Since the dermomyotome constitutes a source of progenitors for embryonic skeletal muscle (Venters and Ordahl, 2002; Gros et al., 2005; Kassar-Duchossoy et al., 2005; Relaix et al., 2005), we wanted to correlate Numb protein expression with the mitotic activity of these cells. In cryosections of somites, the number of mitotic cells with condensed chromosomes was very rare compared to that found in the neural tube (about 1/10 as many). Nevertheless cells with mitotic chromosomes were located in both the cleft of the dorso-medial lip of the dermomyotome (Fig. 3G-I) as well as in the lateral dermomyotome (data not shown). Of 16 cells in the cleft of the dorso-medial lip that had condensed chromosomes, 11 displayed a localized crescent of Numb staining, which in all cases was localized to the basal side of the mitotic figures (3 examples are shown in Fig. 3G-I). The remaining 5 mitotic cells did not have any detectable Numb. Similar findings were recently published by Venters and Ordahl who observed that Numb specifically accumulated on the basal side of cells in the dorso-medial lip of the dermomyotome undergoing mitosis in the basal-apical orientation (which are enriched in the dorso-medial lip of the dermomotome) and is absent from cells undergoing mitosis in a planar orientation (which are enriched in the dermomyotomal sheet; (Venters and Ordahl, 2005)).
Because of the rarity of mitotic figures, BrdU was used to label cells undergoing proliferation during a longer time span (i.e., 30 minutes prior to tissue fixation). Consistent with our analysis of condensed chromosomes, only cells of the dermomyotome and dorso-medial lip of the dermomyotome were BrdU-labeled (Fig. 3J, K). Cells bearing crescents of Numb were BrdU-labeled and therefore were about to undergo mitosis. Cells in the myotome, which displayed a homogeneous Numb distribution, were post-mitotic as indicated by the lack of BrdU staining. Our observations suggest that, upon asymmetric cell division, basally located daughter cells in the dorso-medial lip of the dermomyotome specifically accumulate Numb protein. When cells from the dorso-medial lip join the myotome they become post-mitotic and express Numb protein more uniformly throughout their cytoplasm.
Exposure to myogenic stimuli leads to expression of the Notch ligand, Serrate-2, without significantly altering Numb mRNA levels
To determine if muscle differentiation leads to an induction in Numb mRNA expression, we employed RT-PCR analysis to assay Numb expression in somite cultures exposed to myogenic stimuli. In addition, we also assayed the expression of chick Serrate-2 (a homolog of mouse Jagged-2), as this Notch ligand has been reported to be expressed in the myotome (Hirsinger et al., 2001) . Somites I-III were explanted from a stage 10 chick embryo and infected with an avian retrovirus encoding either Green Fluorescence Protein (GFP) or Wnt-3a. Such explants were cultured in either the absence or presence of Shh for 5 days. While others have observed that Wnt signals are able to induce the expression of the dermomyotomal marker Pax3 in murine presomitic mesoderm cultured for only 24 hours (Fan et al., 1997), when chicken somites are cultured for a more extended period of time (i.e., 5 days), both Wnt and Shh signals are apparently necessary to both maintain the expression of Pax3 and induce the expression of myogenic markers (Fig 4, lanes 1-4). As Shh signals have been suggested to be required for the survival of the myotome in vivo (Teillet et al., 1998), the apparent requirement for Shh to maintain the expression of Pax3 in long term somite cultures may similarly reflect a requirement for Shh to promote either the survival or proliferation of Pax3-expressing cells cultured in vitro. While the expression of Numb mRNA was not significantly affected by pro-myogenic signals, culture of somites with the combination of Wnt-3a and Shh induced both the skeletal muscle differentiation program and induced the robust expression of Serrate-2 (Fig. 4, lanes 1-4). Thus, somitic myogenesis correlates with the induction of the Notch ligand, Serrate-2 but fails to significantly affect Numb mRNA levels.
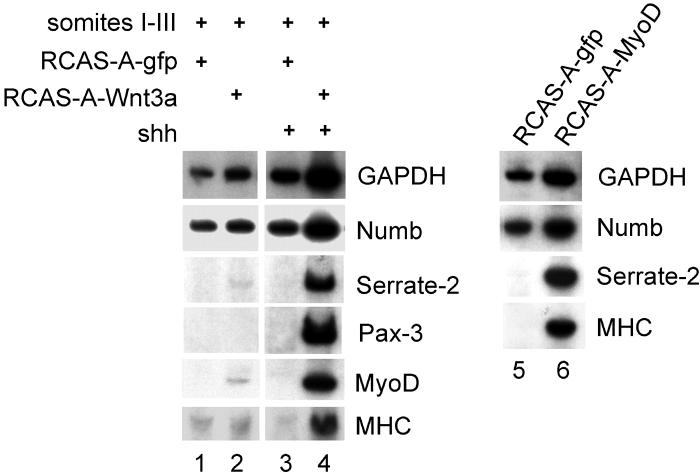
Somites I-III were explanted from a stage 10 chick embryo and infected with an avian retrovirus encoding either Green Fluorescence Protein (GFP), Wnt-3a, or MyoD as indicated and cultured in either the absence or presence of Shh (as indicated). After 5 days culture, the explants were harvested and gene expression was assayed by RT-PCR analysis.
To determine if up-regulation of Serrate-2 is a corollary of somitic skeletal muscle differentiation, we examined if forced expression of MyoD in somites could similarly induce the expression of Serrate-2. Indeed, infection of somites with a retrovirus encoding MyoD led to both the induction of myosin heavy chain and Serrate-2 expression (Fig. 4, lane 6) indicating that MyoD can either directly or indirectly induce the expression of Serrate-2 in myotomal cells. In contrast, MyoD expression failed to significantly affect the expression level of Numb mRNA. In addition, we could detect low levels of the Notch ligands, Delta-1 and Serrate-1, in these somite cultures, but their expression was not significantly affected by either muscle inducing signals or forced MyoD expression (data not shown). These results confirm previous in situ hybridization data showing that Serrate-2 is expressed in the differentiating myotome (Hirsinger et al., 2001).
Numb protein levels are dramatically enhanced in somite cultures exposed to myogenic stimuli
As opposed to the striking up-regulation in Serrate-2 mRNA expression by either myogenic inducing signals or forced MyoD expression, levels of Numb mRNA were not significantly modulated by either of these myogenic stimuli (Fig. 4, lanes 1-6). Therefore, we set out to determine whether signals that induce somitic myogenesis specifically regulate the cytoplasmic distribution of Numb protein. Antibody staining of somite cultures revealed that various myogenic stimuli do indeed enhance the levels of Numb protein accumulation (Fig. 5). Addition of either Shh (n=6; Fig. 5A) or Wnt3a virus alone (n=6; not shown) did not lead to any notable increase in Numb protein. In contrast, the combination of both Wnt-3a and Shh, which induces somitic myogenesis ((Munsterberg et al., 1995); Fig. 4, lane 4), led to a marked increase in the amount of Numb staining in 19 out of 20 cultures (Figs. 5B,C). These Numb-positive, multinucleate cells resembled myotubes and could be co-stained with an anti-myosin heavy chain (MHC) antibody (data not shown). Another way to induce somitic myogenesis is by co-culture of somites I-III with the overlying ectoderm and addition of Noggin-conditioned medium (Reshef et al., 1998). While somites cultured with either Noggin conditioned medium alone (n=6; not shown) or with only the surface ectoderm (n=6; Fig. 5D) did not contain a substantial amount of either Numb or MHC, somites cultured with both ectoderm and Noggin-conditioned medium displayed a robust increase in expression of both Numb and MHC in the resultant myotubes (17 of 20 cultures; Figs. 5E,F). Interestingly, although Numb and MHC were present in the same cells, exact overlap of the two proteins occurred in only a few domains (Fig. 5E), indicating that the two proteins occupy different cellular compartments.
