Abstract
Purpose
Previously, we demonstrated that scleral stem/progenitor cells (SSPCs) from mice have a chondrogenic differentiation potential, which is stimulated by transforming growth factor-β (TGF-β). In the present study, we hypothesized that chondrogenesis in the sclera could be a possible mechanism in myopia development. Therefore, we investigated the association of form-deprivation myopia (FDM) with expressions in mice sclera representing the chondrogenic phenotype: collagen type II (Col2) and α-smooth muscle actin (α-SMA).Methods
The mRNA levels of α-SMA and Col2 in cultured murine SSPCs during chondrogenesis stimulated by TGF-β2 were determined by real-time quantitative RT-PCR (qRT-PCR). The expression patterns of α-SMA and Col2 were assessed by immunohistochemistry in a three dimensional pellet culture. In an FDM mouse model, a western blot analysis and immunofluorescence study were used to detect the changes in the α-SMA and Col2 protein expressions in the sclera. In the RPE-choroid complex, qRT-PCR was used to detect any changes in the TGF-β mRNA expression.Results
The treatment of SSPCs in vitro with TGF-β2 for 24 h at 1 or 10 ng/ml led to increased levels of both the α-SMA and Col2 expressions. In addition, we observed the formation of cartilage-like pellets from TGF-β2-treated SSPCs. Both α-SMA and Col2 were expressed in the pellet. In an in-vivo study, the α-SMA and Col2 protein expressions were significantly increased in the sclera of FDM eyes in comparison to contralateral control eyes. Similarly, the levels of TGF-β in the RPE-choroid complex of an FDM eye were also significantly elevated.Conclusion
Based on the concept of stem cells possessing multipotent differentiation potentials, scleral chondrogenesis induced by SSPCs may play a role in myopia development. The increased expressions of the cartilage-associated proteins Col2 and α-SMA during scleral chondrogenesis may be potential markers for myopia development. In addition, the increased levels of TGF-β mRNA in the RPE-choroid complex might induce the chondrogenic change in the sclera during myopia development.Free full text

Chondrogenesis in scleral stem/progenitor cells and its association with form-deprived myopia in mice
Abstract
Purpose: Previously, we demonstrated that scleral stem/progenitor cells (SSPCs) from mice have a chondrogenic differentiation potential, which is stimulated by transforming growth factor-β (TGF-β). In the present study, we hypothesized that chondrogenesis in the sclera could be a possible mechanism in myopia development. Therefore, we investigated the association of form-deprivation myopia (FDM) with expressions in mice sclera representing the chondrogenic phenotype: collagen type II (Col2) and α-smooth muscle actin (α-SMA).
Methods: The mRNA levels of α-SMA and Col2 in cultured murine SSPCs during chondrogenesis stimulated by TGF-β2 were determined by real-time quantitative RT–PCR (qRT-PCR). The expression patterns of α-SMA and Col2 were assessed by immunohistochemistry in a three dimensional pellet culture. In an FDM mouse model, a western blot analysis and immunofluorescence study were used to detect the changes in the α-SMA and Col2 protein expressions in the sclera. In the RPE-choroid complex, qRT-PCR was used to detect any changes in the TGF-β mRNA expression.
Results: The treatment of SSPCs in vitro with TGF-β2 for 24 h at 1 or 10 ng/ml led to increased levels of both the α-SMA and Col2 expressions. In addition, we observed the formation of cartilage-like pellets from TGF-β2-treated SSPCs. Both α-SMA and Col2 were expressed in the pellet. In an in-vivo study, the α-SMA and Col2 protein expressions were significantly increased in the sclera of FDM eyes in comparison to contralateral control eyes. Similarly, the levels of TGF-β in the RPE-choroid complex of an FDM eye were also significantly elevated.
Conclusion: Based on the concept of stem cells possessing multipotent differentiation potentials, scleral chondrogenesis induced by SSPCs may play a role in myopia development. The increased expressions of the cartilage-associated proteins Col2 and α-SMA during scleral chondrogenesis may be potential markers for myopia development. In addition, the increased levels of TGF-β mRNA in the RPE-choroid complex might induce the chondrogenic change in the sclera during myopia development.
Introduction
Myopia is an important public health issue [1-3]. Recently, the maculopathy of high myopia has become the leading cause of untreatable blindness in East Asia [4-6].
Thinning of the sclera and excessive elongation of the myopic eyeball are accompanied by long-term thinning and stretching of other ocular tissues, especially in the retina and choroid. Consequently, this leads to complications, such as cataracts, glaucoma, retinal detachment, myopic retinal degeneration, visual impairment, and blindness [7-10]. While optical and laser surgical corrective techniques have been used to alter the refractive state of the myopic eye, these therapies do not address the abnormal elongation of the eye and do not treat pathologic changes in the fundus of high myopia patients.
