Abstract
Free full text

Epidemiology and Pathophysiology of COVID-19-Associated Mucormycosis: India Versus the Rest of the World
Abstract
The coronavirus disease 2019 (COVID-19) pandemic has led to a concerning resurgence of mucormycosis. More than 47,000 cases of mucormycosis were reported in three months from India. We update our systematic review on COVID-19-associated mucormycosis (CAM) till June 21st, 2021, comparing cases reported from India and elsewhere. We included individual patient details of 275 cases of CAM, of which 233 were reported from India and 42 from the rest of the world. Diabetes mellitus was the most common underlying risk factor for CAM in India than in other countries. The fatality rate of cases reported from India (36.5%) was less than the globally reported cases (61.9%), probably due to the predominance of rhino-orbital mucormycosis. On a multivariate analysis, we found that pulmonary or disseminated mucormycosis cases and admission to the intensive care unit were associated with increased mortality, while combination medical therapy improved survival. The paucity of pulmonary and disseminated mucormycosis cases from India suggests that these cases were either not diagnosed or reported, further supported by a trend of search data from the Google search engine. In this review, we discuss the factors explaining the substantial rise in cases of CAM. We also propose a hypothetical model describing the epidemiologic triad of CAM.
Supplementary Information
The online version contains supplementary material available at 10.1007/s11046-021-00584-8.
Introduction
The pandemic caused by the newly discovered coronavirus (SARS-CoV-2) continues to rage havoc in several parts of the world, including India. The resulting illness, coronavirus disease (COVID-19), is mostly mild. However, in about 15–20% of the infected individuals, a severe disease characterized by widespread pneumonia and hypoxemia is encountered [1]. The affliction of a large population in a short period has crippled health care globally, especially in developing countries. While we have yet to completely understand the pathogenesis and consequences of SARS-CoV-2 on the human host, different disease complications continue to emerge. The recent surge of COVID-19-associated mucormycosis (CAM) in India is alarming due to its high mortality, the sheer number of cases, and the limited availability of antifungal drugs [2, 3]. India has reported more than 47,000 cases of mucormycosis in three months (May to July 2021), and the actual figures are likely higher. The crisis of CAM was further worsened by an acute scarcity of amphotericin B caused by the large number of cases of mucormycosis [4].
Mucormycosis has always interested the medical community, given the fulminant course and devastating effects on the host. Unfortunately, we understand little about the pathogenesis of CAM. Herein, we update our systematic review of published literature on CAM [2], comparing cases reported from India and elsewhere. We then discuss the possible factors that could explain the tremendous increase in CAM cases. We do not discuss the diagnosis and management of CAM that have been recently reviewed in detail elsewhere [5, 6].
Epidemiology of Cam in India Versus the Rest of the World
Several cases of CAM were reported since our initial publication in the journal [2]. We updated our systematic review (PubMed and Embase databases) till June 21st, 2021, using the term: (“COVID” OR “SARS-CoV” OR “coronavirus”) AND (mucor* OR “zygomycosis”). Only microbiologically or histologically confirmed cases of mucormycosis reported in peer-reviewed publications were included [7]. We arbitrarily defined ‘late CAM’ as mucormycosis diagnosed after seven days of confirmation of COVID-19 (till a maximum of 3 months). The remaining cases where mucormycosis was diagnosed simultaneously or within seven days of COVID-19 were labeled as ‘early CAM.’ We sought individual patient data for the analysis, and when data was not available, we contacted the corresponding author for additional details before exclusion. We extracted the following information from the publications: (1) author and country of publication, (2) age and sex of the subjects, (3) predisposing factors (solid organ transplantation [SOT], hematopoietic stem cell transplant [HSCT], hematological malignancies, diabetes mellitus, and others), (4) details of COVID-19 management, including the presence of hypoxemia, the need for mechanical ventilation, the time to the diagnosis of mucormycosis from symptom onset or confirmation of COVID-19, the dose and duration of glucocorticoids used for COVID-19, and other immunosuppressive agent used, (5) details of mucormycosis, including the site of involvement, details of medical [amphotericin B, isavuconazole, posaconazole, or a combination] and surgical treatment offered, and the (6) case fatality rate of CAM.
We obtained 102 unique citations of peer-reviewed publications after discarding duplicates and identified 2,568 cases (from 39 citations) of COVID-19-associated mucormycosis, with an overall reported mortality of 16.3% (419/2568) [2, 8–43]. We excluded five of these 39 citations, reporting on 2293 cases (3 citations from India [n =
= 2272], one each from Iran [n
2272], one each from Iran [n =
= 10], and Egypt [n
10], and Egypt [n =
= 11]), where individual patient details were not available.[25, 33, 36, 37, 44]. Finally, we included 275 reported cases of CAM, of which 233 were reported from India and 42 from the rest of the world (most commonly from Iran [n
11]), where individual patient details were not available.[25, 33, 36, 37, 44]. Finally, we included 275 reported cases of CAM, of which 233 were reported from India and 42 from the rest of the world (most commonly from Iran [n =
= 19], the USA [n
19], the USA [n =
= 9], and others). The mean age of presentation and sex distribution was not different in India than the global data. The need for supplemental oxygen and mechanical ventilation during COVID-19 were significantly more common in subjects of CAM reported from other countries than India (Table (Table11).