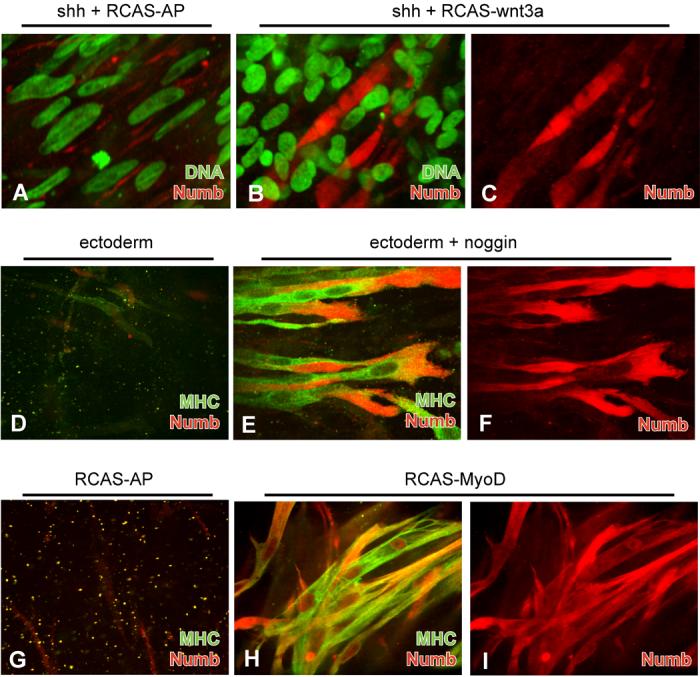
(A-I) Somites I-III were explanted from a stage 10 chick embryo and infected with an avian retrovirus (RCAS) encoding either alkaline phosphatase (AP), Wnt-3a, or MyoD as indicated and cultured in either the absence or presence of Shh, noggin, or overlying ectoderm (as indicated). (A) Addition of Shh and control RCAS-AP virus to somite explants does not enhance Numb protein levels (red). Nuclei were stained with Sytox Green. (B) Addition of both Shh and RCAS-Wnt3a to somite explants leads to significant Numb accumulation in mature myotubes. (C) Same cells as in (B) with Numb staining alone. (D) Co-culture of somites with ectoderm does not lead to enhancement of Numb (red) or MHC (green) expression. (E) Addition of Noggin to somite-ectoderm cultures induces high levels of Numb (red) and MHC expression (green) throughout the myotubes. Although present in the same cells, exact overlap of Numb and MHC (yellow) is visible in only a few domains, indicating that the two proteins occupy different cellular compartments. (F) Same cells as in (E) showing only Numb staining. (G) Control somite cultures infected with RCAS-AP show neither Numb (red) nor MHC (green) expression. (H) Somite cultures infected with RCAS-MyoD display high levels of Numb (red) and MHC (green) expressioin. (I) Same cells as in (H) showing only Numb staining.
Over-expression of the bHLH transcription factor, MyoD, can lead to skeletal muscle differentiation in several cell types (Davis et al., 1987; Weintraub et al., 1989). To see if the enrichment in uniform cytoplasmic Numb protein was downstream of MyoD, we infected chick somites with a MyoD-expressing retrovirus. While a control virus did not lead to any myogenesis or detectable Numb expression (n=12; Fig. 5G), the MyoD-expressing virus induced the formation of large myotubes that expressed high levels of both Numb and MHC proteins in all treated cultures (n=12; Fig. 5H,I). Hence, the high uniform cytoplasmic distribution of Numb protein induced by myogenic stimuli such as Wnt plus Shh, or ectoderm plus Noggin, is likely to occur downstream of MyoD expression.
In summary, Numb protein is expressed uniformly throughout the cytoplasm in somitic cells undergoing myogenesis. Myogenic stimuli that induce MyoD expression (such as Wnt plus Shh or ectoderm plus Noggin) also increase Numb protein levels, but fail to significantly alter the expression of Numb RNA. The enhancement of Numb protein levels by muscle inducing cues likely occurs downstream of MyoD expression since forced expression of MyoD in somite cultures also leads to the accumulation of Numb protein throughout the cytoplasm.
Discussion
Differential Numb protein expression and localization in dermomyotomal versus myotomal cells
In this study, we describe the expression of Numb, an inhibitor of Notch signaling, during muscle formation in the chick embryo. We have found that Numb protein is asymmetrically localized to the basal surface of cells within the dorso-medial lip of the dermomyotome. The dorso-medial lip of the dermomyotome has been shown to contain progenitor cells that either remain within this structure or give rise to the myotome (Denetclaw et al., 2001; Ordahl et al., 2001; Kahane et al., 2002; Venters and Ordahl, 2002; Gros et al., 2004). Fate mapping (Cinnamon et al., 2001; Denetclaw et al., 2001; Gros et al., 2004) and gene expression analyses (Hirsinger et al., 2001; Kassar-Duchossoy et al., 2005; Relaix et al., 2005) have suggested that proliferating Pax-3/Pax-7-positive dermomyotomal cells give rise to Myf-5/MyoD-positive cells in the myotome. Pax-3-positive dermomotomal progeny are similarly thought to give rise to Myf-5/MyoD expressing myoblasts in the limb (Amthor et al., 1998; Delfini et al., 2000) and indeed dermomyotomal cells have recently been fate-mapped to give rise to satellite cells (Gros et al., 2005). While cells that remain within the dorso-medial lip of the dermomyotome maintain the expression of Pax-3/Pax-7 and continue to proliferate, those that migrate into the myotome lose expression of these Pax family members, activate expression of Myf-5 and MyoD, withdraw from the cell cycle, and express skeletal muscle structural proteins such as myosin heavy chain. Prior work has suggested that the BMP antagonist, noggin, may control the transition of a Pax-3-positive dermomyotomal cell into a MyoD-positive myotomal cell (Hirsinger et al., 1997; McMahon et al., 1998; Reshef et al., 1998; Amthor et al., 1999). Recent work has suggested that Notch signals may control this transition in satellite cells, which constitute a stem cell population for adult skeletal muscle (Conboy and Rando, 2002) and that Notch signaling is required for BMP-induced inhibition of myogenic differentation (Dahlqvist et al., 2003). Indeed, forced expression of Delta-1 in the somite can also block the developmental maturation of embryonic muscle precursors (Hirsinger et al., 2001) raising the possibility that the dermomyotomal and myotomal compartments of the somite may be subject to differing levels of endogenous Notch signaling.