The outer coating of the eyeball, the sclera, becomes pathologically thin in high myopia patients [11]. Although historically the sclera has been considered a relatively inert tissue, the role of the sclera is not simply a static container. Recent research has shown the sclera is a dynamic tissue, capable of responding rapidly to changes in the visual environment to alter ocular size and refraction [12,13]. Its biochemical and biomechanical properties determine the shape and size of the eyeball and therefore play an important role in the determination of refraction status [14]. Scleral extracellular matrix remodeling plays an important role in the enlargement of the ocular globe [15-18]. It has been reported that the development of myopia is controlled by local ocular tissue rather than the central nervous system [19-21]. Even though the mechanism of myopia is still under investigation, it is well known that scleral tissue is the target tissue of myopia [18]. The signal from nearby tissues induces scleral tissue remodeling and can result in eyeball elongation. There are several categories and types of experimental manipulations for an induced myopia model. The most relevant models for understanding the role of vision in common forms of myopia are form deprivation (alterations in retinal image contrast) and lens compensation (focus behind the retina) experiments [22].
In eutherian mammals, the inner layer of cartilage is absent, so the entire sclera consists of a fibrous, collagen type I-dominated (90%) extracellular matrix [23].
In contrast, the scleras of most other vertebrates, including chicks, comprise an inner layer of cartilage and an outer fibrous layer [24]. Recently, we successfully identified scleral stem/progenitor cells (SSPCs) from mouse sclera, as well as demonstrated that SSPCs have a chondrogenic differentiation potential, which is stimulated by transforming growth factor-β (TGF-β) [25]. The expression of cartilage-related markers, such as glycosaminoglycan, aggrecan, and collagen type II (Col2), were all increased in the cartilage-like pellet. The expression of Col2 is considered a hallmark of cartilage differentiation [26]. In addition, Col2 is a major fibrillar collagen of the largely cartilaginous avian sclera and has been identified in the scleras of embryonic mice [27]. On the other hand, it has been suggested that the increased expression of alpha- smooth muscle actin (α-SMA) in scleral tissue is associated with aging and myopia [28-30]. As well, α-SMA is also expressed during chondrogenesis in human mesenchymal stem cells and is considered to have the function of maintaining the integrity of cartilage tissue [31].
Therefore, we hypothesize that SSPCs possessing a chondrogenic differentiation potential may play an important role in myopia development in mammals. In this study, we investigated the association between chondrogenesis and form deprivation myopia (FDM) in mice.
Methods
Mice
Male wild-type C57BL/6 mice (Jackson Labs) were used in this study. All procedures were performed in accordance with an institutional IACUC approved protocol, as well as according to the ARVO Statement for the Use of Animals in Ophthalmic and Vision Research.
Scleral stem/progenitor cell (SSPC) isolation and culture
The SSPCs were isolated and cultured, as previously described [25]. In brief, mouse eyes were obtained and scleras were carefully dissected away from the limbus and optic disc under a dissecting microscope. After the retina and choroid tissues were removed, the scleral tissue was cut into small pieces and digested with 1.5 mg/ml of collagenase type I (Worthington) and 2 mg/ml of Dispase (Roche) in a PBS for 1 h at 37 °C to release individual cells. Individual cells were cultured in α-MEM (Gibco), supplemented with 20% lot-selected FBS (Equitech-Bio), glutamine, penicillin/streptomycin, and 55 μM of 2-mercaptoethanol (Gibco) for 8 to 10 d at 5% CO2 and 37 °C .
TGF-β treatment
In the early passages (3 to 5), 1 × 105 SSPCs were seeded into each well of a 12-well plate. Different concentrations of TGF-β2 were added into the 12 wells of SSPCs. After 24 h, the images of cell morphology were recorded. Then, total RNA was extracted for further analysis. In addition, the same conditions were performed in a chamber slide culture for the immunofluorescence study.