9], and others). The mean age of presentation and sex distribution was not different in India than the global data. The need for supplemental oxygen and mechanical ventilation during COVID-19 were significantly more common in subjects of CAM reported from other countries than India (Table (Table11).
Table 1
Comparison of COVID-19-associated mucormycosis (CAM) reported from India and the rest of the world
India (n = = 233) 233) | Rest of the world (n = = 42) 42) | P value | |
|---|---|---|---|
Age in years (n = = 87) 87) | 56.1 (17–88) | 52.8 (14–86) | 0.14 |
| Male sex | 187/233 (80.3) | 32/42 (76.2) | 0.34 |
| COVID-19 details | |||
| Hypoxemia | 93/158 (58.9) | 37/41 (90.2) | 0.0001 |
| Mechanical ventilation | 68/209 (32.5) | 20/40 (50) | 0.03 |
| Glucocorticoid therapy | 184/229 (80.3) | 27/38 (71.1) | 0.19 |
| Cumulative glucocorticoid dosea | 156.1 (0–1343) | 59.4 (12–121) | 0.05 |
| Indication for corticosteroidsb | 0.001 | ||
| Appropriate | 67/233 (28.8) | 10/19 (52.6) | |
| Inappropriate dose or duration | 52/233 (22.3) | 7/19 (36.8) | |
| Not indicated | 52/233 (22.3) | 2/19 (10.5) | |
| No information or not used | 62/233 (26.6) | 23/42 (54.8) | |
| Tocilizumab | 6/233 (2.6) | 5/42 (11.9) | 0.015 |
| Underlying disease | 0.001 | ||
| COVID-19 only | 64/233 (28.8) | 11/42 (26.2) | |
| Diabetes mellitus | 154/233 (66.1) | 23/42 (54.8) | |
| Hematologic malignancy | 2/233 (0.9) | 4/42 (9.5) | |
| SOT | 5/233 (2.1) | 4/42 (9.5) | |
| Othersc | 8/233 (3.4) | 0 | |
| Diabetic ketoacidosis | 29/211 (13.7) | 2/25 (8) | 0.55 |
| Time to mucormycosis from COVID-19, days | 19.9 (-14 to 101) | 17.2 (-4 to 100) | 0.38 |
Late CAM (> 7 days after COVID-19) 7 days after COVID-19) | 185/232 (79.7) | 27/42 (64.3) | 0.028 |
| Site of CAM | 0.001 | ||
| ROM | 137/233 (58.8) | 20/42 (47.6) | |
| ROCM | 69/233 (29.6) | 7/42 (16.7) | |
| Pulmonary | 17/233 (7.3) | 9/42 (21.4) | |
| Othersd | 10/233 (4.3) | 6/42 (14.3) | |
Organism identified (n = = 41) 41) | 0.0001 | ||
| Rhizopus arrhizus | 13/24 (54.2) | 4/17 (23.5) | |
| Rhizopus spp. | 6/24 (25) | 5/17 (29.4) | |
| Rhizopus microsporus | 1/24 (4.2) | 6/17 (35.3) | |
| Otherse | 4/24 (16.6) | 2/17 (11.8) | |
| Treatment | |||
| Amphotericin | 0.89 | ||
| No amphotericin | 26/233 (11.2) | 5/42 (11.9) | |
| Any formulation | 207/233 (88.8) | 37/42 (88.1) | |
| Posaconazole | 80/204 (39.2) | 12/42 (28.6) | 0.19 |
| Isavuconazole | 19/198 (9.6) | 7/41 (17.1) | 0.16 |
| Surgery | 0.31 | ||
| None | 65/233 (27.9) | 15/42 (35.7) | |
| Complete or partial surgery | 168/233 (72.1) | 27/42 (64.3) | |
| Mortality | 85/233 (36.5) | 26/42 (61.9) | 0.002 |
All the values are presented as mean (range) or numbers/total number of patients [n/N] and percentages
CAM COVID-19-associated mucormycosis, COVID coronavirus disease, L-AMB liposomal amphotericin B, ROM rhino-orbital mucormycosis, ROCM rhino-orbito-cerebral mucormycosis, SOT solid organ transplantation
aDose as dexamethasone equivalent
bIndication was classified as (1) not indicated if glucocorticoids were used in the absence of hypoxemia, (2) appropriate if the dose and duration did not exceed the existing evidence (dexamethasone 6 mg OD [or equivalent] for a duration up to ten days), (3) inappropriate, if the dose or duration exceeded the existing evidence
cOther risk factors included trauma, dental procedure, cirrhosis, and immunosuppressive therapies
dOther sites included disseminated, gastrointestinal, cutaneous, and other site
eOthers included Lichtheimia (n =
= 4), and Mucor spp. (n
4), and Mucor spp. (n =
= 2)
2)
Diabetes mellitus was noted more frequently in the cases reported from India than elsewhere (66.1% vs. 54.8%), while hematological malignancies and organ transplant recipients were more commonly reported from the rest of the world. Mucormycosis was diagnosed after a mean of 19.5 days from the diagnosis of COVID-19, and most cases were late CAM. Twenty-five percent of cases of mucormycosis from India were diagnosed within seven days of COVID-19 (early CAM) compared to nearly 36% from the rest of the world (P =
= 0.028). Overall, rhino-orbital (ROM) and rhino-orbito-cerebral mucormycosis (ROCM) were the most frequent presentation of CAM. ROM and ROCM accounted for 89% of cases in India, while globally, they accounted for only 64% (P
0.028). Overall, rhino-orbital (ROM) and rhino-orbito-cerebral mucormycosis (ROCM) were the most frequent presentation of CAM. ROM and ROCM accounted for 89% of cases in India, while globally, they accounted for only 64% (P =
= 0.001). Pulmonary, disseminated, and other sites were less frequently reported from India.