The cells of the dorso-medial medial lip of the dermomyotome are fated to give rise to progeny cells in both the dermomyotome and myotome (Ordahl et al., 2001; Gros et al., 2004). We speculate that such cells may undergo asymmetric cell division to give rise to either another proliferating dermomyotomal progenitor cell or a nascent myotomal cell, which will ultimately withdraw from the cell cycle. Interestingly, we have found that dividing cells that lie in the cleft of the dorso-medial lip of the dermomyotome accumulate a crescent of Numb protein on their basal side. Thus, it seems most likely that the Numb expressing cells that arise in the cleft give rise to nascent myotomal cells. More mature myotomal cells which lie ventral to these progenitors contain a higher level of Numb protein which is uniformly localized throughout their cytoplasm (see Fig. 6A). We propose that asymmetric cell division, as revealed by Numb protein distribution, is an intrinsic property of muscle progenitor cells. Consistent with our results, Numb was also found to be asymmetrically localized in Pax-7-expressing muscle progenitor cells in adult regenerating skeletal muscle (Conboy and Rando, 2002).
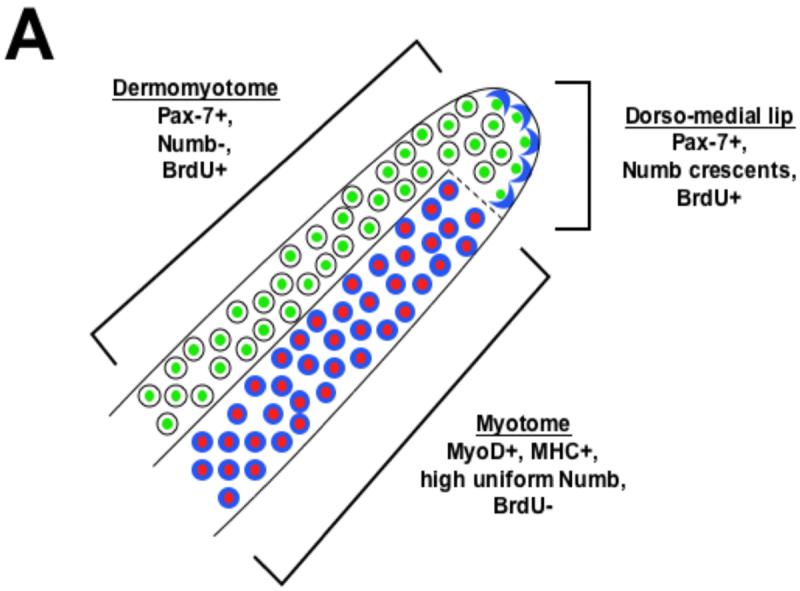
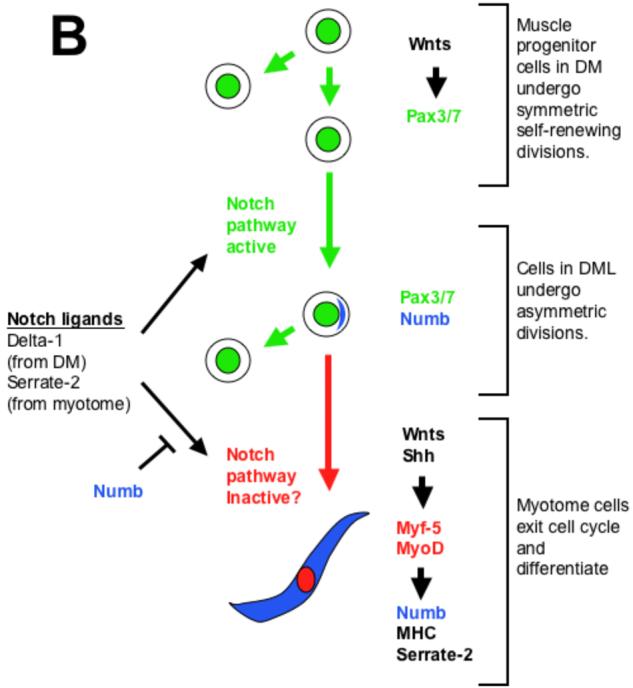
(A) Distribution of Numb protein reveals three somite compartments: Pax-7 expressing (green) dermomyotomal cells which lack Numb protein (blue), Pax-7 expressing cells of the dorso-medial lip of the dermomyotome in which Numb is localized asymmetrically in cortical crescents on the basal side of dividing cells, and MyoD/Myf-5 expressing (red) myotomal cells in which Numb protein accumulates uniformly. (B) A speculative model of some of the signals that modulate myogenesis in the somite. Wnt signals from the surface ectoderm lead to expression of Pax-3/7 in the dermomyotome (DM). Because cells in this compartment lack Numb protein, we speculate that they are sensitive to Notch signals and therefore undergo symmetric cell division. In the dorso-medial lip (DML) of the dermomyotome, localized Numb protein results in differential sensitivity to Notch ligands. We speculate that daughter cells that lack Numb remain in the dorso-medial lip of dermomyotome continue to proliferate, while those that contain Numb join the myotome and withdraw from the cell cycle. Muscle inducing cues from either within the somite (i.e., Wnt5a; (Linker et al., 2003)), the axial tissues (Wnts and Shh; (Munsterberg et al., 1995; Stern et al., 1995; Ikeya and Takada, 1998; Borycki et al., 1999)) or the surface ectoderm (Wnts; (Tajbakhsh et al., 1998)) induce expression of MyoD in Pax7-positive cells that contain a cortical crescent of Numb. The resultant differentiated myotomal cells contain uniformly high levels of Numb protein throughout their cytoplasm and secrete Serrate-2. We propose that Numb blocks the inhibitory effects of Notch signaling on muscle differentiation in the myotome, while Serrate-2 secreted by the myotome may promote the continued proliferation of dermomyotomal progenitors.
These striking differences in Numb protein expression and localization in proliferating dermomyotomal versus post-mitotic myotomal cells suggests that modulation of Notch signaling by Numb may act to modulate whether dermomyotomal precursors continue to proliferate or differentiate into post-mitotic myotomal cells, as has been suggested to be the case in satellite cells (Conboy and Rando, 2002). The asymmetric inheritance of a cortical crescent of Numb located on the basal side of progenitor cells in the dorso-medial lip of the dermomyotome may designate which progeny cells differentiate into myotome. In contrast, the high cytoplasmic level of Numb in differentiated skeletal muscle cells which is first observed in the myotome of stage 22 embryos may play a role in the maintenance of the differentiated phenotype but likely does not play a role in its induction. It has recently been shown that mitotically active Pax3/Pax7-expressing cells which do not express myogenic bHLH factors migrate into the myotome upon de-epithelialization of the dermomyotome (Kassar-Duchossoy et al., 2005; Relaix et al., 2005). While we have not assayed the expression of Numb in such cells, it seems plausible that these myotomal cells remain mitotically active (and fail to differentiate) because they fail to express Numb.
