Abstract
Free full text

MAIT cells are activated during human viral infections
Abstract
Mucosal-associated invariant T (MAIT) cells are abundant in humans and recognize bacterial ligands. Here, we demonstrate that MAIT cells are also activated during human viral infections in vivo. MAIT cells activation was observed during infection with dengue virus, hepatitis C virus and influenza virus. This activation—driving cytokine release and Granzyme B upregulation—is TCR-independent but dependent on IL-18 in synergy with IL-12, IL-15 and/or interferon-α/β. IL-18 levels and MAIT cell activation correlate with disease severity in acute dengue infection. Furthermore, HCV treatment with interferon-α leads to specific MAIT cell activation in vivo in parallel with an enhanced therapeutic response. Moreover, TCR-independent activation of MAIT cells leads to a reduction of HCV replication in vitro mediated by IFN-γ. Together these data demonstrate MAIT cells are activated following viral infections, and suggest a potential role in both host defence and immunopathology.
Mucosal-associated invariant T (MAIT) cells are an abundant and distinctive T cell subset first identified by Porcelli et al.1 in 1993 in humans on the basis of the invariant V-alpha chain (Vα7.2-Jα33) and later described in mice2. MAIT cells comprise ~5–10% of T-cells within the periphery and are found enriched in both the gut and in the liver—where in humans they may represent between ~12% and 50% of T-cells3,4,5,6. In both mouse and man, MAIT cells are restricted to a bacterial riboflavin synthesis pathway-derived ligand presented by the conserved MHC-class I like molecule MR1 (refs 7, 8). In vivo challenge models using riboflavin synthesizing bacteria, such as Klebsiella species and mycobacteria, have confirmed an anti-bacterial role for MAIT cells9,10. MR1 tetramers loaded with the ligand rRL-6-CH2OH have allowed the specific detection of human and murine MAIT cells11.
In recent years, a number of groups have elucidated the distinct phenotypic and functional profile of MAIT cells, characterized by the high expression of the C-type lectin CD161 (KLRB1 or NKR-P1A), and the capacity to secrete multiple cytokines, including IL-17, interferon (IFN)-γ and TNF-α12,13,14,15,16. The distinctive phenotype of MAIT cells appears to be driven by two key transcription factors, RORγt and PLZF3,6,13,17. RORγt expression is linked to development of type 17 function and expression of surface receptors such as IL23R and CCR6 (refs 5, 18). This is consistent with mucosal defence and anti-bacterial functions and also consistent with the bacterial specificity of the receptor. PLZF is critical for development of invariant natural killer T cell (iNKT) cells and may be responsible for a distinct set of ‘innate' phenotypic features, including marked upregulation of the pro-inflammatory cytokine receptors IL-18R and IL-12R19,20. This dual transcriptional drive suggests that MAIT cells may possess multiple parallel functionalities or modes of activation.
Given the specificity of the T-cell receptor (TCR), it appears that activation of MAIT cells in vivo is driven by responsiveness to bacteria (and some yeasts)21. However, given their ‘innate' phenotype, broad range of effector functions, and tissue distribution, we addressed the question of whether they may also have evolved to respond to viral infections in vivo. If so, this would markedly widen the role of MAIT cells in human infection.
Here, we addressed MAIT cell activation in response to a set of major human viral pathogens both in vivo and in vitro. In vivo, MAIT cells are activated in response to dengue virus (DENV), hepatitis C virus (HCV) and influenza virus. In vitro, DENV, HCV and influenza virus readily trigger MAIT cell populations in an IL-18 dependent manner, in concert with other virally-driven pro-inflammatory cytokines, including IFN-α/β. Activation of MAIT cells in acute dengue correlated both with disease outcome and with IL-18 signals. Similarly, in vivo activation of MAIT cells during HCV therapy correlated with specific addition of IFN-α during therapy. Taken together, these data strongly implicate a role for MAIT cells in response to major virus infections of man and provide a mechanism for their virus-responsive nature. Overall, this significantly expands the pathogen response repertoire of this abundant human T-cell subset.
Results
MAIT cell activation during acute viral infections in vivo
Whether human MAIT cells respond during acute viral infections remains unclear. To address this we analysed MAIT cell activation in DENV and influenza virus infection. In this study, MAIT cells were defined as CD161++Vα7.2+ CD8+CD4−CD3+ T cells. DENV infection presents with a rapid onset of a febrile illness, which lasts around a week. After the virus has been cleared and fever drops, patients follow two distinct courses: an uncomplicated resolution (dengue fever; DF) or a severe course associated with organ dysfunction and haemorrhage (dengue haemorrhagic fever; DHF). The determinants driving outcome are still poorly understood. However, an aberrant immune response, around the day of defervescence, is thought to have an important role in the pathogenesis. We analysed MAIT cells in a longitudinal study of peripheral blood mononuclear cells (PBMC) from a cohort of patients with DHF or DF. Blood samples were collected at different time points during the acute phase of infection and dates were named in relation to the day fever subsided (day of defervescence, day 0). Samples taken 10 or more days after the day of defervescence (day >10) were considered convalescent. We found a small but significant reduction in MAIT cell frequencies between the acute phase (day 0) and the convalescent phase (day >10) of infection (Fig. 1a,b), but no differences in MAIT cell frequency between DHF and DF patients (Fig. 1c).
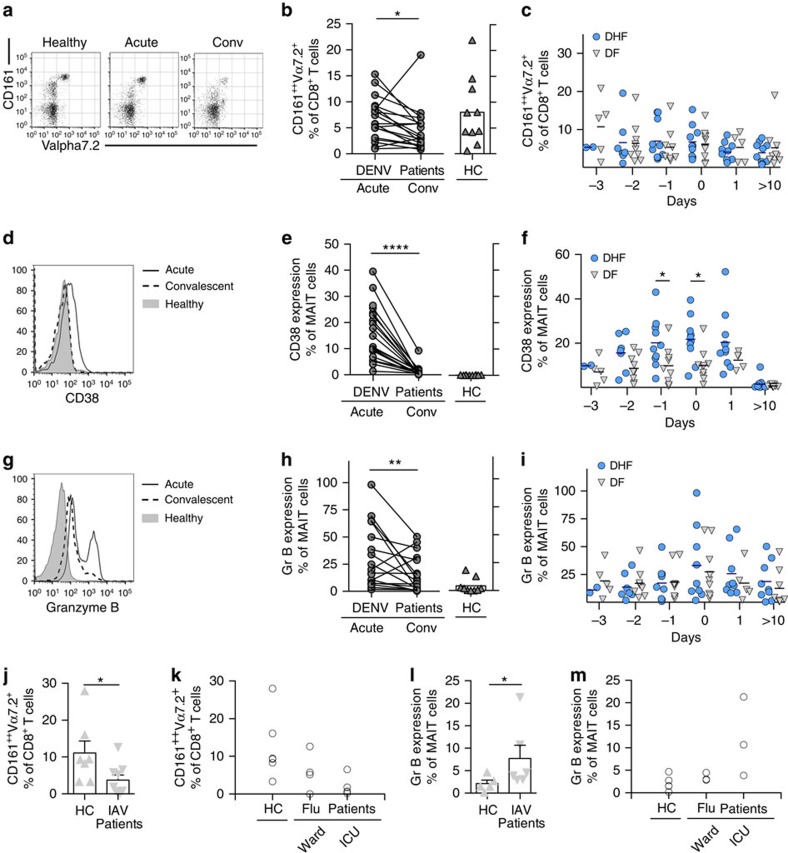
PBMC's from healthy controls (HC, n=5–10), patients suffering from severe dengue (DHF, n=2–10), dengue (DF, n=4–10) or acute, hospitalized patients infected with influenza A virus (n=7–9) were analysed by flow cytometry by gating on live CD3+CD8+CD161++Vα7.2+ (MAIT) cells. (a,d,g) Representative flow cytometry plots. (b,e,h) Comparison between acute (day 0) and convalescent phase (day >10) of infection. (c,f,i) Comparison between DHF and DF patients. (j–m) Acute, hospitalized patients infected with influenza virus. (b,c,j,k) MAIT cell frequency as a proportion of the CD8+ T cell population. (e,f) Percentage of CD38 expression by MAIT cells. (h,i,l,m) Percentage of Granzyme B expression by MAIT cells. Statistical significance was determined with a Wilcoxon matched-paired test (b,e,h) or Mann–Whitney test (j,k). Bars represent means±s.e.m. ns>0.05, *P 0.05, **P≤0.01, ***P≤0.001, ****P≤0.0001. conv, convalescent; Gr B, Granzyme B; HC, healthy control; ICU, intensive care unit.
Analysis of CD38 revealed marked in vivo activation of MAIT cells (Fig. 1d,e), which increased over the course of infection and peaked at a critical moment for DENV infected patients—the day of defervescence. Interestingly, patients who developed the severe form of dengue had higher levels of MAIT cell activation as judged by CD38 expression compared to DF patients over the course of acute infection (Fig. 1f). MAIT cell activation resolved to healthy control levels in the convalescent sample (Fig. 1d,e).
Granzyme B expression was also assessed due to its tight regulation in MAIT cells, and its absence in cells from healthy donors3,22. Furthermore, upregulation of Granzyme B is associated with the acquisition of cytolytic function by MAIT cells22,23. We therefore analysed Granzyme B function in acute dengue and found this followed the same time course as that of CD38 (Fig. 1g–i).
Given their role in mucosal defence, we next addressed the activation of MAIT cells in response to influenza virus, a virus with a segmented genome of negative-sense RNA. Again, patients with acute, severe influenza virus infection had reduced MAIT cell frequencies and an increase in Granzyme B expression on MAIT cells (Fig. 1j–m).
Taken together, our results indicate substantial triggering of MAIT cells in vivo during acute viral infection.
MAIT cell activation during chronic viral infection in vivo
MAIT cells are found at high frequencies within the liver in both healthy and diseased states3,5,13,24. Therefore, we analysed MAIT cell activation during chronic infection with HCV, like DENV, a member of the Flaviviridae family of positive-sense RNA viruses. We examined MAIT cell frequency and phenotype in the PBMC of patients with persistent and resolved HCV infection (spontaneously or after therapy). In all HCV patients, regardless of status, we observed a reduction in MAIT cell frequencies compared to healthy controls (Fig. 2a). However, we only observed upregulation of Granzyme B in patients with prolonged HCV infection (including those who had subsequently responded to antiviral therapy; Fig. 2b,c), and not in those patients with prior short-lived viremia at a distant time-point associated with acute resolving infection (thus, more akin to convalescent DENV infection). Our results indicate substantial activation of MAIT cells in vivo both during acute and chronic viral infections.
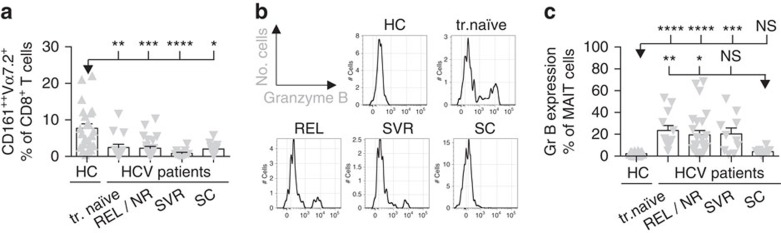
PBMC's from healthy controls (n=20–23) or patients (n=12–25) with persistent (treatment naive, REL, NR) and resolved HCV infection (SVR, SC) were analysed by flow cytometry by gating on live CD3+CD8+CD161++Vα7.2+ (MAIT) cells. (a) MAIT cell frequency as a proportion of the CD8+ T cells. (b,c) Granzyme B expression by MAIT cells. (b) Representative flow cytometry plots. Bars represent means±s.e.m. Statistical significance was determined with the Kruskal–Wallis test followed by the Dunns' test. ns>0.05, *P 0.05, **P≤0.01, ***P≤0.001, ****P≤0.0001. HC, healthy control; Gr B, Granzyme B; REL/NR, relapse/non-response; SC, spontaneous clearance; SVR, sustained virological response; tr., treatment.