Induction of chondrogenic differentiation
At semi-confluence, the second passage SSPCs were trypsinized and counted to make aliquots of 2×105 cells in 2 ml of the growth medium, and they were spun down at 500 g for 10 min to obtain the pellets, as previously described [25]. The pellets were incubated at 37 °C under 5% CO2. Within 12–24 h of incubation, the cells formed an essentially spherical aggregate that did not adhere to the walls of the tube. The culture medium was added to 10 ng/ml TGF-β2 and the medium was changed at 2- to 3-day intervals. The pellets were then harvested at 4 weeks. Subsequently, they were washed twice in a PBS, fixed in 4% paraformaldehyde for 3 h at room temperature, and prepared for paraffin embedding. Furthermore, 8-μm thick sections were obtained for immunohistochemistry,
Immunohistochemistry and immunofluorescence study
We performed immunohistochemistry and immunofluorescence studies to demonstrate the presence of SMA and the Col2 protein during chondrogenesis. For immunohistochemistry, paraffin sections were treated with a 20% blocking goat serum for 30 min and then incubated with primary antibodies, which were rabbit IgG anti-SMA mAb (1:200 dilution; Abcam, Temecula, CA) and mouse IgG2a anti-type II collagen mAb (1:100 dilution; Abcam) at 4 °C overnight. The sections were then treated with horseradish peroxidase (HRP)-conjugated secondary (1:200; Santa Cruz Biotechnology, Santa Cruz, CA) antibodies for 1 h. The DAB reagent (diaminobenzidine tetrahydrochloride) was subsequently used to detect immunoactivity. For immunofluorescence, cryostat sections and rehydrated paraffin sections were treated with blocking serum, incubated with a primary antibody, reacted with the corresponding fluorescein-isothiocyanate-conjugated secondary antibody, and finally evaluated by fluorescence microscopy.
Real-time PCR
Total RNA from the SSPCs or the choroid tissue in each eye was isolated using Trizol (Invitrogen, Carlsbad, CA) according to the manufacture’s protocol. The qRT-PCR analysis was performed using the iScript one-step RT–PCR kit with SYBR Green (Bio-Rad, Hercules, CA) on an ABI PRISM 7900 HT sequence detection system (Applied Biosystems, Foster City, CA), according to the manufacturer’s instructions. Primers used for the experiment are shown in Table 1. GAPDH and β-actin served as controls. The Ct values of the control gene were subtracted from those of α-SMA and Col2 to provide a semi-quantitative analysis, and the fold change relative to no treatment was assessed.
Table 1
| Primer name | Sequence (5′-3′) |
|---|---|
| α-SMA | F: ATGCCTCTGGACGTACAACTG |
| R: CGGCAGTAGTCACGAAGGAAT | |
| Col2 | F: GTCCTTCTGGCCCTAGAGGT |
| R: TGTTTCTCCTGAGCGTCCA | |
| β-actin | F: CATTGCTGACAGGATGCAGA |
| R: CTGATCCACATCTGCTGGAA | |
| GAPDH | F: AACTTTGGCATTGTGGAAGG |
| R: ACACATTGGGGGTAGGAACA |
Induction of Mouse FDM
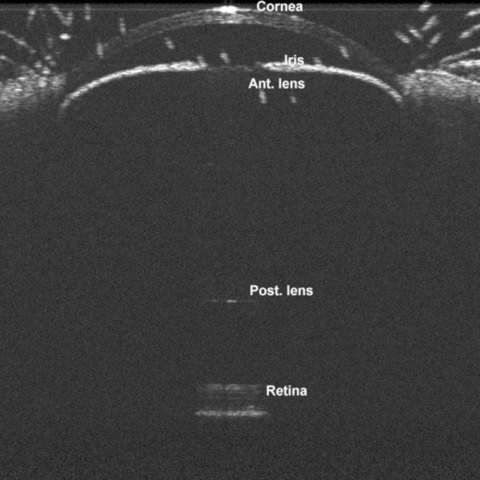
On the day of the experiment (postnatal day [P] 21–24), C57BL/6J mice were anesthetized by an intraperitoneal injection of ketamine (90 mg/kg) and xylazine (10 mg/kg), and diffuser eye patches were sutured to the skin surrounding the right eye with three to six stitches (type: prolene suture; size: 4–0). The left eye served as a control. Hemispherical plastic diffuser eye patches were made from caps of 0.5-ml PCR plastic tubes. Animals were placed on a warming pad during recovery and were monitored until fully mobile. Treated animals were housed in transparent plastic cages under 12 h:12h light-dark conditions (200±15 lx horizontal illuminance) for 21 days. The method of measuring axial length was described by Jiang et al. [32] A spectral-domain optical coherence tomography was used for the ocular biometric measurement before and after FDM induction (Figure 1).