MyoD may induce the accumulation of Numb protein by a post-transcriptional mechanism
We have found that signals that promote somitic myogenesis, such as Wnt plus Shh, ectoderm plus Noggin, or forced expression of MyoD all induce uniform cytoplasmic accumulation of Numb protein in the resultant myotubes. Interestingly, each of these myogenic inducing regimens fails to significantly affect the levels of Numb RNA, suggesting that they act by a post-transcriptional mechanism to induce Numb protein levels. Several molecules have been reported to regulate Numb protein translation as well as stability. Musashi-1 binds Numb mRNA and prevents translation (Imai et al., 2001), while two other proteins, Siah-1 and LNX have been reported to target Numb for ubiquitin-dependent degradation (Susini et al., 2001; Nie et al., 2002). Thus, it is possible that MyoD may induce accumulation of Numb protein (in the absence of significantly affecting Numb RNA levels) by modulating the expression or activity of either Musashi-1, Siah-1 and LNX in the myotome. Alternatively, because the Numb anti-sera we employed also recognizes Numblike, it is possible that MyoD may induce the expression of Numblike in myotubes which would account for the increased Numb/Numblike immunostaining within these cells.
We observed an asymmetric distribution of Numb protein on the basal surface of cells located within the dorso-medial lip of the dermomyotome, while cells within the maturing myotome accumulate Numb both on their basal surface and uniformly throughout their cytoplasm. It was recently reported that transplantation of lateral dermomyotomal cells into the region of the dorso-medial lip of the dermomyotome, resulted in the induction of Numb protein on the basal surface of these cells (Venters and Ordahl, 2005). Taken together, our results and those of Ordahl's group suggest that signals present in the dorso-medial lip of the dermomyotome are sufficient to promote the asymmetric accumulation of Numb in dermomyotomal cells (Venters and Ordahl, 2005) and that subsequent expression of myogenic bHLH factors induces a uniform cytoplasmic accumulation of Numb protein in progeny cells that enter the myotome proper.
Numb protein may act to block Notch signals in both the myotome and cells of the dorso-medial lip of the dermomyotome
Numb is a cytoplasmic protein that binds to the intracellular domain of the Notch receptor and antagonizes Notch signaling (Frise et al., 1996; Guo et al., 1996; Wakamatsu et al., 1999). Numb also binds α-adaptin, a protein involved in receptor-mediated endocytosis (Santolini et al., 2000). Interestingly, when mutant forms of α-adaptin are present in Drosophila embryos, they cause phenotypes similar to those seen in numb mutant embryos (Berdnik et al., 2002). During myogenesis in Drosophila, asymmetric segregation of Numb protein influences the fate of muscle progenitor cells in the larval mesoderm (Carmena et al., 1998). Loss of Numb results in duplicated or deleted muscle founder cells, reminiscent of Notch gain-of-function phenotypes. Over-expression of Numb in satellite cells derived from regenerating adult mouse myofibers causes cell cycle exit and skews cells toward muscle differentiation (Conboy and Rando, 2002). This is a converse phenotype to that observed following over-expression of an activated Notch receptor in either myogenic cell lines (Kopan et al., 1994) or in satellite cells (Conboy and Rando, 2002). Thus, Numb apparently blocks the receipt of Notch signals in a variety of cellular contexts, suggesting that Notch signals are blocked or at least attenuated in Numb expressing cells in the dorso-medial lip of the dermomyotome and the myotome. It will be interesting to determine whether the asymmetric distribution of Numb in dividing cells within the dorso-medial lip of the dermomyotome marks progeny cells that are specifically destined to give rise to the myotome (see Fig. 6B).
MyoD coordinates the expression of a Notch ligand and a Notch antagonist within the myotome
We have found that signals that promote myogenesis, such as Wnt plus Shh, or forced expression of MyoD also induce expression of the Notch ligand, Serrate-2. In Xenopus embryos, Rupp and colleagues have similarly shown that MyoD induces the expression of Delta-1 (Wittenberger et al., 1999) . Secreted forms of Jagged-1 and -2, the mouse homologues of Serrate-1 and -2, have been shown to inhibit myogenesis in a muscle cell line (Shawber et al., 1996; Luo et al., 1997). While it initially seems paradoxical that myotomal cells express a molecule capable of inhibiting skeletal muscle differentiation, we propose that accumulation of Numb protein in these same cells blocks the effects of Notch signaling specifically in myotomal cells. Thus, it seems most likely that Serrate-2 is secreted by the myotome and activates the Notch signaling pathway in either adjacent dermomyotomal or sclerotomal cells, both of which lack Numb protein. What then is the utility of Notch ligands secreted by differentiated skeletal muscle cells? Analogous to other developmental systems, it seems plausible that Notch signals provided by the differentiated myotome may act to maintain the proliferation of dermomyotomal precursor cells. In this way, a balance between the number of skeletal muscle progenitors in the dermomyotome and differentiating myotomal cells can be maintained. Consistent with this hypothesis, regenerating skeletal muscle is known to up-regulate the expression of Delta-1 and thereby promote the proliferation of Pax-3 positive satellite cell precursors (Conboy and Rando, 2002). Studies are in progress to determine if Notch signals are similarly necessary to promote the proliferation of dermomyotomal cells.
Experimental Procedures
In situ hybridization
Whole-mount in situ hybridization for Numb (Wakamatsu et al., 1999) and MyoD (Lin et al., 1989) was carried out as previously described (Stern, 1998). Some stained embryos were embedded in OCT and sectioned (15-25 μm thick).
Cloning of RCAS-MyoD virus
The single myc-tagged mouse MyoD insert was isolated from pCS2-6mt-MyoD using Nco1 and Xho1 (blunted) and sub-cloned into the Nco1/Sma1 site of Slax13 (Riddle et al., 1993). The Slax-13 insert was transferred as a Cla1 fragment into RCASBP-A (Hughes et al., 1987).
Embryo and explant culture
Embryos were staged according to (Hamburger and Hamilton, 1951). Somitic tissue was dissected and cultured for 4 to 5 days as described (Munsterberg et al., 1995). RCAS viruses were harvested from chick embryo fibroblasts, concentrated and titered according to (Morgan and Fekete, 1996). Somites were incubated on ice for one hour with virus prior to plating (108 to 109 viral particles per mL).
Immunohistochemistry and confocal microscopy
Rabbit anti-Numb antibody (Wakamatsu et al., 1999) was used at a dilution of 1:400. Hybridoma supernatants (1:10) containing monoclonal anti-myosin heavy chain (MF20), 12-101, and anti-Pax-7 antibodies, and purified mouse anti-BrdU antibody (1:200) were purchased from the Developmental Studies Hybridoma Bank, University of Iowa. Sytox Green (Molecular Probes) was sometimes used as a nuclear counterstain (50 nM) and added to the secondary antibody solution. Secondary antibodies (1:250) were as follows: TRITC-donkey anti-rabbit antibody, FITC-donkey anti-mouse and Cy3-goat anti-mouse from Jackson Immunoresearch; Alexa 568-goat anti-rabbit and Alexa 488-goat anti-mouse antibodies from Molecular Probes.