Viral MAIT cell activation in vitro
Having observed MAIT cell activation in vivo during acute and chronic viral infections, we next established in vitro models for viral infections using PBMCs or human CD8+ T cells, co-incubated with infected or virus-treated dendritic cells (DCs) or macrophages as antigen presenting cells (APC).
MAIT cells were readily and specifically activated in response to DENV-treated APCs (multiplicity of infection (MOI)=1), as indicated by production of IFN-γ and Granzyme B (Fig. 3a,b), as well as, expression of CD38, with minimal production of TNF-α (Supplementary Fig. 1a–c). Activation was dependent on the presence of APCs as DENV alone did not activate MAIT cells (data not shown).
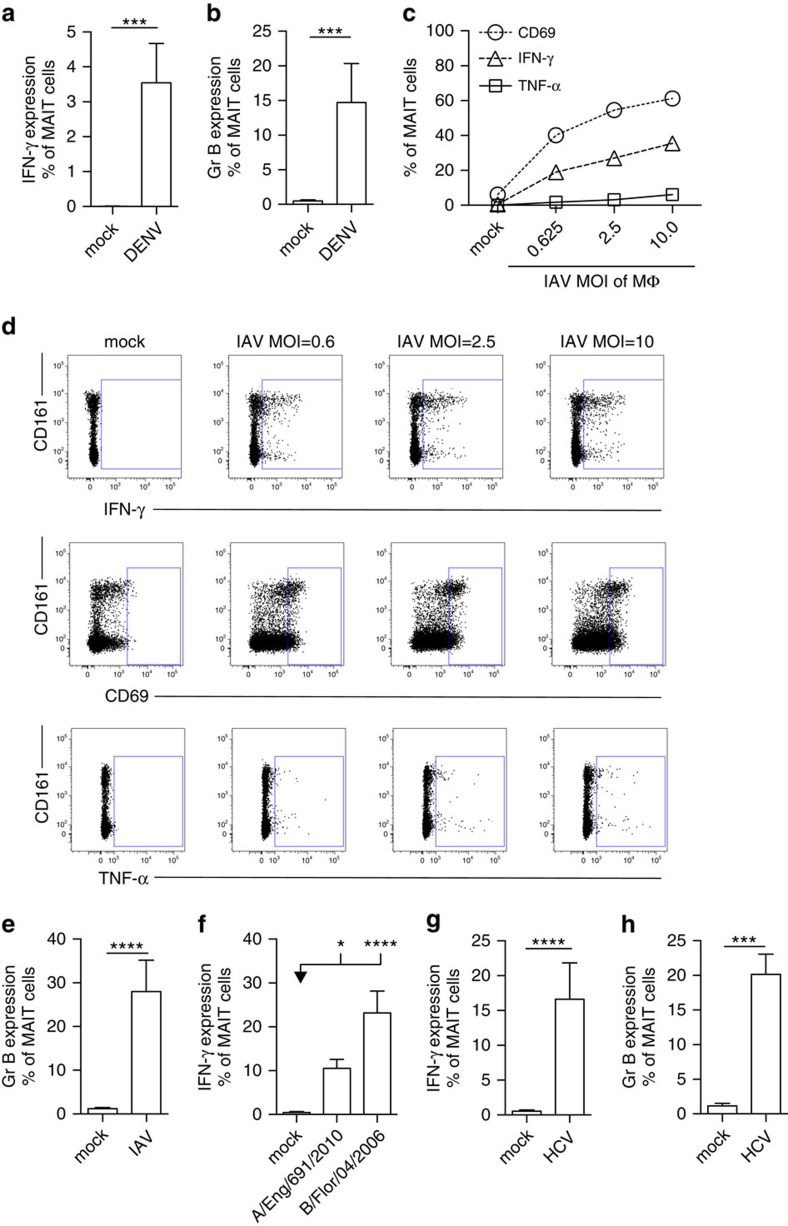
MAIT cells from healthy individuals were analysed by flow cytometry, gated on live CD3+CD161++Vα7.2+ cells. (a,b) PBMC's (n=7) were co-cultured with autologous monocyte-derived DC's exposed to DENV (MOI=1) as described in ‘Methods'. (c–e) CD8+ T cells isolated from PBMC's (n=11–12) were co-cultured with IAV-exposed macrophages (MOI=1) as described in ‘Methods', unless indicated otherwise. (f) CD8+ T cells isolated from PBMC's (n=11–12) were co-cultured with macrophages exposed to the clinical H3N2 influenza A strain (A/England/691/2010 (n=7)) or influenza B (B/Florida/04/2006 (n=8)) (MOI=1) as described in ‘Methods'. (g,h) CD8+ T cells isolated from PBMC's (n=7–12) co-cultured with macrophages exposed to HCV (MOI=1) as described in ‘Methods'. Proportion of MAIT cells producing IFN-γ (a,c,d,f,g), TNF-α (c,d), CD69 (c,d) or Granzyme B (b,e,h). (d) Representative flow cytometry plots. All data are representative from at least two independent experiments. Bars represent means±s.e.m. Statistical significance was determined with the Kruskal–Wallis test followed by the Dunns' test (f) or the Mann–Whitney test (a,b,e,g,h). ns>0.05, *P 0.05, ***P≤0.001, ****P≤0.0001. Eng, England; Flor, Florida; HC, healthy control; Gr B, Granzyme B; MΦ, macrophage; REL/NR, relapse/non-response; SC, spontaneous clearance; SVR, sustained virological response; tr., treatment.
We next examined activation of MAIT cells in vitro in response to influenza virus. APCs incubated with influenza virus (MOI=1) promoted strong MAIT cell activation, inducing upregulation of CD69 and IFN-γ (with minimal TNF-α secretion), in a dose-dependent manner (Fig. 3c,d). Also, Granzyme B expression was induced (Fig. 3e). This activation was not restricted to laboratory-adapted influenza virus strains, but also was seen in response to recently-circulating strains of influenza A and B (Fig. 3f). Interestingly, MAIT cell activation was also readily induced using PBMCs instead of purified APCs (Supplementary Fig. 1d). Moreover, influenza virus exposure of purified monocyte, macrophage and DC populations all led to MAIT cell activation (Supplementary Fig. 1e).
Finally, we tested the impact of HCV on MAIT cells in vitro. MAIT cells expressed IFN-γ and Granzyme B (Fig. 3g,h), as well as CD69 in response APC's treated with HCV (MOI=1), in a dose-dependent and specific manner (Supplementary Fig. 1f–i).
Taken together, we demonstrate that MAIT cells are activated in vitro in response to DENV, influenza virus and HCV-exposed APCs.
Viral MAIT cell activation is dependent on cytokines
Next, we investigated the mechanisms of MAIT cells activation in response to viruses. The ligand presented by MR1 is derived from the riboflavin synthesis pathway, which is not found in host cells or viruses7. Consistent with this, using the models described above, we confirmed by antibody blocking of MR1 that MAIT cell activation was TCR-independent for all the viruses tested (Fig. 4a–c).
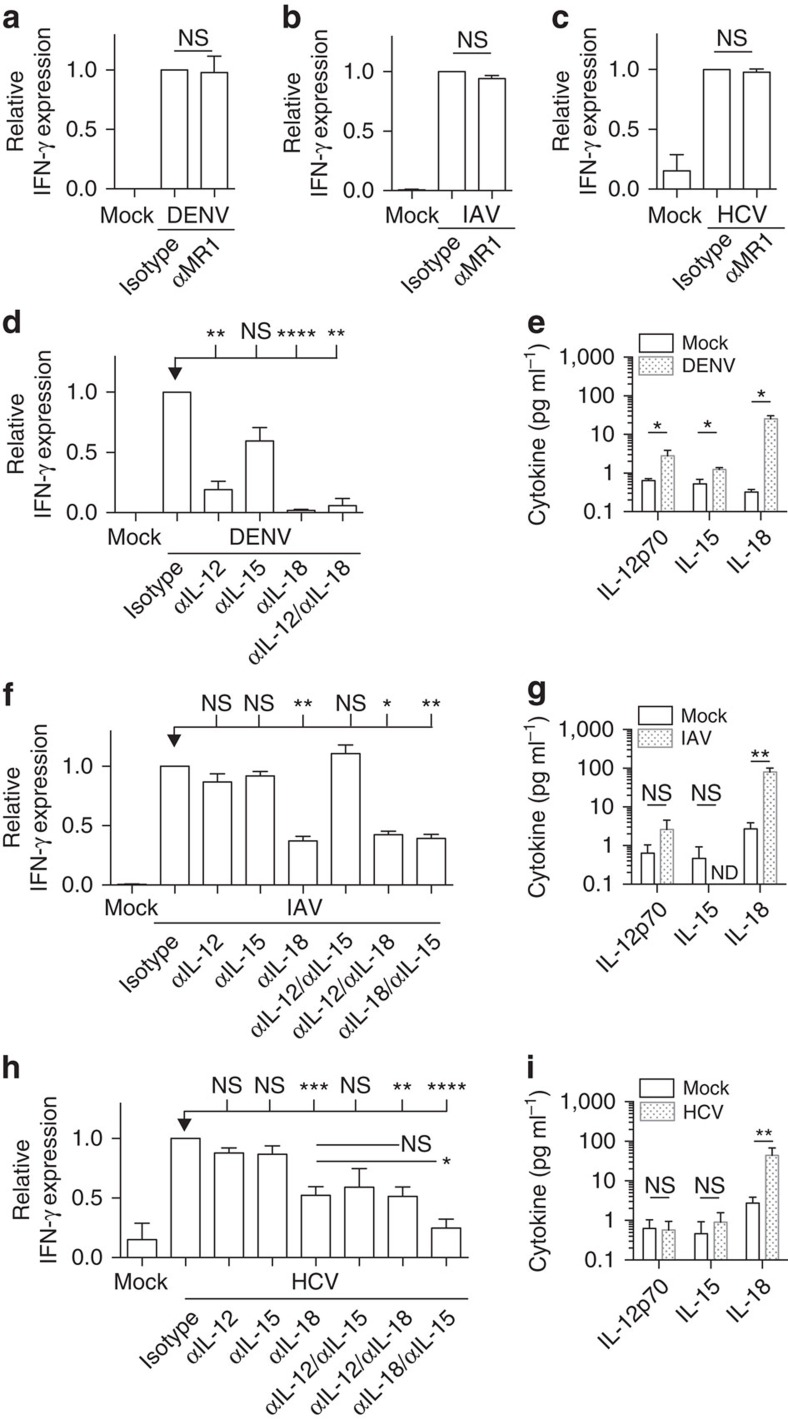
(a–d,f,h) Isotype control, anti-MR1, anti-IL-12, anti-IL-15 and/or anti-IL-18 antibodies were added to a co-culture exposed to DENV (a,d, n=3–6), IAV (b,f, n=5–29) or HCV (c,h, n=7–16). IFN-γ expression by MAIT cells (gated on live CD3+CD8+CD161++Vα7.2+ cells) was analysed by flow cytometry and is shown relative to the isotype control. (e,g,i) IL-12p70, IL-15 and IL-18 levels secreted by virus-exposed APC's. (e) DENV-exposed DC's (n=4) at 42 h. (g) IAV-exposed macrophages (n=7) at 48
h. (g) IAV-exposed macrophages (n=7) at 48 h. (i) HCV-exposed macrophages (n=7) at 48
h. (i) HCV-exposed macrophages (n=7) at 48 h. Data are representative from at least two independent experiments. Bars represent means±s.e.m. Statistical significance was determined with the Mann–Whitney test (a–c,e,g,i) or the Kruskal–Wallis test followed by the Dunns' test or the Mann–Whitney test (d,f,h). ns>0.05, *P 0.05, ***P≤0.001, ****P≤0.0001. ND, not detected.
h. Data are representative from at least two independent experiments. Bars represent means±s.e.m. Statistical significance was determined with the Mann–Whitney test (a–c,e,g,i) or the Kruskal–Wallis test followed by the Dunns' test or the Mann–Whitney test (d,f,h). ns>0.05, *P 0.05, ***P≤0.001, ****P≤0.0001. ND, not detected.