Western blot analysis
The total protein from the scleras was extracted using a RIPA protein extraction buffer. After homogenization of the scleral tissue, the sample was centrifuged and the supernatant was collected. The protein concentration of each sample was measured using a BCATM Protein Assay Kit (Bio-Rad). Scleral protein samples were standardized and electrophoresed on 10% SDS–PAGE gel, then transferred to a polyvinylidene fluoride transfer membrane (Immun-Blot PVDF Membrane, BIO-RAD) at 21 V for 1 h. Membranes were blocked for 1 h at room temperature with 5% dry milk in PBS with 0.1% Tween and incubated at 4 °C overnight with primary antibodies. Membranes were washed and incubated with 1:10,000 goat anti-mouse or anti-rabbit IgG antibodies conjugated to horseradish peroxidase (Santa Cruz) for 1 h at room temperature and washed again. Membranes were developed by chemiluminescence with the reagent Lumigen TMA-6 (GE Healthcare UK limited, Buckinghumshire, UK), and images were captured with the Fujifilm imaging system (LAS-4000; Fujifilm, Tokyo, Japan). Protein bands were quantified using ImageJ software.
Statistical analysis
For in vitro studies, the ANOVA test with a Bonferroni post-hoc test was performed to compare the expressions of α-SMA and Col2 following different concentrations of TGF-β2 treatment. For in vivo studies, the paired t test was used to determine the statistical significance between FDM eyes and contralateral control eyes. A statistical significance was defined as a p value less than 0.05.
Results
Changes in morphology of cultured SSPCs after TGF-β treatment
The cell morphology dramatically changed after TGF-β2 treatment (1–10 ng/ml) for 24 h. In the control and in the 0.1 ng/ml TGF-β2 treatment groups, some SSPCs had the characteristics of a thin spindle shape, and a small portion of cells showed a widened phenotype (Figure 2A,B). In addition, the cytoskeleton filaments of the cells were not obvious (Figure 2E). When the SSPCs were treated with 1 or 10 ng/ml of TGF-β2, nearly all cells became broad and mostly rhombus- or triangle-shaped with prominent cytoskeletal filaments (Figure 2C,D). Immunofluorescence microscopy showed that 10 ng/ml of the TGF-β2 treatment resulted in an increased number of α-SMA protein-positive cells and prominent intracellular α-SMA filament staining (Figure 2F).
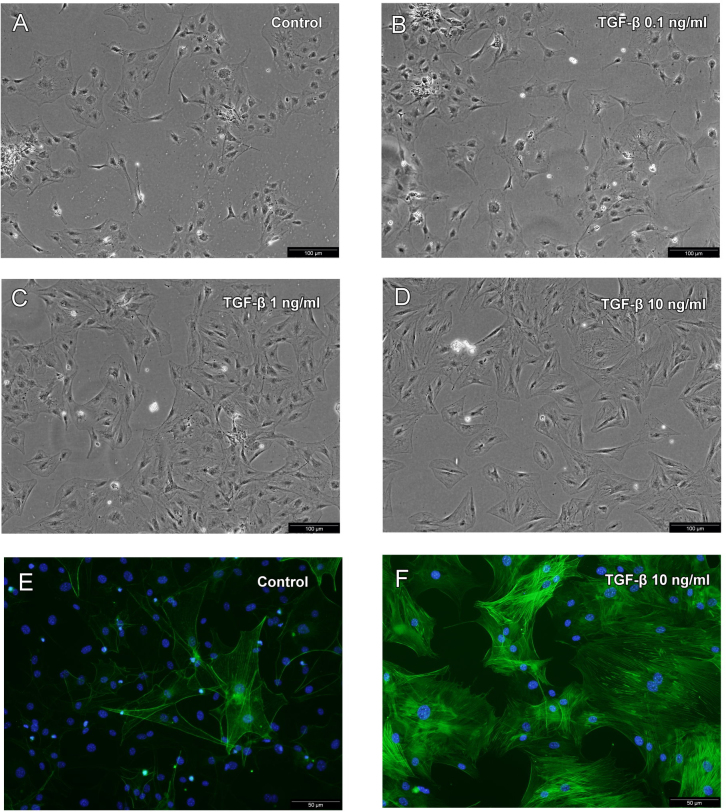
The changes in SSPC morphology following 24 h of treatment with different concentrations of TGF-β. A: Without TGF-β treatment, some SSPCs had the characteristics of thin spindle shapes and some showed a widened phenotype. The cytoskeletal filaments of cells were not obvious (original magnification, ×200). B: Following treatment with 0.1 ng/ml of TGF-β, SSPCs did not change in comparison to no TGF-β treatment. C, D: SSPCs treated with 1 and 10 ng/ml of TGF-β all became broad and mostly rhombus- or triangle-shaped with prominent cytoskeletal filaments. E, F: The expression of the α-SMA protein was determined by immunofluorescence microscopy. Nuclei were stained with DAPI (original magnification, ×400). Representative of three independent experiments.