0.001). Pulmonary, disseminated, and other sites were less frequently reported from India.
The organism responsible for CAM was primarily R. arrhizus, followed by Rhizopus spp. and Rhizopus microsporus. Interestingly, of the seven reported CAM cases by R. microsporus, six were pulmonary (including one disseminated), and the other was a case of cutaneous mucormycosis [2, 14, 20, 28, 31, 43]. Probably, the relatively smaller size of the R. microsporus sporangiospores (compared to R. arrhizus) might have lodged them in the lower airways causing pulmonary infection, which needs to be studied further.
There was no significant difference in the medical or surgical therapy offered to patients from India or elsewhere (Table (Table1).1). The case fatality rate of cases reported from India was significantly less than the globally reported cases (36.5% vs. 61.9%, respectively, P =
= 0.002). We performed a multivariate analysis of possible factors associated with survival in CAM. We found that pulmonary or disseminated mucormycosis cases and admission to the intensive care unit were independently associated with increased mortality (Table (Table2).2). Combination medical therapy (use of amphotericin followed by isavuconazole or posaconazole) was associated with better survival. The reported mortality obtained in our review of literature from India (36.5%) is much lower than the previous Indian data on non-COVID-19 mucormycosis (52%) [45] and a recently reported large study on CAM (45.7%) [3]. The predominance of ROM and timely treatment may partly explain the better survival reported from India. However, a more tenable explanation is the reporting bias and publication bias in these individual case reports.
0.002). We performed a multivariate analysis of possible factors associated with survival in CAM. We found that pulmonary or disseminated mucormycosis cases and admission to the intensive care unit were independently associated with increased mortality (Table (Table2).2). Combination medical therapy (use of amphotericin followed by isavuconazole or posaconazole) was associated with better survival. The reported mortality obtained in our review of literature from India (36.5%) is much lower than the previous Indian data on non-COVID-19 mucormycosis (52%) [45] and a recently reported large study on CAM (45.7%) [3]. The predominance of ROM and timely treatment may partly explain the better survival reported from India. However, a more tenable explanation is the reporting bias and publication bias in these individual case reports.
Table 2
Factors associated with mortality in subjects with COVID-19-associated mucormycosis (CAM)
| Variable | Odds ratio (95% CI) | P value |
|---|---|---|
| Age, in years | 1.018 (0.993–1.043) | 0.17 |
| CAM reported from India | 0.492 (0.207–1.170) | 0.11 |
| Risk factor for mucormycosis | ||
| Diabetes mellitus | Reference category | |
| COVID-19 only | 0.953 (0.472–1.923) | 0.89 |
| Hematological malignancy or organ transplant | 0.620 (0.095–4.042) | 0.62 |
| Others | 3.374 (0.746–15.271) | 0.11 |
| ICU admission or MV for COVID-19 | 2.274 (1.183–4.373) | 0.014 |
| Glucocorticoid usage | 0.832 (0.380–1.823) | 0.65 |
| Days to the diagnosis of mucormycosis from COVID-19 | 0.997 (0.978–1.017) | 0.79 |
| Site of involvement | ||
| Rhino-orbital mucormycosis | Reference category | |
| Rhino-orbito-cerebral mucormycosis | 1.447 (0.332–6.300) | 0.62 |
| Pulmonary | 3.221 (1.009–10.728) | 0.048 |
| Others | 3.299 (1.631–6.671) | 0.001 |
| Surgical therapy for CAM | 0.940 (0.451–1.959) | 0.87 |
| Combination medical therapy for CAM | 0.363 (0.181–0.729) | 0.004 |
CAM COVID-19-associated mucormycosis, CI confidence interval, COVID-19 coronavirus disease, ICU intensive care unit, MV mechanical ventilation
The paucity of pulmonary (Fig. 1) and disseminated mucormycosis cases from India suggests that these cases were either not diagnosed or reported. At the time of publication of our initial report in February 2021 [2], only one additional case of CAM was available from India from the first wave of the COVID-19 pandemic [8]. We believe that there was gross underreporting during the first wave in India. To explore this issue, we analyzed the trend of search data from the Google search engine. We found that the search activity for the term ‘mucormycosis’ had increased significantly, a few weeks following the peak of the first wave of COVID-19 (Fig. 2, blue line). Similarly, the search for ‘mucormycosis’ had also increased tremendously following the second wave. Notably, the search term ‘black fungus’ (misnomer for mucormycosis used by the lay) had shown a similar peak following the second and was not seen in the first wave (Fig. 2). We believe that the medical community had experienced a surge of cases in the first wave too. However, the increase in mucormycosis cases was interpreted as a chance occurrence rather than an outbreak. The widespread media attention and coverage occurred only during the second wave, owing to the tremendous increase in cases.