As activation was TCR-independent, we explored triggering of MAIT cells by cytokines. Previously, we have shown that TLR8 is capable of inducing IFN-γ expression in MAIT cells via IL-12 and IL-18 (ref. 24). In addition, IL-15 can specifically activate distinct functions of MAIT cells in synergy with IL-12 and/or IL-18, in a dose-dependent manner (Supplementary Figs 2 and 3)25. We extended this finding by exploring responses to a range of TLR ligands in PBMCs and found endosomal TLR3 was also a potent activator (Supplementary Fig. 4). As with TLR8, TLR3 induced MAIT cell activation via IL-18 and IL-12 and not MR112,13. TLR sensing by APC's can occur in the absence of viral replication26,27. To assess the requirement of viral replication for MAIT cell activation, we used ultraviolet-irradiation of the viruses, which prevents transcription or replication. Ultraviolet-irradiated DENV was no longer able to activate MAIT cells (Supplementary Fig. 5a). In contrast, ultraviolet-irradiated HCV and influenza virus were still able to activate MAIT cells, although less efficiently compared to untreated virus (Supplementary Fig. 5b,c). Accordingly, DENV productively infects APCs, whereas productive influenza virus and HCV infection is limited in both primary and stem cell-derived human APCs (Supplementary Fig. 5d–f)28,29,30. Furthermore, the level of DENV infection correlated with MAIT cell IFN-γ expression (Supplementary Fig. 5e).
We next assessed blockade of IL-12, IL-18 and IL-15 in the in vitro virus models. Figure 4d reveals that IL-12 and IL-18 had the most significant impact on DENV-dependent MAIT cell activation using DC's, with anti-IL-18 blockade alone leading to complete suppression of IFN-γ expression. IL-18 was the most abundant cytokine induced in DC's in response to DENV (Fig. 4e).
Anti-IL-18 also inhibited activation of MAIT cells induced by influenza virus, a result observed using purified APCs (Fig. 4f) or PBMC's (Supplementary Fig. 6a). Blockade of IL-12 and IL-15 alone or in combination had a limited impact in influenza virus-induced MAIT cell activation using macrophages (Fig. 4f). Accordingly, IL-18, but not IL-12 or IL-15 was secreted by macrophages after incubation with influenza virus (Fig. 4g).
Similar to DENV and influenza virus, IL-18 had a dominant role in HCV-induced MAIT cell activation (Fig. 4h). Addition of further blockade with anti-IL-15 was required to achieve maximal suppression. We assessed expression of IL-12, IL-15 and IL-18 on macrophages after incubation with HCV (Fig. 4i). No IL-15 protein was detected, yet IL-15R (responsible for IL-15 trans-presentation31) was upregulated by macrophages in response to HCV, as measured by quantitative PCR (Supplementary Fig. 6b).
Overall, these experiments indicate a mechanism by which viruses can activate MAIT cells, dependent on viral sensing and cytokines, with a key role for IL-18, and independent of MR1 and TCR.
Virally-triggered IL-18 correlates with MAIT cell activation
Having identified IL-18 as an important cytokine for viral MAIT cell activation in vitro, we further explored its relevance in vivo. In DENV infection, IL-18 levels in plasma were markedly increased during the acute phase compared to the convalescent phase of infection and healthy controls (Fig. 5a). In addition, patients suffering from the severe form of dengue (DHF) had significantly higher levels of IL-18 over the course of acute infection compared to patient with DF (Fig. 5b). Interestingly, expression of the IL-18 receptor (IL-18R) on MAIT cells was upregulated during the acute phase of infection and resolved to healthy control levels in the convalescent samples (Fig. 5c). As observed for IL-18, patients with DHF revealed higher expression of IL-18R on MAIT cells compared to patient with DF (Fig. 5d,e). Thus, levels of IL-18 in plasma and expression of IL-18R on MAIT cells in vivo are upregulated during the acute phase of infection, especially in those patients suffering from DHF. These findings show an association with increased activation of MAIT cells and disease severity in dengue patients (Figs 1f and and5f).5f). These results are not restricted to DENV infection since IL-18 protein (Fig. 5g) and mRNA (Fig. 5h) were found readily in liver tissues from patients with HCV infection.
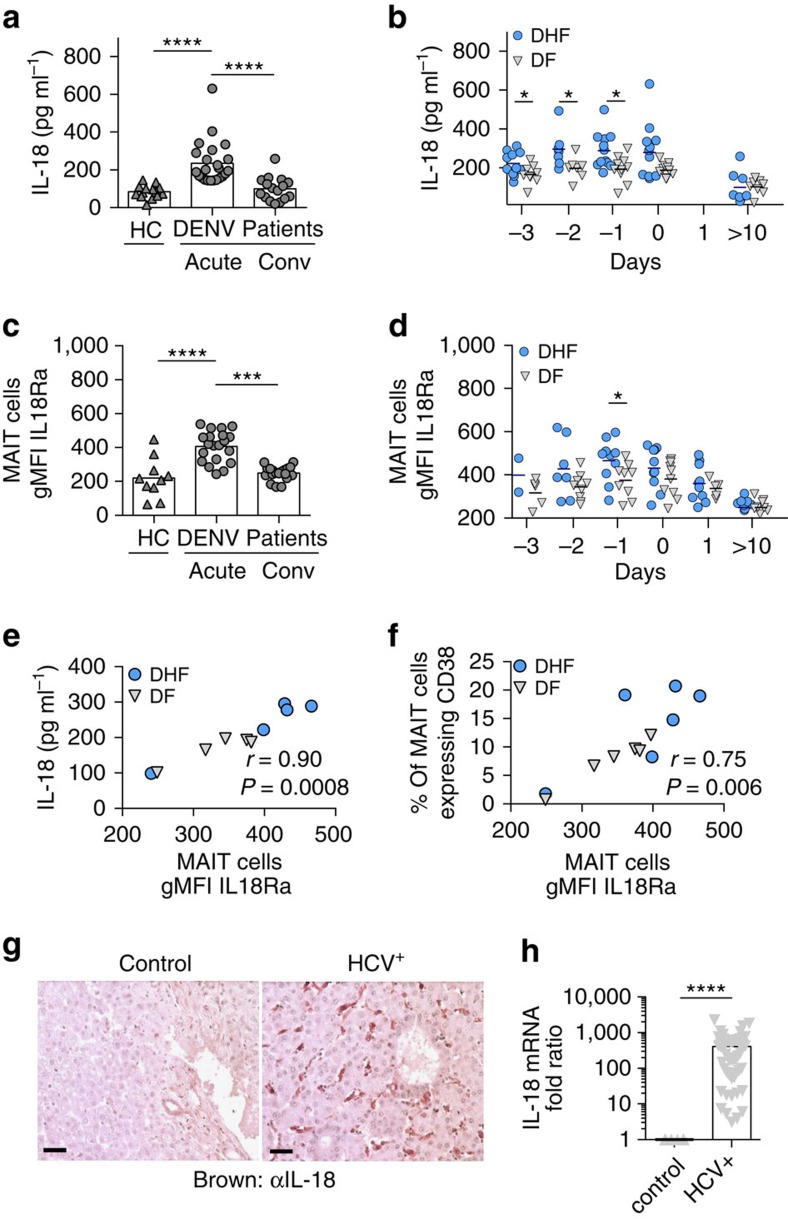
(a–d) PBMC's from healthy controls (n=10) or patients suffering from severe dengue (DHF, n=2–12) or dengue (DF, n=4–12) were analysed. (a,b) Plasma levels of IL-18. (c,d) IL-18Ra expression on MAIT cells. (a,c) Comparison between acute (day 0) and convalescent (conv) phase (day >10) of infection. (b,d) Comparison between DHF and DF patients. Correlation of plasma levels of IL-18 (e) or MAIT cell CD38 expression (f) against MAIT cell IL-18Ra expression by the Spearman-rank correlation test. (g) Paraffin-embedded human liver tissues sections from HCV or control patients were stained for IL-18 (brown-coloured). Scale bar, 10 mm. (h) IL-18 mRNA expression in liver biopsies from HCV patients (n=55) relative to uninfected control liver samples is shown (n=6). Bars represent means±s.e.m. Statistical significance was determined with the Mann–Whitney test (b,d,h) or the Wilcoxon matched-paired test (a,c). ns>0.05, *P 0.05, ***P≤0.001, ****P≤0.0001. conv, convalescent; HC, healthy control.
mm. (h) IL-18 mRNA expression in liver biopsies from HCV patients (n=55) relative to uninfected control liver samples is shown (n=6). Bars represent means±s.e.m. Statistical significance was determined with the Mann–Whitney test (b,d,h) or the Wilcoxon matched-paired test (a,c). ns>0.05, *P 0.05, ***P≤0.001, ****P≤0.0001. conv, convalescent; HC, healthy control.
Overall, these data indicate that IL-18 is not only a dominant factor during in vitro viral MAIT cell activation, but also has an important role in vivo.
MAIT cells respond to type I IFNs in vitro and in vivo
Type I IFNs are also important cytokines known for their strong antiviral activity. Previous reports have determined a crucial role of IFN-α/β during DENV32, influenza33 and HCV34 infections in limiting viral replication and disease severity. We therefore addressed whether IFN-α/β can also influence MAIT cell activation in vitro and in vivo.
First, we tested whether IFN-α/β could trigger MAIT cells alone or in combination. We found IFN-γ secretion by MAIT cells in response to IFN-α/β in combination with IL-12 and IL-18, but not IL-15 (Fig. 6a), in a dose-dependent manner (Supplementary Fig. 7). Next, we confirmed that these TCR-independent responses to cytokines are shared with other CD161++ T-cell subsets, but not CD161− T-cell populations. CD161++CD8+ T cells that lack expression of the Vα7.2Jα33 TCR (Supplementary Fig. 8a), CD161++ Vα7.2Jα33+ CD4 T cells (Supplementary Fig. 8b) and CD4−CD8− T cells (Supplementary Fig. 8c) could all be triggered in response to cytokines, unlike CD161− T-cell populations (Supplementary Fig. 8d). This is in agreement with our previous findings which suggest that CD161-expressing lymphocytes display a shared innate response to cytokines16.