Effect of TGF-β treatment on α-SMA and Col2 expressions in cultured SPPCs
Next, we examined whether there were any alterations to the α-SMA and Col2 gene expressions after TGF-β2 treatment for 24 h. A quantitative RT–PCR analysis showed there were statistically significant dose-dependent increases in mRNA levels for both α-SMA and Col2 after treatments of TGF-β2 (Figure 3; p<0.0001 and p = 0.011 respectively, ANOVA test).
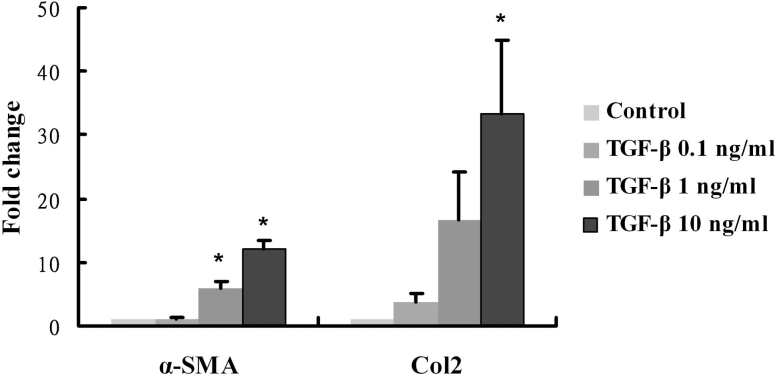
After SSPCs were treated with different concentrations of TGF-β in culture, real-time PCR detected a dose-dependent increase in α-SMA and Col2 mRNA normalized to the β-actin expression. Data are expressed as the fold change over the control sample, as determined by the delta-delta Ct method. Bars, SD * represents statistically significant (p<0.05).
α-SMA and Col2 expressions and localization in 3-D pellets of SSPCs
The cell pellets did not grow in the control medium when cultured for 4 weeks. In contrast, the pellets continued to grow in the medium containing TGF-β2 at 10 ng/ml (TM-pellets) during the 4-week culture period (Figure 4). A histological analysis showed that most cells were located in the peripheral and mid-peripheral areas that surrounded the central matrix tissue in TM pellets. An immunohistochemical analysis showed the Col2 protein localized in a region of the mid-peripheral area of TM pellets (Figure 4C). In contrast, the localization of α-SMA was more extended within the TM pellets, especially in the mid-peripheral and peripheral areas (Figure 4D).
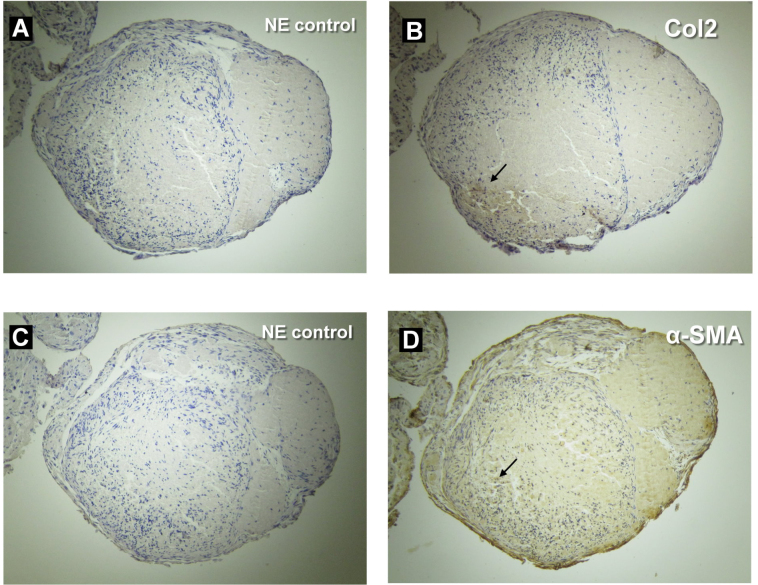
Expressions of the α-SMA and Col2 proteins in the 3-D pellets of SSPCs, as stimulated by TGF-β2. In a culture medium supplemented with 10 ng/ml of TGF-β2, the 3-D pellets grew during the 4-week culturing period. Protein expressions and localization were visualized by a DAB reagent. (A) The negative control without the Col2 antibody. (B) Col2 expressed in the local, mid-peripheral areas of the TM pellets (arrow). (C) The negative control without the α-SMA antibody. (D) The α-SMA expression was more extended within the TM pellets, especially in the mid-peripheral and peripheral areas (arrow).