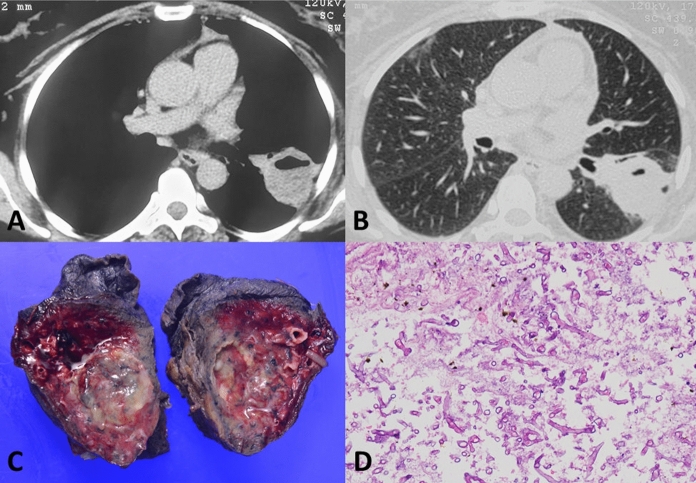
Computed tomography images of a patient with pulmonary mucormycosis (following COVID-19) showing a thick-walled cavity with an air-fluid level in the left lower lobe (A and B). Gross photograph (C) of the resected specimen showing cavity filled with necrotic material. Photomicrograph (D) demonstrating broad aseptate fungal hyphae conforming to the morphology of mucormycosis in a necrotic background (H&E, ×
× 200)
200)
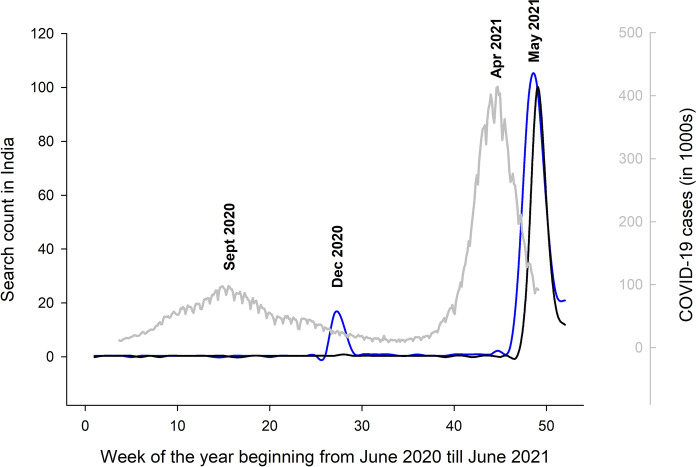
Google search trends from India over one year (52 weeks beginning from June 10th 2020 till June 10th 2021). The vertical axis on the left shows the number of searches in the search engine (obtained from publicly available data). The graph in blue depicts the search count for the term “mucormycosis” in the search engine, while the graph in black color represents the search term “black fungus.” The graphs are superimposed on the number of COVID-19 cases reported from India (in gray color) over the same period. The vertical axis (marked in gray) on the right side of the figure represents the number of COVID-19 cases in thousands
From the systematic review of literature, it appears that there is an unprecedented rise in cases of CAM. The reasons for this surge of mucormycosis cases remain unknown. The ‘epidemiologic triad’ (environment, agent, and host factors) is a practical model to explain the occurrence of a new disease or a re-emerging disease. We discuss the emergence and pathophysiology of CAM using this model.
Environmental Factors
What is Known?
The fungi responsible for mucormycosis belong to the order Mucorales and are saprobes found in decaying matter and soil [46]. The organism rapidly reproduces in the environment, and its spores can become airborne. Despite the fungi being ubiquitous, mucormycosis is mostly encountered as an opportunistic infection in an immune-compromised host [47]. Outbreaks of cutaneous infection by Mucorales have been reported from hospitals associated with contaminated adhesive bandages, linen, and wooden tongue depressors [48]. Less frequently, rhino-orbital or pulmonary mucormycosis has been encountered after exposure to contaminated air (from ongoing construction, contaminated air conditioners, or ventilating systems) [48]. In a study from India, approximately 9% of the mucormycosis (primarily cutaneous) cases were nosocomial [49]. Previous aero-mycological studies from India have demonstrated the presence of pathogenic Mucorales in both hospital and community settings [50, 51]. Rhizopus arrhizus was the most common species identified in this study. Also, the mean spore counts were high in outdoor air samples (0.73 to 8.60 colony-forming unit [CFU]/m3) across different seasons and also in the hospital environment (0.68 to 1.12 CFU/m3 and 0.88 to 1.72 CFU/m3 for air-conditioned and non-air-conditioned wards, respectively) [50].