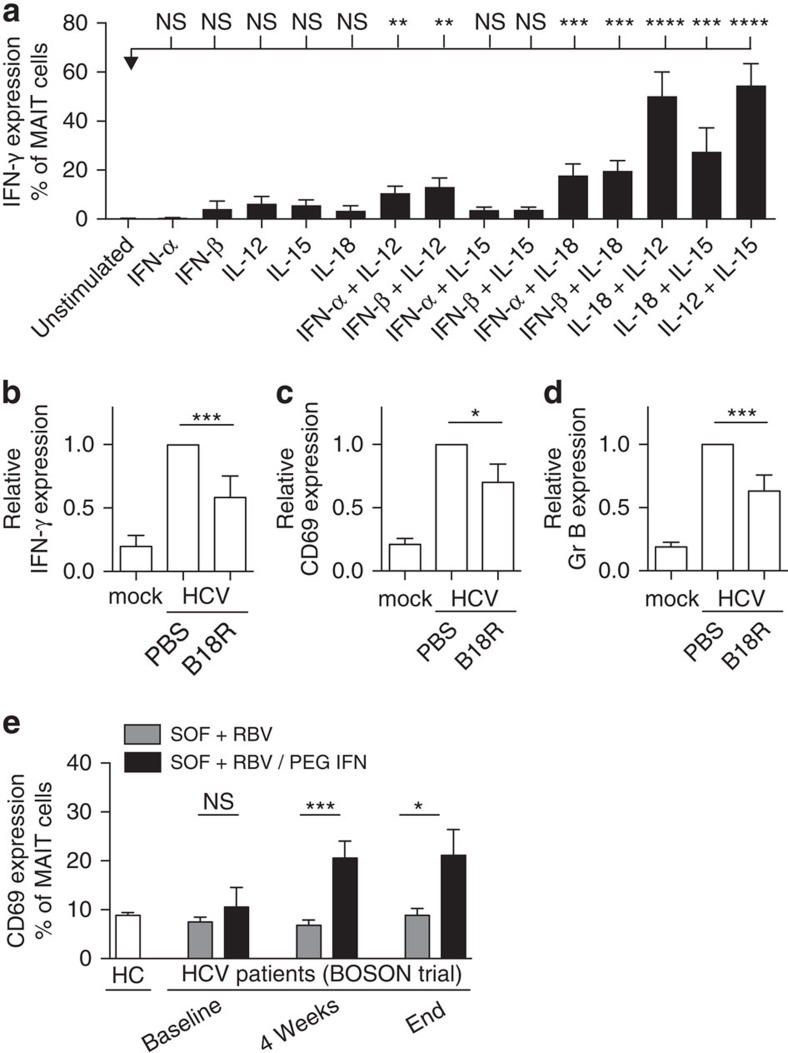
(a) PBMC's from healthy individuals were directly stimulated for 24 h with IFN-α, IFN-β, IL-12, IL-15, IL-18 or indicated combinations thereof. IFN-γ expression by MAIT cells (gated on live CD3+CD8+CD161++Vα7.2+ cells) was analysed by flow cytometry. (b–d) B18R (IFN-α/β neutralizing protein) or PBS control were added to the co-culture (n=6–11). IFN-γ (b), CD69 (c) and Granzyme B (d) expression is shown relative to the control. Data are representative from at least two independent experiments. (e) PBMC's from healthy controls or HCV patients at baseline, during or end of treatment with either SOF+RBV or SOF+RBV/PEG-IFN were analysed by flow cytometry. CD69 expression on MAIT cells (gated on live CD3+CD8+CD161++Vα7.2+ cells) was measured. Bars represent means±s.e.m. Statistical significance was determined with the Kruskal–Wallis test followed by the Dunns' test (a) or the Mann–Whitney test (b–e). ns>0.05, *P 0.05, ***P≤0.001, ****P≤0.0001. HC, healthy controls.
h with IFN-α, IFN-β, IL-12, IL-15, IL-18 or indicated combinations thereof. IFN-γ expression by MAIT cells (gated on live CD3+CD8+CD161++Vα7.2+ cells) was analysed by flow cytometry. (b–d) B18R (IFN-α/β neutralizing protein) or PBS control were added to the co-culture (n=6–11). IFN-γ (b), CD69 (c) and Granzyme B (d) expression is shown relative to the control. Data are representative from at least two independent experiments. (e) PBMC's from healthy controls or HCV patients at baseline, during or end of treatment with either SOF+RBV or SOF+RBV/PEG-IFN were analysed by flow cytometry. CD69 expression on MAIT cells (gated on live CD3+CD8+CD161++Vα7.2+ cells) was measured. Bars represent means±s.e.m. Statistical significance was determined with the Kruskal–Wallis test followed by the Dunns' test (a) or the Mann–Whitney test (b–e). ns>0.05, *P 0.05, ***P≤0.001, ****P≤0.0001. HC, healthy controls.
To further explore the role of type I IFNs during viral MAIT cell triggering, we used a vaccinia virus-encoded, soluble type I IFN receptor (B18R)35. Macrophages treated with HCV in vitro were incubated in the presence of B18R and T cells overnight. Blockade of type I IFNs by B18R inhibited MAIT cell IFN-γ secretion (Fig. 6b), CD69 expression (Fig. 6c) and Granzyme B upregulation (Fig. 6d).
IFN-α has been an important component of HCV treatment. This gave us an opportunity to explore whether type I IFNs could contribute to MAIT cell activation in vivo. We measured the activation marker CD69 on MAIT cells from HCV patients taking part in a clinical trial in which they were treated with either sofosbuvir (SOF)+ribavirin (RBV)+pegylated IFN (PEG-IFN), or only SOF+RBV, without PEG-IFN. Interestingly, CD69 expression on MAIT cells was upregulated only in the treatment group receiving PEG-IFN (Fig. 6e), which corresponded with a significantly higher sustained virologic response rate36. High CD69 upregulation was specific to CD161++MAIT cells and not observed on CD161−CD8+ or CD4+ T cells (Supplementary Fig. 9). These data demonstrate that type I IFNs can contribute to MAIT cell activation in vitro and in vivo.
Activated MAIT cells can limit HCV replication
Finally, we explored whether viral MAIT cell activation can have a functional impact on virus replication. We stimulated sorted MAIT cells in a TCR-independent manner (IL-12 and IL-18) and confirmed activation by measurement of IFN-γ secretion (Fig. 7a). The cell supernatants of unstimulated and stimulated MAIT cells were transferred to a hepatocyte line infected with HCV expressing luciferase, allowing measurement of HCV replication. The activated MAIT cell supernatants were able to potently suppress HCV replication, while no activity from control supernatants (Fig. 7b) or from IL-12 and IL-18 (data not shown) addition to hepatocytes was seen. Diluting the activated MAIT cell supernatant reduced the suppression of HCV replication in a dose-dependent manner (Fig. 7c). Addition of anti-IFN-γ reversed the HCV suppression by activated MAIT cell supernatants (Fig. 7d). In an extension of the experimental set up using virally-induced activation to trigger MAIT cells, supernatants from MAIT cells incubated with HCV-treated macrophages reduced HCV replication in the hepatocyte reporter cell line compared to mock-treated macrophages (Supplementary Fig. 10). These results indicate that activation of MAIT cells by viral triggers induces responses that can impact on viral replication in vitro.
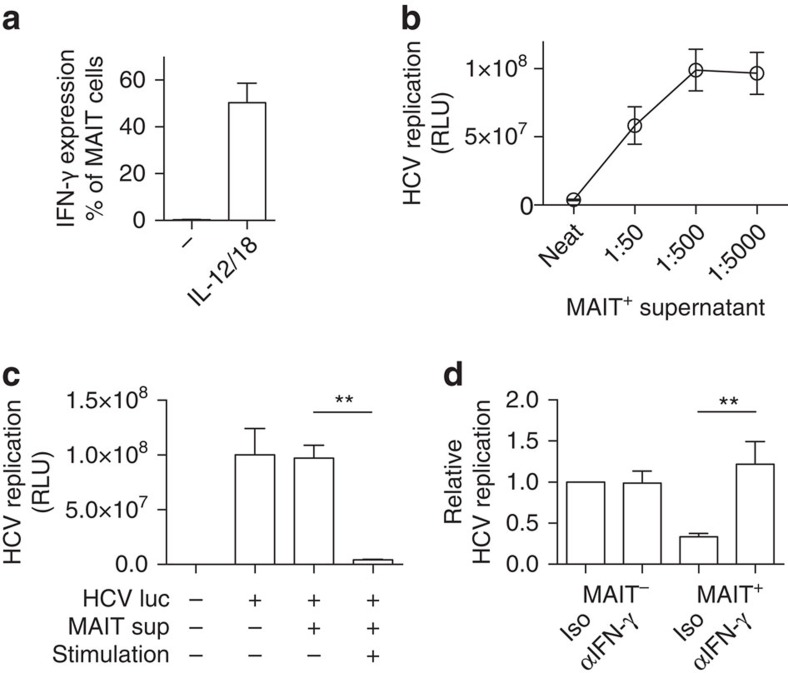
Sorted MAIT cells (CD8+CD161++Vα7.2+) were rested or activated in a TCR-independent manner (IL-12+IL-18 stimulation, MAIT+) for 24 h. (a) IFN-γ expression by MAIT cells (gated on live CD3+CD8+CD161++Vα7.2+ cells) was analysed by flow cytometry. (b,c) Neat or diluted supernatants were transferred to hepatocyte lines infected with HCV expressing luciferase and viral replication measured 4 days post infection. (d) Supernatants were transferred to hepatocyte lines infected with HCV expressing luciferase repeated in the presence of an isotype control or anti-IFN-γ antibody. Data are representative from at least two independent experiments. Bars represent means±s.e.m. Statistical significance was determined with a Wilcoxon matched-paired test (c) or the Mann–Whitney test (d). **P≤0.01. −, unstimulated; luc, luciferase; RLU, relative light units; sup, supernatant.
h. (a) IFN-γ expression by MAIT cells (gated on live CD3+CD8+CD161++Vα7.2+ cells) was analysed by flow cytometry. (b,c) Neat or diluted supernatants were transferred to hepatocyte lines infected with HCV expressing luciferase and viral replication measured 4 days post infection. (d) Supernatants were transferred to hepatocyte lines infected with HCV expressing luciferase repeated in the presence of an isotype control or anti-IFN-γ antibody. Data are representative from at least two independent experiments. Bars represent means±s.e.m. Statistical significance was determined with a Wilcoxon matched-paired test (c) or the Mann–Whitney test (d). **P≤0.01. −, unstimulated; luc, luciferase; RLU, relative light units; sup, supernatant.
Discussion
MAIT cells are an abundant human T-cell population with a semi-invariant TCR determining bacterial specificity. It has previously been considered that MAIT cells were not responsive to viral infection37. However, in this study we provide data to show MAIT cells are also readily and specifically activated by pathogenic viruses in vivo and in vitro. Furthermore, we define mechanisms that allow this virally-driven activation, and show they are distinct in the different viral settings.
The in vivo data shows that there is evidence of MAIT cell activation in response to DENV, HCV and influenza virus. Of these the most striking is the data from acute DENV infection as it is clear that such activation is profound and rapid in this severe clinical setting. Of note, those patients who went on to develop DHF exhibited significantly higher levels of activation as judged by CD38 expression. This suggests the MAIT cell response could contribute to the pathophysiology of this condition. The correlation between levels of IL-18, levels of IL-18R and MAIT cell activation suggests an important role for IL-18 in this activation process, consistent with the in vitro mechanisms demonstrated. In DENV, HCV and influenza virus infection we found clear evidence of activation of the MAIT cells, as judged by upregulation of Granzyme B (which is tightly regulated in MAIT cells22). In addition, we noted a decrease in circulating MAIT cell frequencies. The mechanism for this is not clear. It may result from translocation of some cells from blood to tissues, or it could result from activation-induced cell death as has been suggested for HIV infection38.
The data presented are correlative—however probing a non-redundant role in animal models is not straightforward. In mice the MAIT cell population is a very small minority compared to the frequencies of iNKT cells, which have overlapping functions and innate cytokine responsiveness39. In humans, the situation is reversed, with MAIT cell frequencies exceeding those of iNKT cells by 1–2 logs13. Existing data indicate a functional antiviral role for iNKT cells in control of virus infections in tissues such as lung, in mouse models: given similar molecular mechanisms of activation apply, and similar effector functions are induced, it seems reasonable to extrapolate from this that MAIT cell activity would have a parallel role in human infection40. Emerging models with increased MAIT cell frequencies in mice may help address this question using new in vivo approaches41.