Mouse FDM
The difference in axial length between the two eyes of each animal was initially insignificant (p = 0.378). However, by the 21st day, monocularly deprived eyes had myopia with an axial length of 3055±39 μm, which was significantly longer than the contralateral control eyes (3015±40 μm, n = 6, paired t test, p<0.001).
Col2 and α-SMA protein in the sclera of mouse FDM
The protein expressions of Col2 and α-SMA were detected by western blot (Figure 5A). After 21 days of visual deprivation, the relative expression levels in FDM eyes were significantly higher than in contralateral control eyes of the same animals for Col2 and α-SMA (p = 0.021 and p = 0.042, respectively, by paired t test, n = 6 in each group, 2 repeats, Figure 5B). Immunostaining for the Col2 expression in the scleral area was greater in the FDM eyes than in the control eyes after 21 days of induction (Figure 6A,B). For the α-SMA expression, a greater expression was noted in the scleral and choroid areas of FDM eyes in comparison to control eyes (Figure 6C,D). In addition, the increased expression of α-SMA in the sclera of FDM eyes was mostly located near the choroid side (Figure 6C).
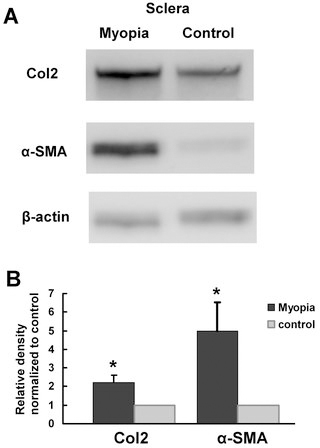
Western blot analysis of Col2 and α-SMA (bands at about 142 kDa and 42 kDa, respectively) in the scleras of mice with form-deprivation myopia (FDM). A: After 21 days of visual deprivation, both the Col2 and α-SMA expression levels were increased. B: A densitometry analysis showed that the Col2 and α-SMA expressions were significantly increased in FDM eyes compared to the control eyes (p<0.05, respectively).
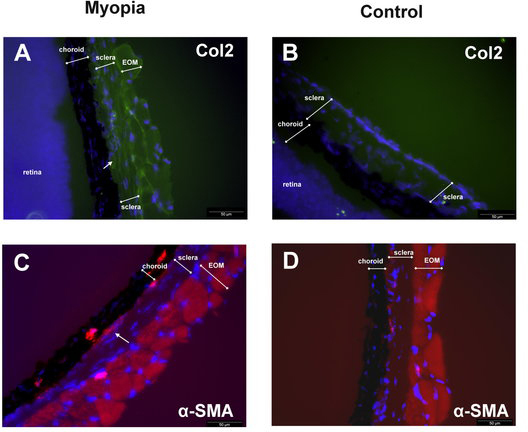
Immunostaining for the Col2 expression in the scleras of FDM mouse eyes on day 21 following induction. Col2 was more highly expressed in the FDM eyes than in the control eyes (Figure 5A,B). For the α-SMA expression, a greater expression was noted in the scleral and choroid areas of FDM eyes in comparison to control eyes. (Figure 5C,D). Some extra-ocular muscle (EOM) tissue existing outside the sclera.
TGF-β mRNA in the choroids of mouse FDM eyes
Due to the α-SMA expression, which was mostly found near the choroid side of the scleras in FDM eyes, the possible influence of the choroid on the sclera was hypothesized, and we investigated the possible changes in TGF-β levels in the choroids of FDM eyes (n = 5 in each group). The relative expression levels of TGF-β1, TGF-β2, and TGF-β3 mRNA in FDM eyes were significantly higher than in contralateral control eyes (2.98, 4.44, and 3.86 fold change, p = 0.042, 0.045, and 0.041, respectively, Figure 7).
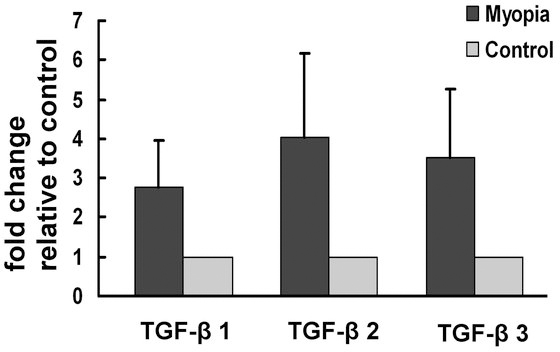
In the RPE-choroid complex of FDM eyes, the levels of the TGF-βs mRNA expression were analyzed by real-time PCR normalized to the β-actin expression. The TGF-β 1, 2, and 3 mRNA levels were all significantly increased in the RPE-choroid complex of FDM eyes in comparison to control eyes. Data are expressed as the fold change over the control sample, as determined by the delta-delta Ct method. Bars, SD * represents statistically significant (Student’s paired t test).