What Remains to be Explored?
A high environmental burden of fungal spores may rarely cause disease, even in immunocompetent individuals; however, vulnerable populations are at the highest risk. Although several states in India experienced the surge in mucormycosis, some were the worst hit (the western Indian states of Gujarat, Maharashtra, followed by Karnataka, are the maximum affected). In contrast, a few others (eastern states, Bengal, Odisha, and the Northeastern states) have been relatively spared. Whether the number of reported CAM cases is proportionate to the COVID-19 cases (Gujarat and Maharashtra are the worst affected states in this aspect) or the environment in the western Indian states is more favorable for the development of mucormycosis remains unknown.
Construction of new hospital zones and restructuring the existing wards to accommodate the large inflow of patients are commonplace in this pandemic and can potentially result in an outbreak [52]. While construction activities in the hospital setting can make the fungal spores airborne, the occurrence of mucormycosis across a vast geographic area and in non-hospitalized individuals suggests that the spores in the home and outdoor environment or multiple unknown factors may be responsible for CAM.
Agent Factors
What is Known?
The etiologic agent responsible for mucormycosis is a group of thermotolerant eukaryotic fungi of the order Mucorales [47]. The order Mucorales comprises 261 species in 55 genera, at least 38 of which have been associated with human infections [53]. In general, the distribution of agents causing mucormycosis varies depending on the geographic area [54]. Rhizopus arrhizus is responsible for most cases of mucormycosis worldwide, followed by fungi of other genera, including Mucor, Rhizomucor, Lichtheimia, Apophysomyces, Saksenaea, Cunninghamella, and others [55]. Any of the pathogenic Mucorales can cause disease at any anatomical site. The fungal sporangiospores enter the human body mainly by inhalation and less commonly by ingestion or direct inoculation. The large spores (e.g., Rhizopus arrhizus) commonly settle in the upper respiratory tract, while the smaller spores (for instance, Cunninghamella) reach the lower respiratory tract [47]. While Mucorales usually do not cause infection in healthy individuals, the capacity of polymorphonuclear leucocytes may be overwhelmed when the hyphal load is high [56]. Certain Mucorales such as Cunninghamella are known to be highly virulent and proliferate rapidly [57].
What Remains to be Explored?
SARS-CoV-2 is constantly undergoing genetic mutations and variations, whereas the genetic makeup of fungi is generally stable. However, fungi may also acquire virulence factors over time. Notably, a recent study had reported microevolution in Cryptococcus neoformans (both in vivo and in vitro), causing hypervirulence [58]. Genus-specific differences in the interaction of fungi and host immune cells are known in Mucorales, and new virulence factors are being discovered [59, 60]. Whether the Mucorales have acquired hypervirulence factors contributing to the current epidemic needs to be studied. Additionally, the role of SARS-CoV-2 in modulating the interaction between Mucorales and the human host is also not known. The immunomodulatory effects of cytomegalovirus (CMV) contribute to the development of invasive fungal infections (including mucormycosis) in transplant recipients [61–64]. Likewise, mucormycosis cases have also been reported following influenza infections [13].
Host Factors and their Interaction with SARS-CoV-2
What is Known?
Innate immunity is primarily responsible for clearing the spores from mucosal surfaces in healthy individuals. Neutrophilic disorders (qualitative or quantitative) are the main risk factors for mucormycosis [65–69], while lymphocytic disorders have rarely been implicated as a predisposing factor for mucormycosis [70]. The conventional risk factors for invasive mold infections include neutropenia, hematological malignancies, solid organ transplantation, hematopoietic stem cell transplant, immunosuppressive therapies targeting T-cells (calcineurin inhibitors, tumor necrosis factor inhibitors, lymphocyte-specific monoclonal antibodies, prolonged use of corticosteroids at a dose of 0.3 mg/kg for >
> 3 weeks in the past two months), and certain inherited immunodeficiency diseases [7].
3 weeks in the past two months), and certain inherited immunodeficiency diseases [7].
Diabetes Mellitus
Diabetes mellitus is a risk factor for severe COVID-19 and is associated with increased mortality due to COVID-19 [1, 71]. Diabetes impairs innate immune function by impairing phagocytic function, which significantly improves following glycemic control [72, 73]. Further, impaired dendritic cell responses delay the timely activation of adaptive immune responses [74]. Conversely, COVID-19 can lead to the onset of diabetes, and diabetic ketoacidosis (DKA) has been precipitated in newly diagnosed diabetes following COVID-19 [75]. Angiotensin-converting enzyme 2 (ACE2) is found in the lungs, and pancreas serves as the entry receptor for COVID-19. The ACE2 protein allows entry of SARS-CoV-2 into pancreatic islet cells and may injure the beta cells [76, 77]. Also, severe COVID-19 increases insulin resistance through enhanced secretion of stress hormones (cortisol and others) and cytokines [78].