MR1 tetramers unequivocally identify MAIT cells, but these reagents are not yet widely available. In this study we used CD161++Vα7.2+ as a surrogate marker for MAIT cells, which is a commonly used staining approach and includes the majority of tetramer-positive MAIT cells6,42. We further defined MAIT cells as CD8+CD4−CD3+ T cells, and for in vitro experiments PBMC's from healthy individuals were used. CD8 is expressed by the majority of MAIT cells, and CD4 only by a small proportion of MAIT cells42. This approach has been used in recent transcriptional studies and allows ready comparison with related CD161-expressing subsets16. Future studies using MR1 tetramers are needed to further establish the distribution of MAIT cell phenotype in virally infected humans in more detail. As we did not have access to the tetramer, the observed loss of MAIT cells in virally infected patients may be potentially explained by down-regulation of CD161, as previously suggested12,43. However, Fernandez et al.44 showed that in HIV-1 infected patients, MR1 tetramers do not bind CD161−Vα7.2+ T cells, and the loss of MAIT cells in the periphery may be due to recruitment to the site of infection or activation-induced cell death. Of note, our data are not restricted to CD161++Vα7.2+ cells but also includes responses from the minority CD161++Vα7.2− cells, which we have previously described as sharing phenotypic and functional properties with MAIT cells and which likely also include some recently described Vα7.2− MR1-restricted T cell populations45,46.
The mechanisms we have explored reveal a central role for IL-18 in virus-induced activation12,16. However, the critical co-stimulation was not restricted to a single cytokine. In the case of HCV we repeatedly observed an important role for IL-15. IL-15 levels in these experimental conditions were very low, but upregulation of IL-15R was seen and importantly antibody-blockade of IL-15 revealed a clear role for this cytokine. In addition, we observed a role for type I IFNs in the activation of MAIT cells in response to HCV. This was clear in both addition and blockade experiments and is consistent with data from natural killer cells, where such cytokine-mediated activation is described47.
The cytokine activation of MAIT cells was mediated by APCs with a role for viral sensing and replication. In the case of DENV, viral replication was required for MAIT cell activation and this was positively correlated with the level of productive infection in the APC. Although we observed a reduction of MAIT cell activation in response to UV-irradiated HCV and IAV, replication does not have a key role in this context, since HCV and influenza poorly replicate in macrophages28,29. As APC have continual and extensive endocytic activity, viral recognition can occur through endocytosis of viral particles as well as through direct infection26,48. Using TLR agonists, we found that both surface and endocytic TLRs can trigger MAIT cells, in particular viral pathogen-associated molecular patterns.
Using murine bone marrow-derived DCs, Le Bourhis et al.37 has previously shown that MAIT cells are not responsive to viral infection. This discrepancy is likely due to the use of murine, rather than human cells, which may reflect differential abilities in secreting, as well as, responding to cytokines.
IFN-α-mediated activation of MAIT cells is clinically and functionally relevant, as demonstrated in vivo in the well-controlled BOSON clinical trial. In addition to directly acting antiviral drugs (SOF in combination with RBV), IFN-α was included in one arm only and associated with MAIT cell activation and an enhanced therapy response36. It is possible that MAIT cells act as local amplifiers of IFN-α-mediated therapies, contributing to an antiviral effect through secretion of further local antiviral cytokines or potentially via clearance of infected cells. The specific activation by type I IFNs appears to be important evidence in favour of an evolution of MAIT cells in response to viral signals. Although type I IFN signalling is not exclusive to virus infections, a wealth of data links IFN pathways with antiviral defence, and we propose that MAIT cells form an important part of this network in humans. Of note, IFN-α signalling alone was insufficient to fully activate MAIT cells, indicating a potentially fail-safe mechanism to prevent harmful over-activation.
The secondary consequences of MAIT cell activation could be of significance for both protection and immunopathology. Activation of MAIT cells in tissues could provide both direct and indirect antiviral effects. HCV, for example, is known to be highly sensitive to T-cell-derived IFN-γ49,50. Accordingly, we found that activation of MAIT cells by HCV-induced responses that limited HCV replication in vitro, in an IFN-γ dependent manner. Thus, the local activation of MAIT cells can be plausibly correlated with a direct antiviral function in vivo.
Loss of MAIT cell frequencies (as we have observed in blood) may contribute to impairment of both viral and bacterial control, whereas excess cytokine secretion by MAIT cells may lead to immunopathologic outcomes. For example, high levels of MAIT cell activation could contribute to the cytokine ‘storm' associated with DHF and severe cases of influenza virus51,52. Activation-induced loss of MAIT cells is potentially relevant in the context of influenza virus infection, which is epidemiologically associated with secondary bacterial pneumonia due to Staphylococcus aureus or Streptococcus pneumonia53,54. Both of these organisms possess the MAIT cell ligand7,37, and it is plausible that depletion of MAIT cells in situ could lead to transient impairment of local control of these pathogens. Interestingly, the transcription factor PLZF, which likely drives the innate phenotype and functionality of MAIT cells, may also contribute to the induction of activation-induced cell death, through upregulation of specific intracellular caspases55.
In conclusion, human MAIT cells are activated in response to virus infections (as illustrated by a simplified model depicted in Supplementary Fig. 11), through overlapping mechanisms dependent on the virus and likely the environment and cell types affected. These data extend the likely functions of these abundant human lymphocytes well beyond their traditional confines. Modulation of this cell subset (and related CD161+ T-cell subsets16,56,57,58) could provide a novel opportunity to promote antiviral defence or limit immunopathology.
Methods
Patient samples
All samples were collected with appropriate patient consent and local research ethics committee approval. Samples were frozen and stored at −80 °C until examined. Healthy donors were supplied by the NHS blood service without any further donor information. The study on DENV patient samples was approved by the Scientific and Ethical Committee of the Khon Kaen Hospital in Thailand and the Riverside Ethics Committee in the UK (06/Q0401/22). Laboratory confirmation of DENV infection was determined by RT–PCR detection of DENV nucleic acid (which also confirmed the infecting serotype) or seroconversion in an ELISA of IgM. Secondary infection was defined based on the ratio of DENV-specific IgM to IgG <1.2 on or after day 6 of illness. Disease severity was classified according to 1997 World Health Organization criteria. Of the patients enroled in the study, 10 patients were classified as mildly symptomatic of dengue fever and 10 patients were classified as severely symptomatic of dengue haemorrhagic fever with plasma leakage and bleeding (Supplementary Table 1). The day of defervescence was defined as day 0, the day before defervescence as day −1, the day after defervescence as day +1, and so forth. PBMCs were isolated from whole blood by Ficoll-Hypaque density-gradient centrifugation at various time points during hospitalization and cryopreserved until further use.
°C until examined. Healthy donors were supplied by the NHS blood service without any further donor information. The study on DENV patient samples was approved by the Scientific and Ethical Committee of the Khon Kaen Hospital in Thailand and the Riverside Ethics Committee in the UK (06/Q0401/22). Laboratory confirmation of DENV infection was determined by RT–PCR detection of DENV nucleic acid (which also confirmed the infecting serotype) or seroconversion in an ELISA of IgM. Secondary infection was defined based on the ratio of DENV-specific IgM to IgG <1.2 on or after day 6 of illness. Disease severity was classified according to 1997 World Health Organization criteria. Of the patients enroled in the study, 10 patients were classified as mildly symptomatic of dengue fever and 10 patients were classified as severely symptomatic of dengue haemorrhagic fever with plasma leakage and bleeding (Supplementary Table 1). The day of defervescence was defined as day 0, the day before defervescence as day −1, the day after defervescence as day +1, and so forth. PBMCs were isolated from whole blood by Ficoll-Hypaque density-gradient centrifugation at various time points during hospitalization and cryopreserved until further use.
Patients with influenza virus infection requiring hospitalization were recruited from two hospitals based in Oxford and Glasgow during the 2009 H1N1 pandemic under ethical approval (09/H0606/92). Influenza virus infection was diagnosed using an in-house generic influenza A real time PCR assay based on the Matrix gene and confirmed with swine-origin H1N1/09 specific Matrix gene PCR59,60. Patients had concomitant conditions as documented in Supplementary Table 2, but were not on immunosuppressants or steroids at the point of sampling. Two patients were pregnant, and one was 6 days post-partum. PBMC samples from HCV patients (Supplementary Table 3) were collected from the Hepatitis Clinic at the John Radcliffe Hospital, Oxford, UK and consented according to a locally approved protocol (COREC 04.OXA.010). Liver biopsy specimens were obtained from patients with HCV infection (n=55) at S. Bortolo Hospital, Vicenza, Italy and scored using the Ishak system. Control samples were normal adjacent tissue from six uninfected volunteers undergoing liver resection for other reasons (source Proteogenex, CA, USA). Trials were conducted in accordance with the Declaration of Helsinki, ethical approval was obtained from local ethics committees, and all patients provided written informed consent. Explanted HCV-diseased liver tissues were obtained from QE transplant programme and non-diseased normal liver tissue was obtained from donor liver tissue surplus to clinical requirements. Normal liver was liver tissue from hepatic resection of colorectal metastasis. For studying MAIT cell activation in the context of HCV infection and type I IFNs in vivo, PBMC from the BOSON clinical trial were used, as part of a collaboration between STOP-HCV and Gilead Sciences36,61. BOSON is a randomized, open-label, phase 3 study testing SOF plus RBV with or without pegylated IFN-α in patients with HCV genotype 3 and treatment-experienced cirrhotic patients with HCV genotype 2 (Supplementary Table 4). This study was registered with the European Clinical Trials Database, number 2013-002641-11.
Tissue staining
Paraffin-embedded human liver tissues sections were dewaxed using Clearene and IMS. Endogenous peroxidase was blocked by using 5% hydrogen peroxide followed by antigen retrieval using 10% EDTA buffer. Slides were blocked using casein buffer. Primary Rabbit IL-18 antibody LS-B2809, 1:50 dilution was applied for an hour followed by Vector Impress anti-rabbit secondary for 30 min in a staining chamber with rocking. Slides were then washed with PBS+Tween and Impress DAB substrate was applied for 2½ mins. Slides were then counterstained in Mayer's haematoxylin and mounted in DPX.
min in a staining chamber with rocking. Slides were then washed with PBS+Tween and Impress DAB substrate was applied for 2½ mins. Slides were then counterstained in Mayer's haematoxylin and mounted in DPX.
Cell culture
Human PBMCs were isolated from leukocyte cones from healthy donors supplied by the NHS blood service. CD8+ T-cells were isolated from PBMCs using positive selection with MACS CD8+ microbeads (Miltenyi). Monocytes were enriched from PBMCs using MACS CD14+ microbeads (Miltenyi). Monocytes were differentiated into macrophages (macrophages or GM-MΦ), by incubating the CD14+ cells for 7 days in X-VIVO15 (Lonza), Pen/strep (Sigma-Aldrich), L-glutamine (Sigma-Aldrich) with 50 ng
ng ml−1 GM-CSF (Miltenyi). Immature dendritic cells (imDC's or DC's) were obtained by culturing CD14+ cells for 4–5 days in the presence of 20–100
ml−1 GM-CSF (Miltenyi). Immature dendritic cells (imDC's or DC's) were obtained by culturing CD14+ cells for 4–5 days in the presence of 20–100 ng
ng ml−1 GM-CSF (Miltenyi/First Link) and 25
ml−1 GM-CSF (Miltenyi/First Link) and 25 ng
ng ml−1 IL-4 (Miltenyi/eBioscience). Where stated, MAIT cells were sorted using a Beckman Coulter MoFlo XDP.
ml−1 IL-4 (Miltenyi/eBioscience). Where stated, MAIT cells were sorted using a Beckman Coulter MoFlo XDP.