Discussion
In this study, the expression levels of Col2 and α-SMA were investigated in the scleras of mice following induction of FDM. It was found that both Col2 and α-SMA were upregulated in mouse sclera after 21 days of form deprivation. This is the first report on the increased expression of the chondrogenic protein, type II collagen, in a mammalian myopia model. In addition, the increased expression of TGF-β from the RPE-choroid complex in myopic eyes might contribute to chondrogenesis in the sclera.
During myopia development, the sclera’s mechanical properties are altered. Mammalian scleras are composed of approximately 90% collagen by weight, consisting predominantly of collagen type I [23,33,34]. Collagen type I fibrils have enormous tensile strength and are stronger than steel, gram for gram [35]. It has been reported that the expression of collagen type I is decreased in myopic scleras [36]. The elastic properties of the scleras in myopic eyes show an increase in scleral elasticity (creep rate) early in the development of myopia. Our study showed an increase in Col2 in mouse myopic eyes. This may explain why in myopic eyes, following a decrease in collagen type I or replacement by an increase in Col2, the myopic sclera stiffness would decrease, and the elasticity would increase. The myopic eyes would therefore be more susceptible to elongation and an increased axial length.
Chondrogenesis might account for the possible mechanism of myopia development. Col2 has been identified in the scleras of embryonic mice and is a major fibrillar collagen of the cartilaginous avian sclera. Even though little or no Col2 has previously been detected in human scleras [23], there is still some evidence that Col2 plays an important role in human myopia development, as Stickler syndrome type II is a disease with a Col2A1 gene mutation and is associated with myopia [37-39]. In addition, it is difficult to explain why myopia progression would not stop or retard until late adolescence [37-39]. If chondrogenesis is the major contributing mechanism of myopia development in humans, the growth pattern should be similar to other cartilage-associated parts of our body. The bony cartilage (growth plates), for example, disappears at the time of adolescence after a burst of pubertal activity [40]. Articular cartilage, similarly, ceases growth in the early twenties, and it is regulated by growth and sex hormones.
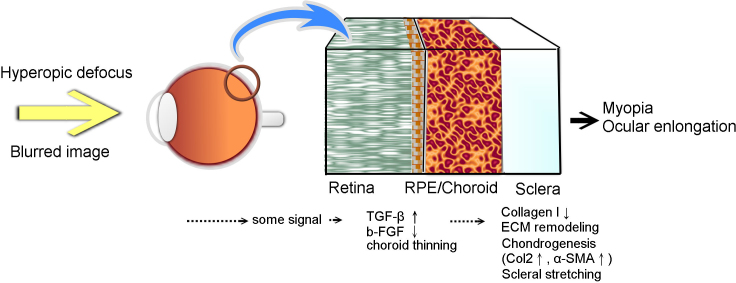
TGF-β plays an important role in myopia development. Several genetic studies showed that the TGF- β gene is associated with myopia [41,42]. In a Marfan syndrome mouse model with TGF- β enhancement, the mice treated with anti-TGF-β antibodies demonstrated a significantly lower axial length [43]. TGF-β has an antagonistic effect on fibroblast growth factor-2 (FGF-2); FGF-2 reduces myopia and TGF-β inhibits the FGF effect on myopia [44-47]. In our study, the increased expression of TGF-β was found in the RPE-choroid complex of FDM eyes, as has also been reported in chickens [46]. McBrien reviewed the role of TGF-β in myopia and highlighted it as a responsible factor for certain critical events in myopia development [48]. However, the levels of TGF-β are decreased in tree shrew scleras but are not significantly changed in the retina and choroid during myopia development [28,49]. Our study showed increased levels of TGF-β in the RPE-choroid complexes of FDM mouse eyes. The possible explanation is that the role of the RPE-choroid complex in myopia development might be due to RPE cells, possessing the ability to express and secrete TGF-β [50,51]. The choroid is the tissue adjacent to the inner sclera and could diffuse soluble proteins to the sclera, especially as the choroid becomes thinned during myopia development [52]. TGF-β is a family of small and soluble proteins. The choroid might play a role in the transportation of TGF-β to the sclera, thus effecting scleral chondrogenesis. This study showed that increased levels of TGF-β in the RPE-choroid complex might be associated with scleral chondrogenesis. Further work to define the origin of TGF-β secretion (i.e., from the RPE or choroid) is needed to elucidate the mechanisms.