Additionally, poorly controlled diabetes mellitus is the most common risk factor for mucormycosis, both for COVID-19 and non-COVID-19 [2, 3, 55]. Patients with DKA are even more prone to mucormycosis [79]. Hyperglycemia increases glucose-regulated 78 kDa protein (GRP78) expression on the human endothelial cells, the essential receptor for vascular invasion by Mucorales through its spore coat protein (CotH) [80, 81]. Rhizopus also interacts with GRP78 on nasal epithelial cells via CotH3 to invade and damage the nasal epithelial cells [82]. The expression of GRP78 and CotH3 is significantly enhanced by high glucose, iron, and ketones (the hallmark of DKA), potentially causing ROM [82].
Iron Metabolism
Hyperferritinemia, due to the profound inflammation, is a characteristic feature of COVID-19 [83] and is associated with increased mortality [84]. Notably, ferritin-associated iron induces defects in innate (neutrophils) and adaptive immunity (T-lymphocytes) in mice models [85, 86]. Interestingly, the acquisition of iron from the host is essential for the growth of Mucorales [87]. Iron deficiency induces apoptosis in R.arrhizus in an in vitro model [88]. The iron chelator deferasirox protects mice from mucormycosis through iron starvation [89], although not beneficial in patients [90, 91]. Patients with iron overload are more prone to mucormycosis [92], especially those receiving the iron chelator deferoxamine [93, 94]. In patients with DKA, acidosis temporarily dislocates iron bound to transferrin [95, 96]. The ketoacid, β-hydroxybutyrate, indirectly compromises the ability of transferrin to chelate iron [97]. The increased iron can permit the growth of R.arrhizus.
Glucocorticoids
Corticosteroids are an important predisposing factor for CAM. They are potent immunosuppressants with a wide range of effects on various aspects of adaptive and innate immunity [98]. Additionally, hyperglycemia induced by corticosteroids further heightens the risk [99]. Apart from the effects of chronic hyperglycemia on phagocyte and neutrophil functions, even short-term glucocorticoids can precipitate hyperglycemia and have been shown to predispose to mucormycosis [100]. Corticosteroids impair the ability of phagocytes to clear the fungi. In a Drosophila melanogaster model, dexamethasone completely inhibited the phagocytosis of Aspergillus and Rhizopus [101]. Dexamethasone and other corticosteroids are widely used to manage COVID-19, and their contribution to the pathogenesis of CAM appears undeniable [3].
What Remains to be Explored?
SARS-CoV-2 as an Immunomodulatory Virus
The tremendous increase in mucormycosis following the COVID-19 pandemic resembles the re-emergence of rare pathogens such as Pneumocystis following the HIV pandemic. Notably, a significant proportion of individuals with CAM lacked the traditional risk factors for invasive mold diseases [3]. The pathogenesis behind the development of CAM thus remains unknown, and our knowledge on the role of SARS-CoV-2 infection on the cells of the innate immunity (macrophage, neutrophils) is limited. The effects of SARS-CoV-2 on innate immunity need to be explored. Of note, high GRP78 levels have been reported in subjects with COVID-19 [102]. GRP78 expression in sinuses and nasal epithelium and their interaction with CotH3 on the fungal surface have been identified to be important in the pathogenesis of mucormycosis in diabetic mice [80, 103]. While the rampant use of immunosuppressive drugs (particularly glucocorticoids) has fueled the epidemic of CAM, more research is needed to delineate the impact of SARS-CoV-2 on host innate immunity. Adaptive immune defects have been described in COVID-19 [104]. Whether deranged adaptive immunity also contributes to the pathogenesis of CAM remains unclear.
Dose of Glucocorticoids Predisposing to CAM
While glucocorticoids are known to be risk factors for invasive mold infections (both aspergillosis and mucormycosis), the harmful dose and duration remain unclear. In the updated consensus definition of invasive fungal diseases, prolonged use of corticosteroids (defined as a dose of ≥
≥ 0.3 mg/kg corticosteroids for
0.3 mg/kg corticosteroids for ≥
≥ 3 weeks in the preceding 60 days) has been listed as a risk factor [7]. A recent large study suggested that doses and duration beyond the current existing recommendation for COVID-19 (dexamethasone 6 mg up to ten days) were associated with increased risk of CAM [3]. Whether even lesser doses may be harmful or additional unknown factors contribute to CAM needs further research.
3 weeks in the preceding 60 days) has been listed as a risk factor [7]. A recent large study suggested that doses and duration beyond the current existing recommendation for COVID-19 (dexamethasone 6 mg up to ten days) were associated with increased risk of CAM [3]. Whether even lesser doses may be harmful or additional unknown factors contribute to CAM needs further research.