Viruses
DENV serotype 2, strain 16681, was grown in C6/36 cells in Leibovitz's L-15 medium with L-glutamine and supplemented with 2% fetal calf serum. Culture medium was centrifuged and stored at −80 °C. The titres of virus were determined by a focus-forming assay on Vero cells and expressed as focus-forming units per ml. Briefly, virus was serially diluted and incubated with Vero cells for 2
°C. The titres of virus were determined by a focus-forming assay on Vero cells and expressed as focus-forming units per ml. Briefly, virus was serially diluted and incubated with Vero cells for 2 h at 37
h at 37 °C. The monolayers were then overlaid with 1.5% carboxymethylcellulose and incubated at 37
°C. The monolayers were then overlaid with 1.5% carboxymethylcellulose and incubated at 37 °C for 3 days. Virus foci were stained with anti-DENV E antibody (4G2) followed by peroxidase-conjugated anti-mouse Ig and visualized by the addition of DAB substrate. Infection rates of DENV infected DCs were assessed using an antibody detecting the non-structural DENV protein, NS3, by flow cytometry. The mouse monoclonal anti-DENV NS3 (E1D8) and anti-DENV E (4G2) were gifts from E. Harris and AFRIMS.
°C for 3 days. Virus foci were stained with anti-DENV E antibody (4G2) followed by peroxidase-conjugated anti-mouse Ig and visualized by the addition of DAB substrate. Infection rates of DENV infected DCs were assessed using an antibody detecting the non-structural DENV protein, NS3, by flow cytometry. The mouse monoclonal anti-DENV NS3 (E1D8) and anti-DENV E (4G2) were gifts from E. Harris and AFRIMS.
A total of 10 ng RNA transcribed from genotype 2a HCV strain J6CF-JFH1 (obtained from Prof. Bartenschlager62) was electroporated into Huh-7.5 cells (obtained from Apath) and were cultured for up to 3 weeks. Cell culture supernatants were collected for up to 20 days post electroporation centrifuged and concentrated using an Amicon Ultra-15 (Millipore). This inoculum was used to infect huh-7.5 cells. The HCV titre was determined by immunofluorescence. Huh-7.5 cells were fixed with methanolacetone, blocked with BSA/phosphate-buffered saline (PBS) solution, washed with PBS, stained with anti-HCV core primary antibody (Cambridge Biosciences), washed with PBS and Alexa Fluor 488 donkey polyclonal secondary antibody to Mouse IgG (ab150105, Abcam). To measure HCV replication a HCV luciferase reporter Jc1FLAGp7-nsGluc2A29 virus was used. Jc1FLAG(p7-nsGluc2A) was grown in naïve Huh-7.5 cells63 and the infectivity titre was determined by limiting dilution titration on naive Huh-7.5 cells as median tissue culture infective dose (TCID50). To measure HCV replication, Huh-7.5 cells were plated in a 96-well plate and infected at an MOI of 0.1 for 6
ng RNA transcribed from genotype 2a HCV strain J6CF-JFH1 (obtained from Prof. Bartenschlager62) was electroporated into Huh-7.5 cells (obtained from Apath) and were cultured for up to 3 weeks. Cell culture supernatants were collected for up to 20 days post electroporation centrifuged and concentrated using an Amicon Ultra-15 (Millipore). This inoculum was used to infect huh-7.5 cells. The HCV titre was determined by immunofluorescence. Huh-7.5 cells were fixed with methanolacetone, blocked with BSA/phosphate-buffered saline (PBS) solution, washed with PBS, stained with anti-HCV core primary antibody (Cambridge Biosciences), washed with PBS and Alexa Fluor 488 donkey polyclonal secondary antibody to Mouse IgG (ab150105, Abcam). To measure HCV replication a HCV luciferase reporter Jc1FLAGp7-nsGluc2A29 virus was used. Jc1FLAG(p7-nsGluc2A) was grown in naïve Huh-7.5 cells63 and the infectivity titre was determined by limiting dilution titration on naive Huh-7.5 cells as median tissue culture infective dose (TCID50). To measure HCV replication, Huh-7.5 cells were plated in a 96-well plate and infected at an MOI of 0.1 for 6 h. After washing the Huh-7.5 cells, supernatants from (un)stimulated MAIT cells were added in the presence or absence of a neutralizing IFN-γ (clone MD-1, 14-7317-85 eBioscience). Supernatants were collected 4 days post infection and luciferase expression determined using the Renilla Luciferase Assay System (Promega) and a Berthold TriStar2 multimode reader LB 942.
h. After washing the Huh-7.5 cells, supernatants from (un)stimulated MAIT cells were added in the presence or absence of a neutralizing IFN-γ (clone MD-1, 14-7317-85 eBioscience). Supernatants were collected 4 days post infection and luciferase expression determined using the Renilla Luciferase Assay System (Promega) and a Berthold TriStar2 multimode reader LB 942.
Influenza A/WSN/33 (H1N1) virus (WSN) was grown in Madin Darby Bovine Kidney (MDBK, obtained from the European Collection of Cell Cultures) cells in Minimum Essential Medium Eagle (Sigma) with 2 mM L-glutamine and 0.5% fetal calf serum. To produce virus stocks, six 175
mM L-glutamine and 0.5% fetal calf serum. To produce virus stocks, six 175 cm2 tissue culture flasks containing sub-confluent MDBK cells were infected at low-multiplicity. After 48
cm2 tissue culture flasks containing sub-confluent MDBK cells were infected at low-multiplicity. After 48 h of culture in a 37
h of culture in a 37 °C humidified incubator, supernatants were collected and clarified by low-speed centrifugation (30
°C humidified incubator, supernatants were collected and clarified by low-speed centrifugation (30 min, 2,000
min, 2,000 g and 30
g and 30 min, 18,000
min, 18,000 g, 4
g, 4 °C). Next, WSN was concentrated through a 30% sucrose cushion by ultracentrifugation (90
°C). Next, WSN was concentrated through a 30% sucrose cushion by ultracentrifugation (90 min, 112,000
min, 112,000 g, 4
g, 4 °C). Finally, WSN was purified on a 30–60% sucrose gradient by ultracentrifugation (150
°C). Finally, WSN was purified on a 30–60% sucrose gradient by ultracentrifugation (150 min, 209,000
min, 209,000 g, 4
g, 4 °C). The visible band of virus was drawn off with a needle. (Hutchinson, E. and Fodor, E., Nature ProtocolExchange, Purification of influenza virions by haemadsorption and ultracentrifugation, ‘ http://www.nature.com/protocolexchange/protocols/3315')64,65. Plaque assays were performed on MDBK cells using standard techniques. Influenza A (H3N2) virus A/England/691/2010 is a clinical strain isolated by Public Health England (PHE), as part of the MOSAIC project. Influenza B virus, B/Florida/04/2006 was derived from reverse genetics systems by de novo synthesis (GeneArt).
°C). The visible band of virus was drawn off with a needle. (Hutchinson, E. and Fodor, E., Nature ProtocolExchange, Purification of influenza virions by haemadsorption and ultracentrifugation, ‘ http://www.nature.com/protocolexchange/protocols/3315')64,65. Plaque assays were performed on MDBK cells using standard techniques. Influenza A (H3N2) virus A/England/691/2010 is a clinical strain isolated by Public Health England (PHE), as part of the MOSAIC project. Influenza B virus, B/Florida/04/2006 was derived from reverse genetics systems by de novo synthesis (GeneArt).
In vitro virus experiments
Unless specified differently, for co-culture experiments using viruses, APCs were treated with virus at a MOI of 1, as determined based on the viral titre and number of cells plated, for 90 to 120 min at 37
min at 37 °C. Virus was washed off and isolated CD8+ T-cells were added for an additional 10–24
°C. Virus was washed off and isolated CD8+ T-cells were added for an additional 10–24 h, unless specified differently. In the case of the DENV experiments, PBMC's were added instead of isolated CD8+ T-cells. In the case of influenza virus, WSN virus was used, unless specified differently.
h, unless specified differently. In the case of the DENV experiments, PBMC's were added instead of isolated CD8+ T-cells. In the case of influenza virus, WSN virus was used, unless specified differently.
In vitro cytokine stimulations
For cytokine stimulation experiments, isolated CD8+ T-cells or PBMC's were stimulated for 24 h with 50
h with 50 ng
ng ml−1 IL-12 (Miltenyi Biotech), 50
ml−1 IL-12 (Miltenyi Biotech), 50 ng
ng ml−1 IL-18 (R&D Systems Europe), 50
ml−1 IL-18 (R&D Systems Europe), 50 ng
ng ml−1 IL-15 (Miltenyi Biotech) or 2,000
ml−1 IL-15 (Miltenyi Biotech) or 2,000 U
U ml−1 IFN-α (Sigma-Aldrich) or 50
ml−1 IFN-α (Sigma-Aldrich) or 50 ng
ng ml−1 IFN-β (Miltenyi Biotech) or combinations thereof. For blocking experiments, neutralizing agents were added into the culture together with the CD8+ T-cells or PBMC's. Blocking antibodies against IL-12p40/70 (508804, eBioscience), IL-12p70 (MAB219-100, R&D), IL-15 (MAB2471, R&D) or IL-18 (D044-3, MBL International, USA), IFN-γ (14-7317-85, eBioscience) or MR1 (361102, Biolegend) were used at 5–10
ml−1 IFN-β (Miltenyi Biotech) or combinations thereof. For blocking experiments, neutralizing agents were added into the culture together with the CD8+ T-cells or PBMC's. Blocking antibodies against IL-12p40/70 (508804, eBioscience), IL-12p70 (MAB219-100, R&D), IL-15 (MAB2471, R&D) or IL-18 (D044-3, MBL International, USA), IFN-γ (14-7317-85, eBioscience) or MR1 (361102, Biolegend) were used at 5–10 μg
μg ml−1. Type I IFN was blocked using 1
ml−1. Type I IFN was blocked using 1 μg
μg ml−1 B18R (34-8185-81, eBioscience).
ml−1 B18R (34-8185-81, eBioscience).
Flow cytometry
Antibodies/dyes and dilutions used were: viability dye live/dead fixable-violet (L34955, Invitrogen, 1:1250), CD3-eFluor450 (48-0038, eBioscience, 1:100), CD3-PECy7 (25-0038, eBioscience, 1:100), CD4-VioGreen (130-096-900, Miltenyi Biotech, 1:50), CD8-VioGreen (130-098-062, Miltenyi Biotech, 1:50), CD8-V450 (560347, BD, 1:50), CD8-PerCP.Cy5.5/PerCP (301032, Biolegend, 1:100), CD38-APC (555462, BD, 1:50), CD69-FITC (11-0699, eBioscience, 1:40), CD161-APC (130-098-908, Miltenyi Biotech, 1:100), CD161-PE (130-099-193, Miltenyi Biotech, 1:100), IFN-γ-FITC (130-091-641, Miltenyi Biotech, 1:50), IFN-γ-APCCy7 (502529 Biolegend, 1:50), Vα7.2-PE/PeCy7/APC/FITC (351705/351711/351707/351703, Biolegend, 1:50). Granzyme B-APC (MHGB05, Invitrogen), IL-18Ra-APC (17-7183-41, eBioscience, 1:50), TNF-α-PeCy7 (502929, Biolegend, 1:100). All data was acquired on a MACSQuant (Miltenyi Biotech) or a BD FACSVerse (BD) and analyzed on FlowJo (Tree Star Inc.). Gating strategy is shown in Supplementary Fig. 12.
Cytokine measurements
Cytokines levels in this study were measured from cell culture supernatants or heparin plasma samples. Cell culture supernatants from macrophages treated with mock, HCV or influenza A were collected at 48 h. Cell culture supernatants from DCs treated with mock or DENV were collected at 42
h. Cell culture supernatants from DCs treated with mock or DENV were collected at 42 h. Heparin plasma samples were collected from DENV infected patients at different phases of illness or from healthy controls. Cell culture supernatants and heparin plasma samples were frozen at −80̊C until further use. Cytokine levels were analysed using Bio-Plex human cytokine kits (BioRad) and acquired on a Luminex 100 (Luminex) or a Bio-Plex 200 reader (BioRad) according to the manufacturer's instructions.
h. Heparin plasma samples were collected from DENV infected patients at different phases of illness or from healthy controls. Cell culture supernatants and heparin plasma samples were frozen at −80̊C until further use. Cytokine levels were analysed using Bio-Plex human cytokine kits (BioRad) and acquired on a Luminex 100 (Luminex) or a Bio-Plex 200 reader (BioRad) according to the manufacturer's instructions.