Embryos were fixed for 2 hours at room temperature in 4% paraformaldehyde in PBS, washed in PBT (PBS with 0.1% Tween-20), incubated in blocking buffer (PBT, 5% goat serum, 1% horse serum, 0.1% sodium azide) for 2 hours at room temp, then incubated in diluted primary antibody in blocking buffer at 4°C overnight with gentle rotation. The following day they were rinsed in PBT and subjected to 6 one-hour washes with PBT. Secondary antibody incubations and subsequent washes were carried out similarly. Embryos were sliced transversely at a thickness of 1 mm using a #10 scalpel blade and placed on a slide in wells created with perforated strips of Sylgard. Mounting medium (Biomeda Gelmount) was added and a coverslip was placed on top.
After fixation and PBT washes, some embryos were frozen in OCT and sectioned at a thickness of 20-25 μm. BrdU staining was carried out as previously described (Hirsinger et al., 2001). Sections were washed with PBT, incubated in blocking buffer for 30 minutes, and incubated in diluted primary antibody overnight at 4°C. They were then washed with PBT, incubated with diluted secondary antibody for 2 hours at room temp, washed with PBT and mounted. For staining of explant cultures, explants embedded in collagen gels were fixed with 4% paraformaldehyde for 15 min at room temp, washed 6 times for 10 min with PBT, and blocked with blocking buffer. Primary and secondary antibody incubations were carried out at 4°C overnight, with 6 ten-minute PBT washes after each incubation. Confocal microscopy was performed using a Nikon TE300 microscope with Perkin Elmer spinning disk confocal attachments and Metamorph Image Acquisition software.
RT-PCR analysis of molecular markers
RNA was extracted from explants using the Qiagen RNeasy Mini Kit and Qiagen DNAse reagents according to the manufacturer's instructions except that for each sample, 100 ng of carrier RNA (Qiagen) was added to the lysis buffer. Reverse transcriptase (RT) reactions and polymerase chain reaction (PCR) analysis were carried out as previously described (Munsterberg et al., 1995) and (Maroto et al., 1997). PCR primers that were not previously published are as follows: Numb (Wakamatsu et al., 1999) 5′-ccg ggc gtt ctc ata cat ctg-3′, 5′-tgg ggg ttg ctc att tcc ttc-3′ (408 bp); Notch-1 (Wakamatsu et al., 1999) 5′-cgc ctc ccc tta cta cca ctg-3′, 5′-ccg tcc cac tca cac tca aa-3′ (453 bp product); Serrate-1 (Myat et al., 1996) 5′-ccc cga taa ata cca gtg ttc-3′, 5′-ggt ttg ccc tca cat tca ttc-3′ (312 bp); Serrate-2 (Hayashi et al., 1996) 5′-gcg acg aaa atg gaa aca aag-3′, 5′-cag ggg tta gat aca caa gca-3′ (400 bp); Delta-1 (Henrique et al., 1997) 5′-gcc gac ccc gcc ttc agc aac- 3′, 5′-aca gag cag cct tcc ccg tag-3′ (272 bp). We used 23 cycles to assay GAPDH and 28-30 cycles for other markers. Amplifications were tested to ensure that they were roughly linear and that no PCR product was obtained without the addition of RT.
Acknowledgements
We are very grateful to Y. Wakamatsu for the generous gift of antibodies and cDNA, to S. Evans and M. Spechler for technical assistance, and to W. Zhong for pointing out that the Numb antisera we employed may also recognize Numblike. Confocal microscopy was carried out at the Nikon Imaging Center at Harvard Medical School and was much facilitated through the help of its director, Dr. J. Waters Shuler. Most of all, we are indebted to the following members of the Lassar laboratory for helpful discussions: G. Di Rocco, J.B. Lazaro, H. Kempf, D.-W. Kim, R. Sohn, and E. Tzahor. T.H. was supported by a joint fellowship from the Foundation for Gene and Cell Therapy and the Canadian Institute for Health Research. L.Z. was supported by an NIH postdoctoral fellowship. This work was funded by a grant from the NIH (GM054879) to support work on somitogenesis awarded to A.B.L.
Grant sponsor: National Institutes of Health; Grant number: GM054879.
References
- Amthor H, Christ B, Patel K. A molecular mechanism enabling continuous embryonic muscle growth - a balance between proliferation and differentiation. Development. 1999;126:1041–1053. [Abstract] [Google Scholar]
- Amthor H, Christ B, Weil M, Patel K. The importance of timing differentiation during limb muscle development. Curr Biol. 1998;8:642–652. [Abstract] [Google Scholar]
- Berdnik D, Torok T, Gonzalez-Gaitan M, Knoblich JA. The endocytic protein alpha-Adaptin is required for numb-mediated asymmetric cell division in Drosophila. Dev Cell. 2002;3:221–231. [Abstract] [Google Scholar]
- Borycki AG, Brunk B, Tajbakhsh S, Buckingham M, Chiang C, Emerson CP., Jr. Sonic hedgehog controls epaxial muscle determination through Myf5 activation. Development. 1999;126:4053–4063. [Abstract] [Google Scholar]
- Carmena A, Murugasu-Oei B, Menon D, Jimenez F, Chia W. Inscuteable and numb mediate asymmetric muscle progenitor cell divisions during Drosophila myogenesis. Genes Dev. 1998;12:304–315. [Europe PMC free article] [Abstract] [Google Scholar]
- Chiang C, Litingtung Y, Lee E, Young KE, Corden JL, Westphal H, Beachy PA. Cyclopia and defective axial patterning in mice lacking Sonic hedgehog gene function. Nature. 1996;383:407–413. [Abstract] [Google Scholar]
- Christ B, Ordahl CP. Early stages of chick somite development. Anat Embryol (Berl) 1995;191:381–396. [Abstract] [Google Scholar]
- Cinnamon Y, Kahane N, Bachelet I, Kalcheim C. The sub-lip domain--a distinct pathway for myotome precursors that demonstrate rostral-caudal migration. Development. 2001;128:341–351. [Abstract] [Google Scholar]
- Conboy IM, Rando TA. The regulation of notch signaling controls satellite cell activation and cell fate determination in postnatal myogenesis. Developmental Cell. 2002;3:397–409. [Abstract] [Google Scholar]
- Conlon RA, Reaume AG, Rossant J. Notch1 is required for the coordinate segmentation of somites. Development. 1995;121:1533–1545. [Abstract] [Google Scholar]