Endothelial Dysfunction
COVID-19 is associated with endothelial dysfunction. Autopsy studies have found severe endothelial injury associated with the presence of intracellular virus and disrupted cell membranes in patients with severe COVID-19 [105]. The vascular endotheliitis may provide an easy route for entry of the Mucorales into the bloodstream, further increasing the risk of complications.
Other Factors
The repurposing of biological agents for COVID-19 treatment, including inhibitors of interleukins (IL-1 and IL-6), tumor necrosis factor, and Janus kinases, may further increase the propensity for infections [106]. The use of several unproven therapies for COVID-19 was commonplace across the world, particularly in India (Figure S1) [107, 108]. The effect of inappropriate use of antibiotics on superadded infections is well known. Staphylococcus aureus and Staphylococcus epidermidis, which are common constituents of the nasal flora, have been shown to inhibit the growth of Rhizopus arrhizus [109, 110]. Inappropriate use of antimicrobial agents (antibacterial and antifungal drugs) may alter the delicate balance of the mycobiome and microbiome of the nasal and respiratory epithelium [111].
Zinc is a crucial element in fungal pathogenesis [112]. During an infection, the human host conceals the available zinc for the fungi to use, thereby conferring a state of ‘nutritional immunity’ [113]. It is possible that the zinc supplementation, particularly in those with acidosis (since some fungal zinc transporters work optimally at acidic pH), may promote the growth of Rhizopus [114–119]. However, no conclusive proof is yet available that zinc therapy has contributed to the tremendous increase in CAM cases.
Another widely discussed factor, albeit unproven in the context of CAM, is steam inhalation. While steam inhalation may offer symptomatic relief in upper respiratory tract infection, the temperature achieved with steam therapy has caused severe inhalational injuries [120]. An ex-vivo study conducted on nasal mucosal and tracheal epithelium showed that the ciliary beat frequency decreases when exposed to temperatures beyond 45 °C and may also result in cell death [121]. Whether such high temperatures decrease the clearance of fungal spores predisposing to rhino-orbital and pulmonary mucormycosis remains unknown.
Finally, industrial oxygen was another widely discussed culprit for the increase in CAM cases. Industrial oxygen is produced like medical-grade oxygen. However, the containers carrying medical oxygen need to comply with certain prescribed norms to not alter the safety, identity, strength, quality, or purity of the oxygen. The impurities in industrial oxygen do not generally refer to microorganisms. High oxygen concentrations do not support the growth of microbes [122], and hyperbaric oxygen has been used as adjunctive therapy in mucormycosis [123]. We do not believe that industrial oxygen led to the surge in cases of CAM.
The Interplay of Host, Environment, and Agent Factors
The occurrence of opportunistic infection is a delicate interplay between the host’s net state of immune suppression and the undue environmental exposure to the causative organism (Fig. 3) [124]. The tropical weather in India is a conducive environment for the growth of Mucoralean spores. The prevalence of diabetes mellitus is also exceptionally high in India (nearly 8% in adults aged ≥
≥ 20 years) [125]. The high numbers of susceptible individuals (diabetes mellitus and COVID-19) have pushed us to the brink of Armageddon. The net state of immunosuppression is increased in COVID-19 individuals because of COVID-19 itself, and its treatment with glucocorticoids (often excessive and inappropriate due to the easy availability of dexamethasone over-the-counter), inappropriate antibiotics, particularly in those with diabetes mellitus, thereby tilting the balance toward the development of invasive mucormycosis.
20 years) [125]. The high numbers of susceptible individuals (diabetes mellitus and COVID-19) have pushed us to the brink of Armageddon. The net state of immunosuppression is increased in COVID-19 individuals because of COVID-19 itself, and its treatment with glucocorticoids (often excessive and inappropriate due to the easy availability of dexamethasone over-the-counter), inappropriate antibiotics, particularly in those with diabetes mellitus, thereby tilting the balance toward the development of invasive mucormycosis.