Statistical analysis
Statistical analysis was carried out with GraphPad Prism software. Statistical significance was reported as ns P>0.05; *P≤0.05; **P≤0.01; ***P≤0.001; ****P≤0.0001. Error bars on graphs represent s.e.m. Mann–Whitney test was used to calculate significance levels between two groups. Wilcoxon matched-paired test was used to calculate significance levels between two paired groups. For comparisons of means from multiple groups against one control group the Kruskal–Wallis with Dunn's multiple comparison post-test analysis was performed. Spearman-rank correlation analysis was used to calculate correlations. Sample sizes were adequate to detect large effects between groups, as determined by the reproducibility and variability of each particular experiment and limited by the availability of patient samples. No randomization or blinding was used.
Additional information
How to cite this article: van Wilgenburg, B. et al. MAIT cells are activated during human viral infections. Nat. Commun. 7:11653 10.1038/ncomms11653 (2016).
Supplementary Material
Supplementary Figures 1-12 and Supplementary Tables 1-4
Acknowledgments
This research was supported by the NIHR Biomedical Research Program (Oxford), the Wellcome Trust (WT091663MA) and National Institutes for Health Research Biomedical Research Centre (Imperial College), the NIH (U19AI082630) and the Oxford Martin School. I.S. contribution was supported by the Wellcome Trust programme Grant PS2186_WMII. The STOP-HCV consortium is funded by a grant from the Medical Research Council. E.C.H. contribution was supported by MRC programme Grant MR/K000241/1 awarded to Ervin Fodor. L.H. contribution was supported by MRC Project Grant G0600371. Y.H.O. contribution was supported by MRC intermediate fellowship programme. The authors wish to acknowledge the role of the HCV Research UK Biobank in collecting and making available the samples/data used in the generation of this publication. We thank Wendy Barclay for providing Influenza A (H3N2) virus A/England/691/2010 and Influenza B virus, B/Florida/04/2006. We thank Mark Ainsworth for overseeing the HCV patients at the JR hospital. We thank Amin Moghaddam, Jacob Hurst and Leo Swadling for their scientific advice. We also want to acknowledge Nattaya Tangthawornchaikul and Thaneeya Duangchinda.
Footnotes
Author contributions Conceived and designed the experiments: B.W, I.S., P.K., C.W., G.S., J.M.; Performed the experiments: B.W, I.S, C.L, T.L, A.K., C.K., P.F, F.T.; Analyzed the data: B.W., I.S., A.K., C.W.; Contributed to the collection of clinical samples: S.C., S.V., W.L., P.M., D.Y., L.D., STOP-HCV, P.F., M.G., Y.O., L.H.; Contributed other reagents/materials/analysis tools: E.H., N.R., S.M.L., L.B.D., M.D.; Contributed to the writing of this manuscript: P.K., B.W., C.W., G.S., I.S., J.M., L.B.D.
References
- Porcelli S., Yockey C. E., Brenner M. B. & Balk S. P. Analysis of T cell antigen receptor (TCR) expression by human peripheral blood CD4-8- alpha/beta T cells demonstrates preferential use of several V beta genes and an invariant TCR alpha chain. J. Exp. Med. 178, 1–16 (1993). [Europe PMC free article] [Abstract] [Google Scholar]
- Tilloy F. et al.. An invariant T cell receptor alpha chain defines a novel TAP-independent major histocompatibility complex class Ib-restricted alpha/beta T cell subpopulation in mammals. J. Exp. Med. 189, 1907–1921 (1999). [Europe PMC free article] [Abstract] [Google Scholar]
- Dusseaux M. et al.. Human MAIT cells are xenobiotic-resistant, tissue-targeted, CD161hi IL-17-secreting T cells. Blood 117, 1250–1259 (2011). [Abstract] [Google Scholar]
- Treiner E. et al.. Selection of evolutionarily conserved mucosal-associated invariant T cells by MR1. Nature 422, 164–169 (2003). [Abstract] [Google Scholar]
- Tang X.-Z. et al.. IL-7 licenses activation of human liver intrasinusoidal mucosal-associated invariant T cells. J. Immunol. 190, 3142–3152 (2013). [Abstract] [Google Scholar]
- Martin E. et al.. Stepwise development of MAIT cells in mouse and human. PLoS Biol. 7, e54 (2009). [Europe PMC free article] [Abstract] [Google Scholar]
- Kjer-Nielsen L. et al.. MR1 presents microbial vitamin B metabolites to MAIT cells. Nature 491, 717–723 (2012). [Abstract] [Google Scholar]
- Corbett A. J. et al.. T-cell activation by transitory neo-antigens derived from distinct microbial pathways. Nature 509, 361–365 (2014). [Abstract] [Google Scholar]
- Georgel P., Radosavljevic M., Macquin C. & Bahram S. The non-conventional MHC class I MR1 molecule controls infection by Klebsiella pneumoniae in mice. Mol. Immunol. 48, 769–775 (2011). [Abstract] [Google Scholar]
- Chua W.-J. et al.. Polyclonal MAIT Cells have unique innate functions in bacterial infection. Infect. Immun. 80, 3256–3267 (2012). [Europe PMC free article] [Abstract] [Google Scholar]
- Rahimpour A. et al.. Identification of phenotypically and functionally heterogeneous mouse mucosal-associated invariant T cells using MR1 tetramers. J. Exp. Med. 212, 1095–1108 (2015). [Europe PMC free article] [Abstract] [Google Scholar]
- Ussher J. E. et al.. CD161++ CD8+ T cells, including the MAIT cell subset, are specifically activated by IL-12+IL-18 in a TCR-independent manner. Eur. J. Immunol. 44, 195–203 (2014). [Europe PMC free article] [Abstract] [Google Scholar]
- Billerbeck E. et al.. Analysis of CD161 expression on human CD8+ T cells defines a distinct functional subset with tissue-homing properties. Proc. Natl Acad. Sci. USA 107, 3006–3011 (2010). [Europe PMC free article] [Abstract] [Google Scholar]
- Takahashi T., Dejbakhsh-Jones S. & Strober S. Expression of CD161 (NKR-P1A) defines subsets of human CD4 and CD8 T cells with different functional activities. J. Immunol. 176, 211–216 (2006). [Abstract] [Google Scholar]
- Lanier L. L., Chang C. & Phillips J. H. Human NKRP1A. A disulfide-linked homodimer of the C-type lectin superfamily expressed by a subset of NK and T lymphocytes. J. Immunol. 153, 2417–2428 (1994). [Abstract] [Google Scholar]
- Fergusson J. R. et al.. CD161 defines a transcriptional and functional phenotype shared across distinct human T cell lineages. Cell Rep. 9, 1075–1088 (2014). [Europe PMC free article] [Abstract] [Google Scholar]
- Ivanov I. I. et al.. The orphan nuclear receptor RORgammat directs the differentiation program of proinflammatory IL-17+ T helper cells. Cell 126, 1121–1133 (2006). [Abstract] [Google Scholar]
- Singh S. P. et al.. PLZF regulates CCR6 and is critical for the acquisition and maintenance of the Th17 phenotype in human cells. J. Immunol. 194, 4350–4361 (2015). [Europe PMC free article] [Abstract] [Google Scholar]
- Savage A. K. et al.. The transcription factor PLZF directs the effector program of the NKT cell lineage. Immunity 29, 391–403 (2008). [Europe PMC free article] [Abstract] [Google Scholar]
- Gleimer M., von Boehmer H. & Kreslavsky T. PLZF controls the expression of a limited number of genes essential for NKT cell function. Front. Immunol. 3, 374 (2012). [Europe PMC free article] [Abstract] [Google Scholar]
- Patel O. et al.. Recognition of vitamin B metabolites by mucosal-associated invariant T cells. Nat. Commun. 4, 2142 (2013). [Abstract] [Google Scholar]
- Kurioka A. et al.. MAIT cells are licensed through granzyme exchange to kill bacterially sensitized targets. Mucosal Immunol. 8, 429–440 (2015). [Europe PMC free article] [Abstract] [Google Scholar]
- Le Bourhis L., Dusseaux M. & Bohineust A. MAIT cells detect and efficiently lyse bacterially-infected epithelial cells. PLoS Pathog. 9, e1003681 (2013). [Europe PMC free article] [Abstract] [Google Scholar]
- Jo J. et al.. Toll-like receptor 8 agonist and bacteria trigger potent activation of innate immune cells in human liver. PLoS Pathog. 10, e1004210 (2014). [Europe PMC free article] [Abstract] [Google Scholar]
- Sattler A., Dang-Heine C., Reinke P. & Babel N. IL-15 dependent induction of IL-18 secretion as a feedback mechanism controlling human MAIT-cell effector functions. Eur. J. Immunol. 45, 2286–2298 (2015). [Abstract] [Google Scholar]
- Chattergoon M. A. et al.. HIV and HCV activate the inflammasome in monocytes and macrophages via endosomal Toll-like receptors without induction of type 1 interferon. PLoS Pathog. 10, e1004082 (2014). [Europe PMC free article] [Abstract] [Google Scholar]
- Hidmark A. S. et al.. Early alpha/beta interferon production by myeloid dendritic cells in response to UV-inactivated virus requires viral entry and interferon regulatory factor 3 but not MyD88. J. Virol. 79, 10376–10385 (2005). [Europe PMC free article] [Abstract] [Google Scholar]
- Short K. R., Brooks A. G., Reading P. C. & Londrigan S. L. The fate of influenza A virus after infection of human macrophages and dendritic cells. J. Gen. Virol. 93, 2315–2325 (2012). [Abstract] [Google Scholar]
- Marukian S. et al.. Cell culture-produced hepatitis C virus does not infect peripheral blood mononuclear cells. Hepatology 48, 1843–1850 (2008). [Europe PMC free article] [Abstract] [Google Scholar]
- van Wilgenburg B., Browne C., Vowles J. & Cowley S. A. Efficient, long term production of monocyte-derived macrophages from human pluripotent stem cells under partly-defined and fully-defined conditions. PLoS ONE 8, e71098 (2013). [Europe PMC free article] [Abstract] [Google Scholar]
- Stonier S. W. & Schluns K. S. Trans-presentation: a novel mechanism regulating IL-15 delivery and responses. Immunol. Lett. 127, 85–92 (2010). [Europe PMC free article] [Abstract] [Google Scholar]
- Liang Z. et al.. Activation of Toll-like receptor 3 impairs the dengue virus serotype 2 replication through induction of IFN-β in cultured hepatoma cells. PLoS ONE 6, e23346 (2011). [Europe PMC free article] [Abstract] [Google Scholar]
- García-Sastre A. Induction and evasion of type I interferon responses by influenza viruses. Virus Res. 162, 12–18 (2011). [Europe PMC free article] [Abstract] [Google Scholar]
- Castet V. et al.. Alpha interferon inhibits hepatitis C virus replication in primary human hepatocytes infected in vitro. J. Virol. 76, 8189–8199 (2002). [Europe PMC free article] [Abstract] [Google Scholar]
- Symons J. A., Alcamí A. & Smith G. L. Vaccinia virus encodes a soluble type I interferon receptor of novel structure and broad species specificity. Cell 81, 551–560 (1995). [Abstract] [Google Scholar]
- Foster G. R. et al.. Efficacy of sofosbuvir plus ribavirin with or without peginterferon-alfa in patients with HCV genotype 3 infection and treatment-experienced patients with cirrhosis and HCV genotype 2 infection. Gastroenterology 149, 1462–1470 (2015). [Abstract] [Google Scholar]
- Le Bourhis L. et al.. Antimicrobial activity of mucosal-associated invariant T cells. Nat. Immunol. 11, 701–708 (2010). [Abstract] [Google Scholar]