- Coutelle O, Blagden CS, Hampson R, Halai C, Rigby PW, Hughes SM. Hedgehog signalling is required for maintenance of myf5 and myoD expression and timely terminal differentiation in zebrafish adaxial myogenesis. Dev Biol. 2001;236:136–150. [Abstract] [Google Scholar]
- Dahlqvist C, Blokzijl A, Chapman G, Falk A, Dannaeus K, Ibanez CF, Lendahl U. Functional Notch signaling is required for BMP4-induced inhibition of myogenic differentiation. Development. 2003;130:6089–6099. [Abstract] [Google Scholar]
- Davis RL, Weintraub H, Lassar AB. Expression of a single transfected cDNA converts fibroblasts to myoblasts. Cell. 1987;51:987–1000. [Abstract] [Google Scholar]
- de la Pompa JL, Wakeham A, Correia KM, Samper E, Brown S, Aguilera RJ, Nakano T, Honjo T, Mak TW, Rossant J, Conlon RA. Conservation of the Notch signalling pathway in mammalian neurogenesis. Development. 1997;124:1139–1148. [Abstract] [Google Scholar]
- Delfini M, Hirsinger E, Pourquie O, Duprez D. Delta 1-activated notch inhibits muscle differentiation without affecting Myf5 and Pax3 expression in chick limb myogenesis. Development. 2000;127:5213–5224. [Abstract] [Google Scholar]
- Denetclaw WF, Jr., Berdougo E, Venters SJ, Ordahl CP. Morphogenetic cell movements in the middle region of the dermomyotome dorsomedial lip associated with patterning and growth of the primary epaxial myotome. Development. 2001;128:1745–1755. [Abstract] [Google Scholar]
- Duprez D, Fournier-Thibault C, Le Douarin N. Sonic Hedgehog induces proliferation of committed skeletal muscle cells in the chick limb. Development. 1998;125:495–505. [Abstract] [Google Scholar]
- Fan C-M, Lee CS, Tessier-Lavigne M. A role for Wnt proteins in induction of dermomyotome. Dev. Biol. 1997;191:160–165. [Abstract] [Google Scholar]
- Frise E, Knoblich JA, Younger-Shepherd S, Jan LY, Jan YN. The Drosophila Numb protein inhibits signaling of the Notch receptor during cell-cell interaction in sensory organ lineage. Proc Natl Acad Sci U S A. 1996;93:11925–11932. [Europe PMC free article] [Abstract] [Google Scholar]
- Goulding MD, Chalepakis G, Deutsch U, Erselius JR, Gruss P. Pax-3, a novel murine DNA binding protein expressed during early myogenesis. EMBO. 1991;10:1135–1147. [Europe PMC free article] [Abstract] [Google Scholar]
- Gros J, Manceau M, Thome V, Marcelle C. A common somitic origin for embryonic muscle progenitors and satellite cells. Nature. 2005;435:954–958. [Abstract] [Google Scholar]
- Gros J, Scaal M, Marcelle C. A two-step mechanism for myotome formation in chick. Dev Cell. 2004;6:875–882. [Abstract] [Google Scholar]
- Guo M, Jan LY, Jan YN. Control of daughter cell fates during asymmetric division: interaction of Numb and Notch. Neuron. 1996;17:27–41. [Abstract] [Google Scholar]
- Gustafsson MK, Pan H, Pinney DF, Liu Y, Lewandowski A, Epstein DJ, Emerson CP., Jr Myf5 is a direct target of long-range Shh signaling and Gli regulation for muscle specification. Genes Dev. 2002;16:114–126. [Europe PMC free article] [Abstract] [Google Scholar]
- Hamburger V, Hamilton HL. A series of normal stages in the development of the chick embryo. Journal of Morphology. 1951;88:49–92. [Abstract] [Google Scholar]
- Hayashi H, Mochii M, Kodama R, Hamada Y, Mizuno N, Eguchi G, Tachi C. Isolation of a novel chick homolog of Serrate and its coexpression with C-Notch-1 in chick development. Int J Dev Biol. 1996;40:1089–1096. [Abstract] [Google Scholar]
- Henrique D, Hirsinger E, Adam J, Le Roux I, Pourquié O, Ish-Horowicz D, Lewis J. Maintenance of neuroepithelial progenitor cells by Delta-Notch signalling in the embryonic chick retina. Current Biol. 1997;7:661–670. [Abstract] [Google Scholar]
- Hirsinger E, Duprez D, Jouve C, Malapert P, Cooke J, Pourquie O. Noggin acts downstream of Wnt and Sonic Hedgehog to antagonize BMP4 in avian somite patterning. Development. 1997;124:4605–4614. [Abstract] [Google Scholar]
- Hirsinger E, Malapert P, Dubrulle J, Delfini MC, Duprez D, Henrique D, Ish-Horowicz D, Pourquie O. Notch signalling acts in postmitotic avian myogenic cells to control MyoD activation. Development. 2001;128:107–116. [Abstract] [Google Scholar]
- Hrabe de Angelis M, McIntyre J, 2nd, Gossler A. Maintenance of somite borders in mice requires the Delta homologue DII1. Nature. 1997;386:717–721. [Abstract] [Google Scholar]
- Hughes SH, Greenhouse JJ, Petropoulos CJ, Sutrave P. Adaptor Plasmids Simplify the Insertion of Foreign DNA into Helper-Independent Retroviral Vectors. Journal of Virology. 1987;61:3004–3012. [Europe PMC free article] [Abstract] [Google Scholar]
- Ikeya M, Takada S. Wnt signaling from the dorsal neural tube is required for the formation of the medial dermomyotome. Development. 1998;125:4969–4976. [Abstract] [Google Scholar]
- Imai T, Tokunaga A, Yoshida T, Hashimoto M, Mikoshiba K, Weinmaster G, Nakafuku M, Okano H. The neural RNA-binding protein musashi1 translationally regulates mammalian numb gene expression by interacting with its mRNA. Mol. and Cell Biol. 2001;21:3888–3900. [Europe PMC free article] [Abstract] [Google Scholar]
- Jostes B, Walther C, Gruss P. The murine paired box gene, Pax7, is expressed specifically during the development of the nervous and muscular system. Mech. of Develop. 1991;33:27–38. [Abstract] [Google Scholar]
- Kahane N, Cinnamon Y, Kalcheim C. The roles of cell migration and myofiber intercalation in patterning formation of the postmitotic myotome. Development. 2002;129:2675–2687. [Abstract] [Google Scholar]
- Kao HY, Ordentlich P, Koyano-Nakagawa N, Tang Z, Downes M, Kintner CR, Evans RM, Kadesch T. A histone deacetylase corepressor complex regulates the Notch signal transduction pathway. Genes Dev. 1998;12:2269–2277. [Europe PMC free article] [Abstract] [Google Scholar]
- Kassar-Duchossoy L, Giacone E, Gayraud-Morel B, Jory A, Gomes D, Tajbakhsh S. Pax3/Pax7 mark a novel population of primitive myogenic cells during development. Genes Dev. 2005;19:1426–1431. [Europe PMC free article] [Abstract] [Google Scholar]
- Kiefer JC, Hauschka SD. Myf-5 is transiently expressed in nonmuscle mesoderm and exhibits dynamic regional changes within the presegmented mesoderm and somites I-IV. Dev Biol. 2001;232:77–90. [Abstract] [Google Scholar]