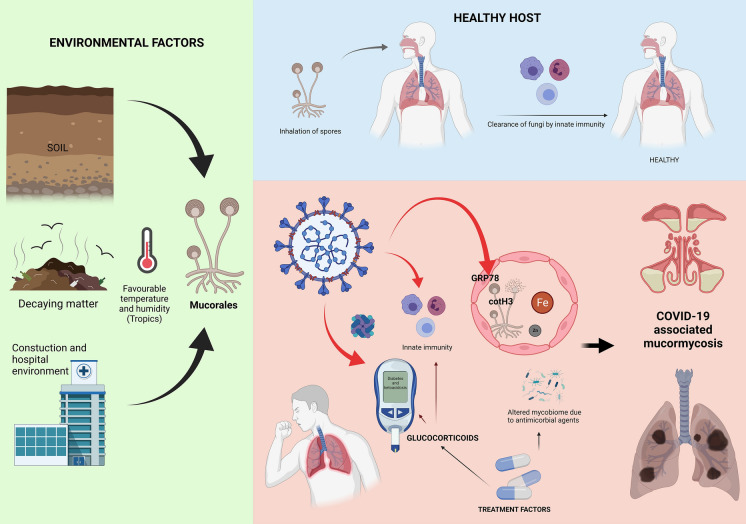
A proposed pathophysiological model of COVID-19-associated mucormycosis. The Mucorales spores are ubiquitous in the environment and are cleared by the innate immunity of a healthy host (Top panel). In a SARS-CoV-2 infected individual, several factors may be involved in the pathogenesis of COVID-19-associated mucormycosis (CAM). Glucose-regulated 78 kDa protein (GRP78) on the human endothelial cells is an essential receptor for vascular invasion by Mucorales through its spore coat protein (CotH3). Hyperglycemia, diabetic ketoacidosis, and the presence of iron favor this interaction and promote the growth of the fungi. SARS-CoV-2 may also be directly (marked by red arrows) increase expression of GRP78, precipitate diabetic ketoacidosis, and possibly alter innate immune cell functions. Additionally, glucocorticoids used in the treatment of COVID-19 cause hyperglycemia and immunosuppression
Conclusions
In conclusion, we believe that systematic data collection and uniform reporting would be the initial step in providing significant clues while analyzing the basis of re-emergence of mucormycosis in the setting of COVID-19. The burden of mucormycosis (non-COVID-19 and COVID-19-associated) is exceptionally high in India and has been neglected for several decades. More research is needed to identify the pathophysiologic basis of CAM, including the role of SARS-CoV-2 on host innate immunity and the interaction with the different species of Mucorales. Other research questions would include the role of hospital outbreaks as a contributor to the increase in cases and the relation of certain species in causing CAM (e.g., R.microsporus in pulmonary mucormycosis following COVID-19).
Supplementary Information
Below is the link to the electronic supplementary material.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
Full text links
Read article at publisher's site: https://doi.org/10.1007/s11046-021-00584-8
Read article for free, from open access legal sources, via Unpaywall:
https://link.springer.com/content/pdf/10.1007/s11046-021-00584-8.pdf
Citations & impact
Impact metrics
Citations of article over time
Alternative metrics

Discover the attention surrounding your research
https://www.altmetric.com/details/112077577
Smart citations by scite.ai
Explore citation contexts and check if this article has been
supported or disputed.
https://scite.ai/reports/10.1007/s11046-021-00584-8
Article citations
Projecting phytochemical bacoside A anti-mucorale agent: An in-silico and in-vitro assessment.
Heliyon, 10(16):e36553, 22 Aug 2024
Cited by: 0 articles | PMID: 39262981 | PMCID: PMC11388571
A Cluster of Diutina catenulata Funguria in Patients with Coronavirus Disease 2019 (COVID-19) Hospitalized in a Tertiary Reference Hospital from Rio de Janeiro, Brazil.
Curr Microbiol, 81(10):338, 03 Sep 2024
Cited by: 0 articles | PMID: 39223407
Epidemiology and prognostic factors of mucormycosis in France (2012-2022): a cross-sectional study nested in a prospective surveillance programme.
Lancet Reg Health Eur, 45:101010, 07 Aug 2024
Cited by: 1 article | PMID: 39220434 | PMCID: PMC11363841
Risk Factors of COVID-19 associated mucormycosis in Iranian patients: a multicenter study.
BMC Infect Dis, 24(1):852, 22 Aug 2024
Cited by: 0 articles | PMID: 39174954 | PMCID: PMC11340102
Dependability and Prognostic Value of Biomarkers in COVID-19 Associated Rhino-Orbito- Cerebral Mucormycosis- A Long Term Ambispective Study.
Indian J Otolaryngol Head Neck Surg, 76(5):4559-4568, 27 Jul 2024
Cited by: 0 articles | PMID: 39376422
Go to all (112) article citations
Data
Data behind the article
This data has been text mined from the article, or deposited into data resources.
BioStudies: supplemental material and supporting data
Similar Articles
To arrive at the top five similar articles we use a word-weighted algorithm to compare words from the Title and Abstract of each citation.
Multicenter Epidemiologic Study of Coronavirus Disease-Associated Mucormycosis, India.
Emerg Infect Dis, 27(9):2349-2359, 04 Jun 2021
Cited by: 235 articles | PMID: 34087089 | PMCID: PMC8386807
Coronavirus disease-associated mucormycosis (CAM): A case control study during the outbreak in India.
J Assoc Physicians India, 70(4):11-12, 01 Apr 2022
Cited by: 1 article | PMID: 35443518
COVID-19-associated mucormycosis in India: Why such an outbreak?
J Mycol Med, 33(3):101393, 09 May 2023
Cited by: 4 articles | PMID: 37182234 | PMCID: PMC10168193
Review Free full text in Europe PMC
Mucormycosis in COVID-19: A systematic review of cases reported worldwide and in India.
Diabetes Metab Syndr, 15(4):102146, 21 May 2021
Cited by: 460 articles | PMID: 34192610 | PMCID: PMC8137376
Review Free full text in Europe PMC