- Cosgrove C. et al.. Early and nonreversible decrease of CD161++ /MAIT cells in HIV infection. Blood 121, 951–961 (2013). [Europe PMC free article] [Abstract] [Google Scholar]
- Reilly E. C., Wands J. R. & Brossay L. Cytokine dependent and independent iNKT cell activation. Cytokine 51, 227–231 (2010). [Europe PMC free article] [Abstract] [Google Scholar]
- Paget C. et al.. Potential role of invariant NKT cells in the control of pulmonary inflammation and CD8+ T cell response during acute influenza A virus H3N2 pneumonia. J. Immunol. 186, 5590–5602 (2011). [Abstract] [Google Scholar]
- Cui Y. et al.. Mucosal-associated invariant T cell-rich congenic mouse strain allows functional evaluation. J. Clin. Invest. 125, 4171–4185 (2015). [Europe PMC free article] [Abstract] [Google Scholar]
- Reantragoon R. et al.. Antigen-loaded MR1 tetramers define T cell receptor heterogeneity in mucosal-associated invariant T cells. J. Exp. Med. 210, 2305–2320 (2013). [Europe PMC free article] [Abstract] [Google Scholar]
- Leeansyah E. et al.. Activation, exhaustion, and persistent decline of the antimicrobial MR1-restricted MAIT-cell population in chronic HIV-1 infection. Blood 121, 1124–1135 (2013). [Europe PMC free article] [Abstract] [Google Scholar]
- Fernandez C. S. et al.. MAIT cells are depleted early but retain functional cytokine expression in HIV infection. Immunol. Cell Biol. 93, 177–188 (2015). [Abstract] [Google Scholar]
- Gherardin N. A. et al.. Diversity of T cells restricted by the MHC class i-related molecule MR1 facilitates differential antigen recognition. Immunity 44, 32–45 (2016). [Abstract] [Google Scholar]
- Fergusson J. R. et al.. CD161 defines a transcriptional and functional phenotype across distinct human T cell lineages. Cell Rep. 9, 1075–1088 (2014). [Europe PMC free article] [Abstract] [Google Scholar]
- Matikainen S. et al.. IFN-alpha and IL-18 synergistically enhance IFN-gamma production in human NK cells: differential regulation of Stat4 activation and IFN-gamma gene expression by IFN-alpha and IL-12. Eur. J. Immunol. 31, 2236–2245 (2001). [Abstract] [Google Scholar]
- Jian Y.-R., Chang S.-Y., Lin P.-Y., Yang Y.-H. & Chuang Y.-H. Inactivated influenza virus vaccine is efficient and reduces IL-4 and IL-6 in allergic asthma mice. Influenza Other Respi. Viruses 7, 1210–1217 (2013). [Europe PMC free article] [Abstract] [Google Scholar]
- Thimme R. Determinants of viral clearance and persistence during acute hepatitis C virus infection. J. Exp. Med. 194, 1395–1406 (2001). [Europe PMC free article] [Abstract] [Google Scholar]
- Guidotti L. G. Viral clearance without destruction of infected cells during acute HBV infection. Science 284, 825–829 (1999). [Abstract] [Google Scholar]
- Pang T., Cardosa M. J. & Guzman M. G. Of cascades and perfect storms: the immunopathogenesis of dengue haemorrhagic fever-dengue shock syndrome (DHF/DSS). Immunol. Cell Biol. 85, 43–45 (2007). [Abstract] [Google Scholar]
- Wang S. et al.. Influenza virus-cytokine-protease cycle in the pathogenesis of vascular hyperpermeability in severe influenza. J. Infect. Dis. 202, 991–1001 (2010). [Europe PMC free article] [Abstract] [Google Scholar]
- Murray R. J. et al.. Community-acquired pneumonia due to pandemic A(H1N1)2009 influenzavirus and methicillin resistant Staphylococcus aureus co-infection. PLoS ONE 5, e8705 (2010). [Europe PMC free article] [Abstract] [Google Scholar]
- Morens D. M., Taubenberger J. K. & Fauci A. S. Predominant role of bacterial pneumonia as a cause of death in pandemic influenza: implications for pandemic influenza preparedness. J. Infect. Dis. 198, 962–970 (2008). [Europe PMC free article] [Abstract] [Google Scholar]
- Gérart S. et al.. Human iNKT and MAIT cells exhibit a PLZF-dependent proapoptotic propensity that is counterbalanced by XIAP. Blood 121, 614–623 (2013). [Europe PMC free article] [Abstract] [Google Scholar]
- Fergusson J. R., Fleming V. M. & Klenerman P. CD161-expressing human T cells. Front. Immunol. 2, 36 (2011). [Europe PMC free article] [Abstract] [Google Scholar]
- Tsai C.-Y. et al.. Type I IFNs and IL-18 regulate the antiviral response of primary human γδ T cells against dendritic cells infected with Dengue virus. J. Immunol. 194, 3890–3900 (2015). [Abstract] [Google Scholar]
- Fergusson J. R. et al.. CD161(int)CD8+ T cells: a novel population of highly functional, memory CD8+ T cells enriched within the gut. Mucosal Immunol. 9, 401–413 (2015). [Europe PMC free article] [Abstract] [Google Scholar]
- Denney L. et al.. Reduction of natural killer but not effector CD8 T lymphocytes in three consecutive cases of severe/lethal H1N1/09 influenza A virus infection. PLoS ONE 5, e10675 (2010). [Europe PMC free article] [Abstract] [Google Scholar]
- Zhao Y. et al.. High levels of virus-specific CD4+ T cells predict severe pandemic influenza A virus infection. Am. J. Respir. Crit. Care Med. 186, 1292–1297 (2012). [Abstract] [Google Scholar]
- Foster G. et al.. Treatment of decompensated HCV cirrhosis in patients with diverse genotypes: 12 weeks sofosbuvir and NS5A inhibitors with/without ribavirin is effective in HCV genotypes 1 and 3. J. Hepatol. 62, (2015). [Google Scholar]
- Pietschmann T. et al.. Construction and characterization of infectious intragenotypic and intergenotypic hepatitis C virus chimeras. Proc. Natl Acad. Sci. USA 103, 7408–7413 (2006). [Europe PMC free article] [Abstract] [Google Scholar]
- Lindenbach B. D. et al.. Complete replication of hepatitis C virus in cell culture. Science 309, 623–626 (2005). [Abstract] [Google Scholar]
- Hutchinson E. C. et al.. Conserved and host-specific features of influenza virion architecture. Nat. Commun. 5, 4816 (2014). [Europe PMC free article] [Abstract] [Google Scholar]
- Hutchinson E. C. et al.. Mapping the phosphoproteome of influenza A and B viruses by mass spectrometry. PLoS Pathog. 8, e1002993 (2012). [Europe PMC free article] [Abstract] [Google Scholar]
Articles from Nature Communications are provided here courtesy of Nature Publishing Group
Citations & impact
Impact metrics
Article citations
Protective effect of TCR-mediated MAIT cell activation during experimental autoimmune encephalomyelitis.
Nat Commun, 15(1):9287, 28 Oct 2024
Cited by: 0 articles | PMID: 39468055 | PMCID: PMC11519641
Riboflavin for women's health and emerging microbiome strategies.
NPJ Biofilms Microbiomes, 10(1):107, 18 Oct 2024
Cited by: 0 articles | PMID: 39420006 | PMCID: PMC11486906
Review Free full text in Europe PMC
MAIT cells: Conserved watchers on the wall.
J Exp Med, 222(1):e20232298, 24 Oct 2024
Cited by: 0 articles | PMID: 39446132 | PMCID: PMC11514058
Review Free full text in Europe PMC
Mucosal-associated invariant T cells are functionally impaired in pediatric and young adult patients following allogeneic hematopoietic stem cell transplantation and their recovery correlates with clinical outcomes.
Haematologica, 109(10):3222-3236, 01 Oct 2024
Cited by: 0 articles | PMID: 38813718
Quantifiable blood TCR repertoire components associate with immune aging.
Nat Commun, 15(1):8171, 17 Sep 2024
Cited by: 0 articles | PMID: 39289351 | PMCID: PMC11408526
Go to all (297) article citations
Other citations
Data
Data behind the article
This data has been text mined from the article, or deposited into data resources.
BioStudies: supplemental material and supporting data
European Clinical Trials
- (1 citation) EU Clinical Trials Register - 2013-002641-11
Similar Articles
To arrive at the top five similar articles we use a word-weighted algorithm to compare words from the Title and Abstract of each citation.
Human liver CD8+ MAIT cells exert TCR/MR1-independent innate-like cytotoxicity in response to IL-15.
J Hepatol, 73(3):640-650, 02 Apr 2020
Cited by: 29 articles | PMID: 32247824
Human mucosal-associated invariant T cells contribute to antiviral influenza immunity via IL-18-dependent activation.
Proc Natl Acad Sci U S A, 113(36):10133-10138, 19 Aug 2016
Cited by: 158 articles | PMID: 27543331 | PMCID: PMC5018778
Activation and Impaired Tumor Necrosis Factor-α Production of Circulating Mucosal-Associated Invariant T Cells in Patients with Trauma.
J Innate Immun, 11(6):506-515, 14 May 2019
Cited by: 3 articles | PMID: 31085907 | PMCID: PMC6758945
MAIT Cells in Respiratory Viral Infections in Mouse and Human.
Crit Rev Immunol, 41(5):19-35, 01 Jan 2021
Cited by: 7 articles | PMID: 35381137 | PMCID: PMC7612767
Review Free full text in Europe PMC
Funding
Funders who supported this work.
Medical Research Council (13)
Biomarkers to predict severity in dengue infection
Professor Gavin Screaton, Imperial College London
Grant ID: G0801508
Immunity in the face of diversity and the development of novel potent HCV vaccines
Professor Eleanor Barnes, University of Oxford
Grant ID: MR/K010239/1
A study of the human antibody response in Dengue haemorrhagic fever
Dr Juthathip Mongkolsapaya, Imperial College London
Grant ID: G0600000
Stratified Medicine to Optimise Treatment for Hepatitis C Virus Infection
Professor Eleanor Barnes, University of Oxford
Grant ID: MR/K01532X/1
Confidence in Concept 2012 - University of Oxford
Professor Alastair Buchan, University of Oxford
Grant ID: MC_PC_12020
Replication of influenza virus at the molecular level
Professor Ervin Fodor, University of Oxford
Grant ID: MR/K000241/1
Death receptors and immune function in health and disease
Professor Gavin Screaton, Imperial College London
Grant ID: G0400720
Respiratory infections
Professor Pablo Murcia, University of Glasgow
Grant ID: MC_UU_12014/9
The dynamic of anti-dengue antibodies over time.
Dr Juthathip Mongkolsapaya, Imperial College London
Grant ID: MR/N012658/1
The role of neutralising antibodies in hepatitis C virus infection
Professor Jane McKeating, University of Birmingham
Grant ID: G0400802
Contribution of NKT cells to modulation of immunepathology in influenza infection
Professor Ling-Pei Ho, University of Oxford
Grant ID: G0600371
Intrahepatic signals involved in the recruitment and differentiation of IL-17 secreting T cells and Regulatory T cells i
Professor Ye Oo, University of Birmingham
Grant ID: G1002552
The role of Hepatitis C virus glycoprotein-receptor polymorphism in viral pathogenesis
Professor Jane McKeating, University of Birmingham
Grant ID: G1100247
NIAID NIH HHS (1)
Grant ID: U19 AI082630
National Institute for Health Research (NIHR) (1)
Grant ID: NF-SI-0515-10005
Wellcome Trust (3)
Function and specificity of T cells in persistent virus infection.
Professor Paul Klenerman, University of Oxford
Grant ID: 091663
Function of novel human T cell subsets in host defence
Professor Paul Klenerman, University of Oxford
Grant ID: 109965/Z/15/Z
Grant ID: WT091663MA