- Kopan R, Nye JS, Weintraub H. The intracellular domain of mouse Notch: a constitutively activated repressor of myogenesis directed at the basic helix-loop-helix region of MyoD. Development. 1994;120:2385–2396. [Abstract] [Google Scholar]
- Kruger M, Mennerich D, Fees S, Schafer R, Mundlos S, Braun T. Sonic hedgehog is a survival factor for hypaxial muscles during mouse development. Development. 2001;128:743–752. [Abstract] [Google Scholar]
- Kuroda K, Tani S, Tamura K, Minoguchi S, Kurooka H, Honjo T. Delta-induced Notch signaling mediated by RBP-J inhibits MyoD expression and myogenesis. J Biol Chem. 1999;274:7238–7244. [Abstract] [Google Scholar]
- Lin AY, Dechesne CA, Eldridge J, Paterson BM. An avian muscle factor related to MyoD1 activates muscle-specific promoters in nonmuscle cells of different germ-layer origin and in BrdU-treated myoblasts. Genes and Development. 1989;3:986–996. [Abstract] [Google Scholar]
- Linker C, Lesbros C, Stark MR, Marcelle C. Intrinsic signals regulate the initial steps of myogenesis in vertebrates. Development. 2003;130:4797–4807. [Abstract] [Google Scholar]
- Luo B, Aster JC, Hasserjian RP, Kuo F, Sklar J. Isolation and functional analysis of a cDNA for human Jagged2, a gene encoding a ligand for the Notch1 receptor. Mol Cell Biol. 1997;17:6057–6067. [Europe PMC free article] [Abstract] [Google Scholar]
- Marcelle C, Ahlgren S, Bronner-Fraser M. In vivo regulation of somite differentiation and proliferation by Sonic Hedgehog. Dev Biol. 1999;214:277–287. [Abstract] [Google Scholar]
- Marcelle C, Stark MR, Bronner-Fraser M. Coordinate actions of BMPs, Wnts, Shh and noggin mediate patterning of the dorsal somite. Development. 1997;124:3955–3963. [Abstract] [Google Scholar]
- Maroto M, Reshef R, Münsterberg AE, Koester S, Goulding M, Lassar AB. Ectopic Pax-3 activates MyoD and Myf-5 Expression in embryonic mesoderm and neural tissue. Cell. 1997;89:139–148. [Abstract] [Google Scholar]
- McMahon JA, Takada S, Zimmerman LB, Fan CM, Harland RM, McMahon AP. Noggin-mediated antagonism of BMP signaling is required for growth and patterning of the neural tube and somite. Genes Dev. 1998;12:1438–1452. [Europe PMC free article] [Abstract] [Google Scholar]
- Morgan BA, Fekete DM. Manipulating gene expression with replication-competent retroviruses. Methods Cell Biol. 1996;51:185–218. [Abstract] [Google Scholar]
- Mumm JS, Kopan R. Notch signaling: from the outside in. Dev Biol. 2000;228:151–165. [Abstract] [Google Scholar]
- Munsterberg AE, Kitajewski J, Bumcrot DA, McMahon AP, Lassar AB. Combinatorial signaling by Sonic hedgehog and Wnt family members induces myogenic bHLH gene expression in the somite. Genes Dev. 1995;9:2911–2922. [Abstract] [Google Scholar]
- Myat A, Henrique D, Ish-Horowicz D, Lewis J. A chick homologue of Serrate and its relationship with Notch and Delta homologues during central neurogenesis. Dev Biol. 1996;174:233–247. [Abstract] [Google Scholar]
- Nie J, McGill MA, Dermer M, Dho SE, Wolting CD, MC J. LNX functions as a RING type E3 ubiquitin ligase that targets the cell fate determinant Numb for ubiquitin-dependent degradation. EMBO J. 2002;21:93–102. [Europe PMC free article] [Abstract] [Google Scholar]
- Nofziger D, Miyamoto A, Lyons KM, Weinmaster G. Notch signaling imposes two distinct blocks in the differentiation of C2C12 myoblasts. Development. 1999;126:1689–1702. [Abstract] [Google Scholar]
- Oka C, Nakano T, Wakeham A, de la Pompa JL, Mori C, Sakai T, Okazaki S, Kawaichi M, Shiota K, Mak TW, Honjo T. Disruption of the mouse RBP-J kappa gene results in early embryonic death. Development. 1995;121:3291–3301. [Abstract] [Google Scholar]
- Ordahl CP, Berdougo E, Venters SJ, Denetclaw WF., Jr The dermomyotome dorsomedial lip drives growth and morphogenesis of both the primary myotome and dermomyotome epithelium. Development. 2001;128:1731–1744. [Abstract] [Google Scholar]
- Relaix F, Rocancourt D, Mansouri A, Buckingham M. A Pax3/Pax7-dependent population of skeletal muscle progenitor cells. Nature. 2005;435:948–953. [Abstract] [Google Scholar]
- Reshef R, Maroto M, Lassar AB. Regulation of dorsal somitic fates: BMPs and Noggin control the timing and pattern of myogenic regulator expression. Genes Dev. 1998;12:290–303. [Europe PMC free article] [Abstract] [Google Scholar]
- Riddle RD, Johnson RL, Laufer E, Tabin C. Sonic hedgehog mediates the polarizing activity of the ZPA. Cell. 1993;75:1401–1416. [Abstract] [Google Scholar]
- Rudnicki MA, Schnegelsberg PNJ, Stead RH, Braun T, Arnold H, Jaenisch R. MyoD or Myf-5 is Required for the Formation of Skeletal Muscle. Cell. 1993;75:1351–-1359. [Abstract] [Google Scholar]
- Ruiz Gomez M, Bate M. Segregation of myogenic lineages in Drosophila requires numb. Development. 1997;124:4857–4866. [Abstract] [Google Scholar]
- Santolini E, Puri C, Salcini AE, Gagliani MC, Pelicci PG, Tacchetti C, Di Fiore PP. Numb is an endocytic protein. J Cell Biol. 2000;151:1345–1352. [Europe PMC free article] [Abstract] [Google Scholar]
- Sassoon D, Lyons G, Wright W, Lin V, Lassar A, Weintraub H, Buckingham M. Expression of two myogenic regulatory factors, myogenin and MyoD1, during mouse embryogenesis. Nature. 1989;341:303–307. [Abstract] [Google Scholar]
- Shawber C, Nofziger D, Hsieh JJ, Lindsell C, Bogler O, Hayward D, Weinmaster G. Notch signaling inhibits muscle cell differentiation through a CBF1-independent pathway. Development. 1996;122:3765–3773. [Abstract] [Google Scholar]
- Spana EP, Doe CQ. Numb antagonizes Notch signaling to specify sibling neuron cell fates. Neuron. 1996;17:21–26. [Abstract] [Google Scholar]
- Stern CD. Detection of multiple gene products simultaneously by in situ hybridization and immunohistochemistry in whole mounts of avian embryos. Curr Top Dev Biol. 1998;36:223–243. [Abstract] [Google Scholar]