From this study, we propose a novel potential mechanism for myopia development. The blurred image imposed on the retina results in a signal transmitted through the choroid to induce choroid thinning and constriction. In conjunction with or following choroidal thinning, the TGF-β expression increases in the RPE-choroid complex and diffuses to the sclera. The SSPCs inside the sclera initiate chondrogenic differentiation induced by TGF-β from the choroid. Then, the scleral changes including collagen type I decrease, and extracellular matrix (ECM) remodeling and chondrogenesis occur in the sclera followed by myopia development (Figure 8). The connective tissue of the sclera is composed by ECM secreted by scleral cells (SSPCs or fibroblasts). ECM molecules previously believed unique to cartilage, such as aggrecan and PRELP, have been identified in human scleras [34,53,54]. Cartilaginous components have been retained in the sclera through evolution and might serve important biochemical and biomechanical functions [12]. Increased aggrecan (cartilage proteoglycan) production in the scleras of myopic chicks and guinea pigs has been reported [44,55]. Therefore, this suggests chondrogenesis in the sclera would result in extracellular matrix changes of scleral remodeling. However, to identify the signaling pathways and the causal relationship, further studies are necessary.
In conclusion, the concept of SSPCs having multipotent differentiation potentials infers scleral chondrogenesis may play a role in myopia development. Increased cartilage-associated proteins, such as Col2 and α-SMA, have the potential to be representative markers in the sclera during myopia development. In addition, increased levels of TGF-β in the RPE-choroid complex might be associated with the chondrogenic change in the sclera during myopia development.
Acknowledgments
This work was partly supported by a grant from National Science Council in Taiwan (NSC 101–2314-B-182A-063) to PW, National Institutes of Health grant R01EY09828 to MEF and an unrestricted grant from Research to Prevent Blindness (RPB) to the University of Southern California.
References
Articles from Molecular Vision are provided here courtesy of Emory University and the Zhongshan Ophthalmic Center, Sun Yat-sen University, P.R. China
Citations & impact
Impact metrics
Citations of article over time
Alternative metrics
Article citations
Association of choroidal thickness and blood flow features with asymmetric axial lengths in children with unilateral myopic anisometropia.
BMC Ophthalmol, 24(1):329, 07 Aug 2024
Cited by: 0 articles | PMID: 39112923 | PMCID: PMC11304658
The human lens is capable of trilineage differentiation towards osteo-, chondro-, and adipogenesis-a model for studying cataract pathogenesis.
Front Bioeng Biotechnol, 11:1164795, 31 May 2023
Cited by: 0 articles | PMID: 37324433 | PMCID: PMC10264667
RNA-sequencing analysis reveals the long noncoding RNA profile in the mouse myopic retina.
Front Genet, 13:1014031, 13 Oct 2022
Cited by: 3 articles | PMID: 36313450 | PMCID: PMC9606684
Choroidal thickness and choriocapillaris vascular density in myopic anisometropia.
Eye Vis (Lond), 8(1):48, 02 Dec 2021
Cited by: 14 articles | PMID: 34857053 | PMCID: PMC8638271
Comparative proteome analysis of form-deprivation myopia in sclera with iTRAQ-based quantitative proteomics.
Mol Vis, 27:494-505, 01 Sep 2021
Cited by: 3 articles | PMID: 34526757 | PMCID: PMC8410231
Go to all (14) article citations
Data
Similar Articles
To arrive at the top five similar articles we use a word-weighted algorithm to compare words from the Title and Abstract of each citation.
Regulation of scleral cell contraction by transforming growth factor-beta and stress: competing roles in myopic eye growth.
J Biol Chem, 284(4):2072-2079, 14 Nov 2008
Cited by: 28 articles | PMID: 19011237
The Role of the RhoA/ROCK Signaling Pathway in Mechanical Strain-Induced Scleral Myofibroblast Differentiation.
Invest Ophthalmol Vis Sci, 59(8):3619-3629, 01 Jul 2018
Cited by: 21 articles | PMID: 30029249
Transforming growth factor-beta in the chicken fundal layers: an immunohistochemical study.
Exp Eye Res, 90(6):780-790, 27 Mar 2010
Cited by: 11 articles | PMID: 20350541
RPE and Choroid Mechanisms Underlying Ocular Growth and Myopia.
Prog Mol Biol Transl Sci, 134:221-240, 23 Jul 2015
Cited by: 62 articles | PMID: 26310157 | PMCID: PMC4755498
Review Free full text in Europe PMC
Funding
Funders who supported this work.
NEI NIH HHS (2)
Grant ID: R01EY09828
Grant ID: R01 EY009828

 1
1