- Stern HM, Brown AMC, Hauschka SD. Myogenesis in paraxial mesoderm: Preferential induction by dorsal neural tube and by cells expressing Wnt-1. Development. 1995;121:3675–3686. [Abstract] [Google Scholar]
- Sun J, Kamei CN, Layne MD, Jain MK, Liao JK, Lee ME, Chin MT. Regulation of myogenic terminal differentiation by the hairy-related transcription factor CHF2. J Biol Chem. 2001;276:18591–18596. [Abstract] [Google Scholar]
- Susini L, Passer BJ, Amzallag-Elbaz N, Juven-Gershon T, Prieur S, Privat N, Tuynder M, Gendron M-C, Israel A, Amson R, Oren M, Telerman A. Siah-1 binds and regulates the function of Numb. Proc. Natl. Acad. Sci. USA. 2001;98:15067–15072. [Europe PMC free article] [Abstract] [Google Scholar]
- Tajbakhsh S, Borello U, Vivarelli E, Kelly R, Papkoff J, Duprez D, Buckingham M, Cossu G. Differential activation of Myf5 and MyoD by different Wnts in explants of mouse paraxial mesoderm and the later activation of myogenesis in the absence of Myf5. Development. 1998;125:4155–4162. [Abstract] [Google Scholar]
- Tajbakhsh S, Buckingham M. The birth of muscle progenitor cells in the mouse: spatiotemporal considerations. Curr Top Dev Biol. 2000;48:225–268. [Abstract] [Google Scholar]
- Tajbakhsh S, Rocancourt D, Cossu G, Buckingham M. Redefining the genetic hierarchies controlling skeletal myogenesis: Pax-3 and myf-5 act upstream of MyoD. Cell. 1997;89:127–138. [Abstract] [Google Scholar]
- Teboul L, Summerbell D, Rigby PW. The initial somitic phase of Myf5 expression requires neither Shh signaling nor Gli regulation. Genes Dev. 2003;17:2870–2874. [Europe PMC free article] [Abstract] [Google Scholar]
- Teillet M, Watanabe Y, Jeffs P, Duprez D, Lapointe F, Le Douarin NM. Sonic hedgehog is required for survival of both myogenic and chondrogenic somitic lineages. Development. 1998;125:2019–2030. [Abstract] [Google Scholar]
- Tonegawa A, Takahashi Y. Somitogenesis controlled by Noggin. Dev Biol. 1998;202:172–182. [Abstract] [Google Scholar]
- Venters SJ, Ordahl CP. Persistent myogenic capacity of the dermomyotome dorsomedial lip and restriction of myogenic competence. Development. 2002;129:3873–3885. [Abstract] [Google Scholar]
- Venters SJ, Ordahl CP. Asymmetric cell divisions are concentrated in the dermomyotome dorsomedial lip during epaxial primary myotome morphogenesis. Anat Embryol (Berl) 2005;209:449–460. [Abstract] [Google Scholar]
- Wakamatsu Y, Maynard TM, Jones SU, Weston JA. NUMB localizes in the basal cortex of mitotic avian neuroepithelial cells and modulates neuronal differentiation by binding to NOTCH-1. Neuron. 1999;23:71–81. [Abstract] [Google Scholar]
- Weintraub H, Tapscott SJ, Davis RL, Thayer MJ, Adam MA, Lassar AB, Miller AD. Activation of muscle-specific genes in pigment, nerve, fat, liver, and fibroblast cell lines by forced expression of MyoD. Proc. Natl. Acad. Sci. USA. 1989;86:5434–5438. [Europe PMC free article] [Abstract] [Google Scholar]
- Williams R, Lendahl U, Lardelli M. Complementary and combinatorial patterns of Notch gene family expression during early mouse development. Mech. of Develop. 1995;53:357–368. [Abstract] [Google Scholar]
- Wilson-Rawls J, Molkentin JD, Black BL, Olson EN. Activated notch inhibits myogenic activity of the MADS-Box transcription factor myocyte enhancer factor 2C. Mol Cell Biol. 1999;19:2853–2862. [Europe PMC free article] [Abstract] [Google Scholar]
- Wittenberger T, Steinbach OC, Authaler A, Kopan R, Rupp RA. MyoD stimulates delta-1 transcription and triggers notch signaling in the Xenopus gastrula. EMBO J. 1999;18:1915–1922. [Europe PMC free article] [Abstract] [Google Scholar]
- Zhong W, Jiang MM, Weinmaster G, Jan LY, Jan YN. Differential expression of mammalian Numb, Numblike and Notch1 suggests distinct roles during mouse cortical neurogenesis. Development. 1997;124:1887–1897. [Abstract] [Google Scholar]
Full text links
Read article at publisher's site: https://doi.org/10.1002/dvdy.20672
Read article for free, from open access legal sources, via Unpaywall:
https://europepmc.org/articles/pmc2561193?pdf=render
Citations & impact
Impact metrics
Citations of article over time
Alternative metrics
Article citations
The Multitasker Protein: A Look at the Multiple Capabilities of NUMB.
Cells, 12(2):333, 15 Jan 2023
Cited by: 2 articles | PMID: 36672267 | PMCID: PMC9856935
Review Free full text in Europe PMC
NUMB as a Therapeutic Target for Melanoma.
J Invest Dermatol, 142(7):1882-1892.e5, 07 Dec 2021
Cited by: 5 articles | PMID: 34883044 | PMCID: PMC9704357
Angelica Sinensis Polysaccharide Prevents Hematopoietic Stem Cells Senescence in D-Galactose-Induced Aging Mouse Model.
Stem Cells Int, 2017:3508907, 11 Apr 2017
Cited by: 22 articles | PMID: 28491095 | PMCID: PMC5405396
WNT Signaling in Lung Aging and Disease.
Ann Am Thorac Soc, 13 Suppl 5:S411-S416, 01 Dec 2016
Cited by: 29 articles | PMID: 28005418
Review
Numb-deficient satellite cells have regeneration and proliferation defects.
Proc Natl Acad Sci U S A, 110(46):18549-18554, 29 Oct 2013
Cited by: 28 articles | PMID: 24170859 | PMCID: PMC3831958
Go to all (32) article citations
Data
Similar Articles
To arrive at the top five similar articles we use a word-weighted algorithm to compare words from the Title and Abstract of each citation.
Regulation of dorsal somitic cell fates: BMPs and Noggin control the timing and pattern of myogenic regulator expression.
Genes Dev, 12(3):290-303, 01 Feb 1998
Cited by: 141 articles | PMID: 9450925 | PMCID: PMC316485
Compartmentalization of the somite and myogenesis in chick embryos are influenced by wnt expression.
Dev Biol, 228(1):86-94, 01 Dec 2000
Cited by: 46 articles | PMID: 11087628
Expression of (beta)-catenin in the developing chick myotome is regulated by myogenic signals.
Development, 127(19):4105-4113, 01 Oct 2000
Cited by: 44 articles | PMID: 10976043
Formation and differentiation of avian somite derivatives.
Adv Exp Med Biol, 638:1-41, 01 Jan 2008
Cited by: 20 articles | PMID: 21038768
Review
Funding
Funders who supported this work.
NIGMS NIH HHS (3)
Grant ID: GM054879
Grant ID: R01 GM054879-08
Grant ID: R01 GM054879





